Advanced Synthesis of Rosuvastatin Intermediates: A Scalable Commercial Route
Introduction to Patent CN1821242A
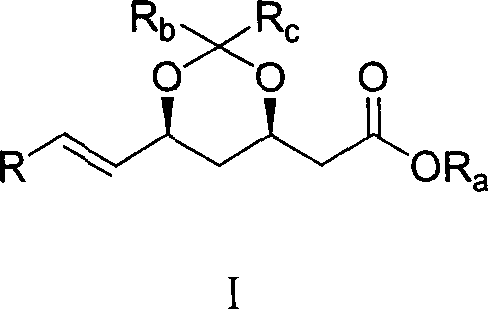
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex therapeutic agents, particularly in the realm of lipid-lowering medications. Patent CN1821242A discloses a novel and highly effective method for the preparation of dihydroxy acid HMG-CoA reductase inhibitors, a class of compounds that includes the widely prescribed super-statin, Rosuvastatin. This technology addresses critical bottlenecks in existing manufacturing processes by introducing a streamlined synthetic route that centers on the preparation of a key unsaturated ester intermediate, designated as Formula (I). By optimizing the coupling of specific side chains with the statin core, this method offers a robust solution for producing high-purity intermediates essential for the final Active Pharmaceutical Ingredient (API). The strategic design of this synthesis not only improves overall yield but also simplifies the operational complexity, making it an attractive option for commercial scale-up.
For procurement specialists and supply chain managers, the implications of this patent are profound, as it represents a shift towards more reliable and cost-effective sourcing of pharmaceutical intermediates. The ability to produce these critical building blocks with fewer steps and under milder conditions directly translates to enhanced supply chain stability and reduced lead times. As the global demand for cardiovascular medications continues to rise, securing a supply partner capable of executing such advanced synthetic methodologies is paramount. This report analyzes the technical merits of this invention, demonstrating how it serves as a cornerstone for the commercial scale-up of complex pharmaceutical intermediates while maintaining the rigorous quality standards required by international regulatory bodies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for HMG-CoA reductase inhibitors have historically been plagued by significant inefficiencies that hinder mass production. Prior art methods often involve lengthy reaction sequences with multiple protection and deprotection steps, each introducing potential yield losses and increasing the accumulation of impurities. Many conventional processes rely on harsh reaction conditions, including extreme temperatures or the use of expensive and toxic transition metal catalysts, which necessitate complex downstream purification to meet safety specifications. These factors collectively drive up the cost of goods sold (COGS) and create vulnerabilities in the supply chain, as any deviation in a long sequence can compromise the entire batch. Furthermore, the environmental footprint of these older methods is often substantial due to the generation of hazardous waste, posing compliance challenges for modern manufacturing facilities striving for greener chemistry.
The Novel Approach
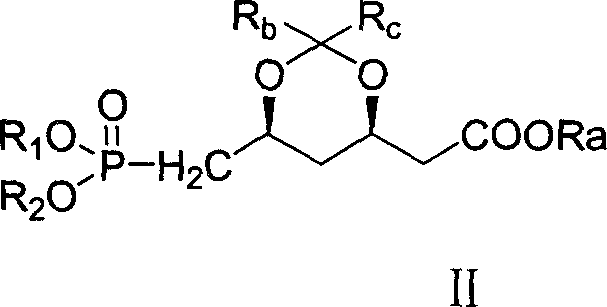
In stark contrast, the method disclosed in CN1821242A introduces a streamlined strategy that effectively bypasses many of these historical hurdles. The core innovation lies in the efficient construction of the carbon-carbon double bond via a coupling reaction between a specific aldehyde and a phosphonate ester, as illustrated by the reaction of Formula (II) with the oxidized side chain. This approach drastically reduces the number of synthetic steps required to reach the key intermediate, thereby minimizing material loss and processing time. By utilizing readily available reagents such as Pyridinium Chlorochromate (PCC) for oxidation and Lithium Chloride (LiCl) for the coupling step, the process avoids the need for exotic catalysts. The result is a synthesis that is not only chemically elegant but also operationally superior, offering a clear pathway for cost reduction in API manufacturing through simplified processing and higher throughput.
Mechanistic Insights into Phosphonate-Mediated Olefination
The heart of this synthetic breakthrough is the precise execution of a Horner-Wadsworth-Emmons (HWE) type olefination, which forms the critical unsaturated linkage in the statin side chain. The process begins with the selective oxidation of a primary alcohol precursor to the corresponding aldehyde using PCC in dichloromethane, a reaction carefully controlled between 0°C and 50°C to prevent over-oxidation to the carboxylic acid. This aldehyde is then subjected to coupling with the phosphonate ester (Formula II) in the presence of LiCl and an organic base like triethylamine. The use of LiCl is particularly noteworthy, as it acts as a Lewis acid additive that can enhance the reactivity and stereoselectivity of the phosphonate anion, ensuring the formation of the desired geometric isomer with high fidelity. This mechanistic precision is vital for maintaining the biological activity of the final drug, as the stereochemistry of the side chain dictates its binding affinity to the HMG-CoA reductase enzyme.
Furthermore, the preservation of chiral integrity throughout this sequence is a testament to the mildness of the reaction conditions. The coupling reaction proceeds efficiently at temperatures ranging from -10°C to 30°C, which prevents the racemization of sensitive chiral centers present in the molecule. Following the coupling, the acetonide protecting group is strategically removed under acidic or basic conditions to reveal the active dihydroxy acid moiety. This step-wise deprotection allows for the isolation of stable intermediates that can be rigorously quality-controlled before final conversion to the calcium salt, such as Rosuvastatin Calcium. By understanding these mechanistic nuances, R&D directors can appreciate the robustness of this route, which minimizes the formation of difficult-to-remove diastereomers and ensures a clean impurity profile suitable for pharmaceutical applications.
How to Synthesize Rosuvastatin Intermediate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters outlined in the patent to ensure maximum efficiency and product quality. The process is designed to be modular, allowing for the independent preparation of the aldehyde and phosphonate components before their convergence in the key coupling step. Operators must maintain anhydrous conditions during the phosphonate formation and coupling stages to prevent hydrolysis of the reactive intermediates, which could otherwise lead to significant yield degradation. The detailed standardized synthetic steps below provide a roadmap for replicating the high yields reported in the patent examples, serving as a foundational guide for process chemists aiming to transfer this technology to pilot or production scales.
- Oxidize the starting alcohol R-CH2OH to the corresponding aldehyde R-CHO using Pyridinium Chlorochromate (PCC) in dichloromethane at 0-50°C.
- React the resulting aldehyde with the phosphonate ester (Formula II) using LiCl and triethylamine in a halogenated solvent at -10 to 30°C to form the unsaturated ester.
- Perform deprotection and hydrolysis using acid or base treatment to yield the final dihydroxy acid or its pharmaceutically acceptable salt, such as Rosuvastatin Calcium.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers transformative benefits for organizations managing the procurement of complex statin intermediates. The elimination of cumbersome reaction steps and the use of commodity chemicals significantly lower the barrier to entry for large-scale production, creating a more competitive pricing environment. For supply chain heads, the robustness of this chemistry means fewer batch failures and a more predictable production schedule, which is critical for maintaining continuous supply to downstream API manufacturers. The ability to source these intermediates from a supplier utilizing this patented route ensures a level of reliability that is often absent in legacy supply chains dependent on fragile, multi-step syntheses.
- Cost Reduction in Manufacturing: The economic impact of this method is driven primarily by the reduction in unit operations and the avoidance of costly noble metal catalysts. By streamlining the synthesis to fewer steps, manufacturers save substantially on solvent consumption, energy usage for heating and cooling, and labor hours required for monitoring and purification. The high yields achieved in each step, particularly the coupling reaction, mean that less raw material is wasted, directly improving the atom economy of the process. These efficiencies compound to offer a significantly lower cost base, allowing procurement teams to negotiate better terms and secure long-term supply agreements at sustainable price points without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials such as substituted pyrimidines and simple phosphonates mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Unlike processes dependent on bespoke reagents with long lead times, this route utilizes chemicals that are readily sourced from multiple vendors, ensuring business continuity even during market fluctuations. The operational simplicity of the reaction conditions further reduces the risk of unplanned downtime due to equipment failure or safety incidents, providing a steady flow of material to meet global demand. This resilience makes the supplier a strategic partner capable of supporting Just-In-Time manufacturing models for major pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from laboratory to industrial tonnage is facilitated by the absence of exothermic hazards and the use of standard reactor materials compatible with halogenated solvents. The reduced generation of heavy metal waste aligns with increasingly stringent environmental regulations, lowering the costs associated with waste disposal and treatment. This eco-friendly profile not only enhances the corporate social responsibility (CSR) standing of the supply chain but also future-proofs the manufacturing site against tightening regulatory frameworks. Consequently, partners can confidently expand production capacity to meet surging market needs for cardiovascular therapeutics without facing significant environmental permitting hurdles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis route. These answers are derived directly from the experimental data and advantageous effects described in the patent documentation, providing clarity for stakeholders evaluating this technology. Understanding these details is crucial for making informed decisions about integrating this intermediate into your broader API manufacturing strategy.
Q: What are the key advantages of this synthesis method over conventional routes?
A: This method significantly reduces reaction steps and operates under mild conditions (0-50°C), avoiding harsh reagents that complicate purification. It achieves high chiral purity and yield, minimizing waste and lowering overall production costs compared to traditional multi-step syntheses.
Q: Is this process suitable for large-scale manufacturing of Rosuvastatin intermediates?
A: Yes, the process utilizes common solvents like dichloromethane and toluene and avoids expensive transition metal catalysts, making it highly scalable. The operational simplicity and high yields demonstrated in the examples support robust commercial-scale production.
Q: How does this method ensure high purity for the final API?
A: The route employs specific protecting group strategies (acetonide) and selective coupling reactions that minimize side-product formation. The final crystallization steps described in the patent ensure the removal of impurities, meeting stringent pharmaceutical quality standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful commercialization of life-saving medications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from development to full-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Rosuvastatin intermediate meets the exacting standards required for global regulatory submission. We are committed to being more than just a vendor; we are a technical ally dedicated to optimizing your supply chain for efficiency and reliability.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this superior route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make a data-driven decision that strengthens your position in the competitive cardiovascular therapy market.
