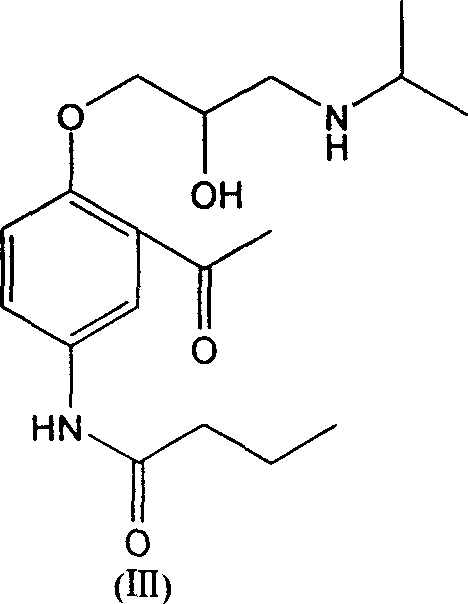
Strategic Upgrade in Acebutolol Intermediate Manufacturing via Liquid-Phase Acylation
The pharmaceutical industry constantly seeks robust synthetic routes for critical beta-blocker intermediates, and patent CN1475478A presents a transformative approach to manufacturing 2-alkanoyl-4-alkanoylaminophenols. This specific class of compounds serves as the foundational backbone for synthesizing Acebutolol, a widely prescribed selective β1 adrenoceptor blocking agent used in managing hypertension and arrhythmias. The traditional reliance on solid-state rearrangements has long plagued manufacturers with inconsistent yields and operational hazards, but this new methodology introduces a streamlined liquid-phase protocol that fundamentally alters the production landscape. By integrating amidation with a concurrent Friedel-Crafts acylation and demethylation sequence, the process achieves exceptional purity profiles while drastically simplifying the engineering requirements for reactor systems. For R&D directors and supply chain leaders, understanding this shift from heterogeneous solid-phase chemistry to homogeneous solution-phase catalysis is essential for securing a competitive edge in the global cardiovascular drug market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
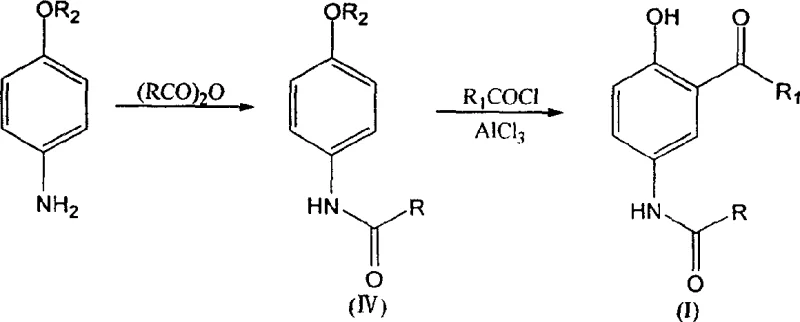
Historically, the synthesis of 2-alkanoyl-4-alkanoylaminophenol derivatives relied heavily on a three-step sequence culminating in a solid-phase Fries rearrangement, a process fraught with significant chemical engineering challenges. In this legacy workflow, the final transformation involves heating a p-alkanoylaminophenol ester with aluminum chloride in a solid state, which creates a highly viscous reaction mass that resists efficient heat transfer and mechanical agitation. This physical limitation leads to severe hot spots within the reactor, promoting uncontrolled side reactions that generate dark-colored impurities and degrade the overall quality of the crude product. Furthermore, the inability to effectively stir the viscous melt restricts the batch size, forcing manufacturers to operate with small, inefficient loads that drive up unit costs and extend production cycles. The cumulative effect of these inefficiencies is a dismal total yield of approximately 57%, alongside a purification burden that requires extensive downstream processing to meet the stringent specifications required for pharmaceutical active ingredients.
The Novel Approach
In stark contrast, the innovative pathway disclosed in the patent data replaces the problematic solid-state rearrangement with a sophisticated two-step liquid-phase strategy that leverages the reactivity of p-aminophenyl alkyl ethers. The first stage involves a clean amidation reaction between the ether and an acid anhydride in a refluxing solvent like ethyl acetate, achieving near-quantitative conversion rates of up to 98% with minimal byproduct formation. The true breakthrough occurs in the second step, where the resulting amide undergoes a simultaneous Friedel-Crafts acylation and demethylation in a halogenated hydrocarbon solvent under the catalysis of anhydrous aluminum chloride. This homogeneous reaction environment ensures uniform temperature distribution and efficient mixing, allowing for precise control over the exothermic acylation process while seamlessly cleaving the methyl ether group to reveal the phenolic hydroxyl. The result is a dramatic improvement in operational safety and product quality, with isolated yields for the final step ranging from 69% to over 83%, depending on the specific alkyl substituents employed.
Mechanistic Insights into AlCl3-Catalyzed Acylation and Demethylation
The core chemical innovation lies in the dual functionality of the aluminum chloride catalyst, which orchestrates both the electrophilic aromatic substitution and the ether cleavage within a single reaction vessel. Mechanistically, the Lewis acidic AlCl3 first complexes with the acid chloride to generate a highly reactive acylium ion, which then attacks the aromatic ring ortho to the alkoxy group due to the directing effects of the ether oxygen. Concurrently, the strong Lewis acidity facilitates the coordination with the ether oxygen, weakening the carbon-oxygen bond and enabling nucleophilic attack by chloride ions or other species present in the medium to effect demethylation. This concerted mechanism bypasses the need for a separate esterification and subsequent high-temperature rearrangement, effectively collapsing two distinct chemical transformations into one thermodynamically favorable process. The choice of halogenated solvents is critical here, as they stabilize the ionic intermediates and maintain the catalyst in solution, preventing the precipitation of inactive complexes that often plague solid-state Lewis acid catalysis.
From an impurity control perspective, this liquid-phase mechanism offers superior selectivity compared to the thermal chaos of the Fries rearrangement. In the traditional solid-phase method, the lack of solvent mediation allows for random radical pathways and polymerization at high temperatures, leading to complex tar formation. However, the solution-phase approach described here maintains the reactants in a molecularly dispersed state, ensuring that the acylium ion reacts primarily at the activated ortho position before any significant degradation can occur. The mild temperature range, spanning from room temperature to 150°C depending on the specific substrate reactivity, further suppresses thermal decomposition pathways. This results in a crude product that is significantly lighter in color and contains fewer structural isomers, thereby reducing the load on crystallization steps and enhancing the overall recovery of high-purity material suitable for subsequent coupling reactions in Acebutolol synthesis.
How to Synthesize 2-Acetyl-4-butyrylaminophenol Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and solvent selection to maximize the efficiency of the demethylation-acylation cascade. The process begins with the protection of the amine functionality via amidation, followed by the critical aluminum chloride-mediated transformation in a chlorinated solvent system. Operators must ensure the gradual addition of the Lewis acid to manage the exotherm effectively, maintaining the reaction mixture within the optimal temperature window to prevent over-chlorination or solvent degradation. While the general principles are straightforward, the precise execution demands adherence to standardized protocols to ensure batch-to-batch consistency and regulatory compliance.
- Perform amidation reaction between p-aminophenyl alkyl ether and acid anhydride in a refluxing solvent such as ethyl acetate.
- Conduct Friedel-Crafts acylation using acid chloride and anhydrous AlCl3 in a halogenated hydrocarbon solvent.
- Execute simultaneous demethylation during the acylation step to generate the final 2-alkanoyl-4-alkanoylaminophenol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel liquid-phase methodology represents a substantial opportunity to optimize the cost structure and reliability of the Acebutolol supply chain. The elimination of the solid-phase Fries rearrangement removes a major bottleneck in production capacity, allowing for larger batch sizes and faster turnaround times without the need for specialized high-torque mixing equipment designed for viscous melts. This operational simplification translates directly into reduced capital expenditure on reactor infrastructure and lower maintenance costs associated with cleaning stubborn, tarry residues from reaction vessels. Furthermore, the significant increase in chemical yield means that less raw material is required to produce the same amount of finished intermediate, providing a direct buffer against volatility in the pricing of starting materials like acid chlorides and anhydrides.
- Cost Reduction in Manufacturing: The shift to a homogeneous liquid-phase reaction eliminates the need for energy-intensive grinding or specialized solid-state heating apparatus, leading to a drastic reduction in utility consumption per kilogram of product. By avoiding the formation of dark, tarry byproducts common in Fries rearrangements, the downstream purification process is streamlined, requiring fewer recrystallization cycles and less solvent for washing, which cumulatively lowers the cost of goods sold. Additionally, the higher selectivity of the reaction minimizes the loss of valuable intermediates to waste streams, ensuring that a greater proportion of input costs are converted into saleable high-purity inventory.
- Enhanced Supply Chain Reliability: The robustness of the liquid-phase process significantly de-risks the manufacturing timeline by removing the variability associated with heat transfer in viscous solid masses. This consistency allows for more accurate production forecasting and shorter lead times, as the reaction endpoints are sharper and easier to monitor via standard analytical techniques like HPLC or TLC. The use of common halogenated solvents and readily available Lewis acids ensures that the supply of critical reagents remains stable, preventing disruptions that could arise from sourcing niche catalysts or specialized equipment parts required for legacy solid-phase reactors.
- Scalability and Environmental Compliance: Scaling this process from pilot to commercial production is inherently safer and more predictable due to the fluid nature of the reaction mixture, which facilitates efficient heat removal and prevents thermal runaway scenarios. The ability to recover and recycle solvents like ethyl acetate and chloroform through standard distillation columns aligns with modern green chemistry initiatives, reducing the volume of hazardous waste generated per ton of product. This improved environmental profile not only lowers disposal costs but also simplifies regulatory compliance audits, making the facility more resilient to increasingly stringent environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route for 2-alkanoyl-4-alkanoylaminophenols. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification.
Q: What is the primary advantage of this new method over the traditional Fries rearrangement?
A: The new method utilizes a liquid-phase reaction system instead of a difficult-to-control solid-phase Fries rearrangement. This eliminates issues with viscosity, heat transfer, and stirring, resulting in significantly higher yields (up to 83% in the second step versus a total of 57% previously) and superior product purity.
Q: Which solvents are recommended for the acylation and demethylation step?
A: The patent specifies the use of halogenated hydrocarbon solvents such as dichloromethane, chloroform, carbon tetrachloride, or various dichloroethanes. These solvents facilitate the homogeneous reaction conditions required for the simultaneous acylation and demethylation catalyzed by aluminum chloride.
Q: How does this process impact the scalability of Acebutolol intermediate production?
A: By shifting from a solid-state reaction to a solution-phase process, the method allows for much larger batch sizes with precise temperature control. The elimination of viscous reactant masses ensures consistent mixing and safety, making commercial scale-up from pilot plants to multi-ton production far more feasible and reliable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Acetyl-4-butyrylaminophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of cardiovascular therapeutics depends on the availability of high-quality intermediates produced via scalable and robust processes. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical supply chains. We are committed to delivering 2-acetyl-4-butyrylaminophenol with stringent purity specifications, utilizing advanced analytical capabilities in our rigorous QC labs to verify that every batch meets the exacting standards required for API synthesis. Our facility is equipped to handle the specific solvent systems and catalytic conditions outlined in this patent, guaranteeing a consistent supply of this critical building block.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to this liquid-phase method for your Acebutolol production. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to validate the superior quality and reliability of our intermediates before committing to large-scale orders.
