Scalable Synthesis of Novel Chromen-2-one Derivatives for High-Purity Pharmaceutical Intermediates
Scalable Synthesis of Novel Chromen-2-one Derivatives for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust synthetic routes that balance molecular complexity with manufacturing feasibility. Patent CN101679407B introduces a significant advancement in the preparation of novel chromen-2-one derivatives, specifically targeting the synthesis of monoamine neurotransmitter reuptake inhibitors. This technology addresses a critical bottleneck in medicinal chemistry: the transition from milligram-scale discovery to kilogram-scale production without compromising purity. By utilizing a 2,2,2-trichloroethyl carbamate protecting group strategy, the invention enables the preparation of crystalline intermediates that can be isolated without the need for expensive and time-consuming chromatographic purification. For R&D directors and process chemists, this represents a paradigm shift towards more efficient, scalable, and cost-effective manufacturing of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (8-H-8-aza-bicyclo[3.2.1]oct-3-yloxy)-benzopyran-2-one derivatives, as disclosed in prior art such as WO 2006/035034, relied heavily on methyl or tert-butoxycarbonyl (t-Boc) protecting groups. While effective for small-scale laboratory synthesis, these traditional methods present substantial challenges when applied to industrial manufacturing. The removal of methyl groups often necessitates harsh demethylation conditions involving strong Lewis acids or nucleophiles, which can degrade sensitive functional groups on the benzopyran core. Furthermore, t-Boc deprotection typically generates isobutylene gas and requires acidic conditions that may lead to salt formation issues or require extensive work-up procedures. Most critically, these conventional routes frequently yield oils or amorphous solids that demand column chromatography for purification, a technique that is economically unviable and operationally difficult to implement on a multi-kilogram or ton scale.
The Novel Approach
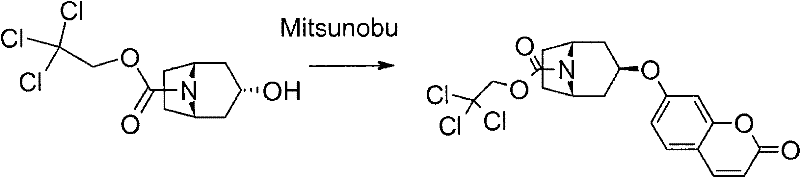
The methodology described in CN101679407B overcomes these limitations through the strategic implementation of a 2,2,2-trichloroethyl (Troc) protecting group on the azabicyclic nitrogen. This approach facilitates a highly stereoselective Mitsunobu coupling reaction, converting the endo-alcohol starting material into the desired exo-ether configuration with high fidelity. The Troc group provides exceptional stability under the basic conditions of the Mitsunobu reaction while remaining susceptible to mild reductive cleavage using zinc dust. This dual characteristic ensures that the intermediate remains intact during coupling but can be cleanly removed in the final step to reveal the free amine. The result is a process that yields crystalline products at multiple stages, allowing for purification via simple filtration and washing rather than chromatography.
Mechanistic Insights into Zinc-Mediated Deprotection and Stereochemical Control
The core of this synthetic innovation lies in the mechanistic interplay between the Mitsunobu reaction and the subsequent zinc-mediated deprotection. In the first key transformation, the endo-3-hydroxy-8-aza-bicyclo[3.2.1]octane derivative undergoes inversion of configuration. The Mitsunobu reagents, typically triphenylphosphine and an azodicarboxylate like DEAD, activate the hydroxyl group, allowing the phenolic oxygen of the hydroxycoumarin to attack from the opposite face. This stereospecific SN2-type displacement is crucial for establishing the correct pharmacophore geometry required for biological activity. The presence of the electron-withdrawing Troc group on the nitrogen atom further enhances the electrophilicity of the bridgehead carbon, facilitating the substitution without causing elimination side reactions that are common in strained bicyclic systems.
Following the etherification, the removal of the Troc group is achieved through a single-electron transfer mechanism mediated by zinc metal. In a solvent system comprising THF, water, and acetic acid, zinc dust reduces the trichloroethyl moiety, leading to the fragmentation of the carbamate linkage. This reaction releases the free secondary amine and generates chloroacetaldehyde byproducts, which are easily separated during the aqueous work-up. The mild acidity of the reaction medium ensures that the liberated amine is immediately captured as a stable hydrochloride salt, which precipitates from the solution. This precipitation drives the equilibrium forward and simultaneously purifies the product, as impurities remain in the mother liquor. This mechanism exemplifies how careful selection of protecting groups can streamline downstream processing.
How to Synthesize Exo-3-(2-oxo-2H-benzopyran-7-yloxy) Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical three-step sequence designed for operational simplicity and high yield. The process begins with the protection of tropine derivatives, followed by the pivotal Mitsunobu coupling, and concludes with the reductive deprotection. Each step has been optimized to maximize crystallinity and minimize impurity carryover. The detailed standardized synthesis steps below outline the specific reagents, stoichiometry, and conditions required to replicate this high-efficiency route in a pilot or production setting.
- Protect endo-3-hydroxy-8-aza-bicyclo[3.2.1]octane with 2,2,2-trichloroethyl chloroformate to form the stable Troc-carbamate intermediate.
- Perform Mitsunobu coupling between the protected alcohol and 7-hydroxycoumarin using triphenylphosphine and DEAD to invert stereochemistry to the exo-form.
- Execute zinc-mediated reductive cleavage of the trichloroethyl carbamate group in THF/acetic acid to yield the final free amine hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible economic and logistical benefits. The primary advantage stems from the elimination of chromatographic purification, which is often the most costly and time-consuming unit operation in fine chemical manufacturing. By relying on crystallization and filtration, the process significantly reduces solvent consumption, waste generation, and processing time. This translates directly into lower manufacturing costs and a reduced environmental footprint, aligning with modern green chemistry principles. Furthermore, the robustness of the crystalline intermediates ensures consistent quality across batches, reducing the risk of supply disruptions due to failed quality control tests.
- Cost Reduction in Manufacturing: The replacement of chromatography with crystallization drastically lowers the cost of goods sold (COGS). Chromatography requires expensive silica gel, large volumes of high-purity solvents, and specialized equipment, all of which add significant overhead. In contrast, the crystallization-based workflow described in this patent utilizes standard reactor vessels and filtration equipment found in any multipurpose chemical plant. Additionally, the high overall yield achieved through this route minimizes raw material waste, further enhancing cost efficiency in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as tropine and 7-hydroxycoumarin, are commercially available commodities with stable supply chains. The use of robust reagents like zinc dust and triphenylphosphine ensures that the process is not dependent on exotic or scarce catalysts. This availability reduces the risk of raw material shortages and price volatility. Moreover, the ability to produce crystalline intermediates allows for easier storage and transportation, as solids are generally more stable and less hazardous than oils or solutions, thereby improving overall supply chain resilience.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on heterogeneous reactions and solid-liquid separations. The zinc-mediated deprotection, for instance, can be easily managed in large-scale reactors with appropriate temperature control. From an environmental perspective, the avoidance of chlorinated solvents often associated with chromatography and the use of recyclable zinc salts contribute to a cleaner production profile. This facilitates regulatory compliance and simplifies the waste disposal process, making it an attractive option for sustainable chemical production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of adopting this route for commercial production.
Q: Why is the 2,2,2-trichloroethyl (Troc) protecting group preferred over methyl or t-Boc groups for this synthesis?
A: The Troc group offers a unique balance of stability during the Mitsunobu coupling and mild removability via zinc reduction. Unlike methyl groups which require harsh demethylation conditions, or t-Boc groups which generate gaseous byproducts, the Troc group allows for clean, crystalline product isolation without chromatography, significantly enhancing process scalability.
Q: How does this process ensure high purity without chromatographic purification?
A: The process relies on the crystallization properties of the intermediates. The Troc-protected intermediate and the final hydrochloride salt both precipitate as crystalline solids from the reaction mixture or upon anti-solvent addition. This physical state allows for impurities to be washed away effectively, meeting stringent purity specifications required for API manufacturing.
Q: Is the zinc-mediated deprotection step scalable for industrial production?
A: Yes, the use of zinc dust in THF/acetic acid is a well-established heterogeneous reduction method suitable for large-scale reactors. The exothermic nature is manageable, and the removal of zinc salts is straightforward via filtration, making it highly compatible with commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chromen-2-one Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert process chemists has extensively evaluated the technology disclosed in CN101679407B and confirmed its potential for large-scale application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from the laboratory to the marketplace. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of chromen-2-one derivatives meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in pharmaceutical intermediates can accelerate your timeline and optimize your budget.
