Advanced Fluorinated Aromatic Diamine Synthesis for High-Performance Polyimide Manufacturing
The landscape of high-performance polymer materials is continuously evolving, driven by the demanding requirements of the microelectronics and aerospace sectors. Patent CN101270059B introduces a significant breakthrough in the synthesis of fluorine-containing unsymmetrical aromatic diamines, which serve as critical monomers for advanced polyimide production. Traditional polyimides often suffer from high melting temperatures and poor solubility due to rigid molecular chains, limiting their processability in complex electronic packaging applications. This patented technology addresses these fundamental limitations by strategically incorporating ether bonds, trifluoromethyl groups, and asymmetric structures into the polymer backbone. The resulting materials demonstrate exceptional solubility, optical transparency, and thermal stability, making them ideal for next-generation display and optoelectronic devices. As a reliable electronic chemical supplier, understanding this molecular innovation is key to sourcing materials that balance performance with manufacturability. The compound, characterized by its specific structural formula, remains stable at room temperature and offers a melting point range of 138 to 140°C, ensuring safe handling and storage during logistics.
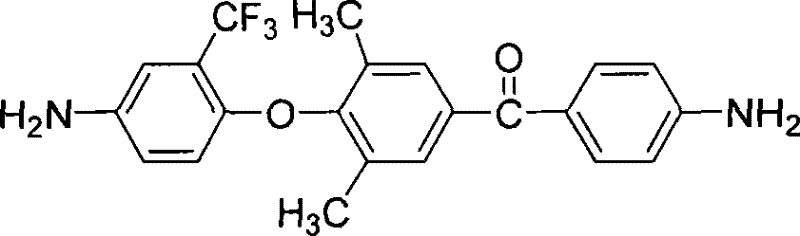
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diamines for polyimide production has relied on symmetrical structures that promote tight molecular packing and strong intermolecular interactions. While these materials offer high thermal resistance, they frequently exhibit poor solubility in common organic solvents, necessitating the use of aggressive processing conditions or specialized solvents that increase environmental and safety risks. The rigid benzene and imide rings in conventional polymers lead to high melting temperatures, often exceeding the thermal limits of standard processing equipment. This inflexibility restricts their application in fields requiring precise film formation or coating, such as flexible displays or microelectronic encapsulation. Furthermore, the lack of functional groups like ether linkages or fluorine atoms in traditional monomers limits the ability to tune the dielectric and optical properties of the final polymer. Consequently, manufacturers face significant challenges in achieving the desired balance between mechanical strength and processability, often resulting in higher production costs and longer development cycles for new electronic materials.
The Novel Approach
The patented process outlined in CN101270059B offers a transformative solution by employing a multi-step synthesis route that introduces key structural modifications to the aromatic diamine backbone. This novel approach utilizes a Friedel-Crafts acylation followed by a nucleophilic substitution and a catalytic reduction, allowing for the precise insertion of trifluoromethyl groups and ether linkages. These modifications effectively disrupt the regularity of the polymer chain, reducing intermolecular forces and significantly enhancing solubility in solvents like N,N-dimethylacetamide and chloroform. The asymmetric nature of the resulting diamine prevents close packing of the molecular chains, which lowers the melting temperature and improves film-forming capabilities without compromising thermal stability. This method enables the production of colorless or light yellow films with excellent optical transparency, a critical requirement for optoelectronic applications. By adopting this advanced synthesis route, manufacturers can achieve cost reduction in electronic chemical manufacturing through improved yield and simplified processing steps, while also meeting stringent performance specifications for high-end industrial applications.
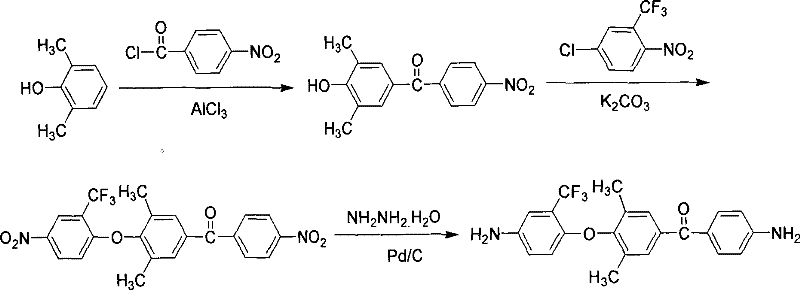
Mechanistic Insights into Fluorine-containing Unsymmetrical Aromatic Diamines Synthesis
The core of this technological advancement lies in the precise control of reaction conditions and catalytic mechanisms during the three-step synthesis process. The initial step involves the reaction of 2,6-dimethylphenol with p-nitrobenzoyl chloride in the presence of aluminum trichloride as a Lewis acid catalyst. This Friedel-Crafts acylation occurs at controlled temperatures between 10 and 20°C, ensuring high selectivity and minimizing side reactions that could lead to impurities. The subsequent nucleophilic substitution reaction utilizes potassium carbonate in a dimethylformamide solution to couple the intermediate ketone with 2-chloro-5-nitro trifluorotoluene. This step is critical for introducing the trifluoromethyl group, which enhances the hydrophobicity and dielectric properties of the final polymer. The reaction is conducted at elevated temperatures of 100 to 150°C to overcome the activation energy barrier, ensuring complete conversion of the reactants. Finally, the reduction of the nitro groups to amino groups is achieved using hydrazine hydrate and a palladium on carbon catalyst. This catalytic hydrogenation alternative is highly efficient and operates at moderate temperatures of 75 to 85°C, preserving the integrity of the sensitive fluorine-containing structures while delivering high-purity diamine products.
Impurity control is a paramount concern in the production of high-purity electronic chemical intermediates, and this patent details specific measures to ensure product quality. The use of recrystallization steps with methanol and ethanol solvents after each reaction stage effectively removes unreacted starting materials and by-products. For instance, the intermediate ketone is purified via steam distillation and recrystallization, yielding a light yellow solid with a defined melting point range. The final diamine product undergoes hot filtration to remove the palladium catalyst, followed by concentration and precipitation to isolate the pure compound. Analytical data, including FT-IR and NMR spectroscopy, confirms the structural integrity and purity of the synthesized diamine, with characteristic peaks corresponding to the amino, carbonyl, and trifluoromethyl groups. This rigorous purification protocol ensures that the resulting polyimides exhibit consistent mechanical and thermal properties, which is essential for maintaining supply chain reliability and meeting the stringent quality standards of global electronics manufacturers. The ability to produce materials with low coloration and high optical transparency further validates the effectiveness of this impurity control strategy.
How to Synthesize Fluorinated Aromatic Diamine Efficiently
Implementing this synthesis route requires careful attention to reagent ratios, temperature control, and safety protocols to maximize efficiency and yield. The process begins with the preparation of the reaction vessel under inert atmosphere conditions to prevent oxidation of sensitive intermediates. Operators must strictly adhere to the specified molar ratios, such as using 2.1 to 2.3 times the molar amount of aluminum trichloride relative to the phenol substrate, to ensure complete catalysis. The detailed standardized synthesis steps involve precise addition rates for reagents like hydrazine hydrate to manage exothermic reactions safely. For R&D teams looking to replicate or scale this process, understanding the solubility profiles of intermediates in solvents like 1,2-dichloroethane and ethanol is crucial for optimizing workup procedures. The following guide outlines the critical operational parameters derived from the patent examples, providing a roadmap for successful laboratory and pilot-scale production.
- Conduct Friedel-Crafts acylation of 2,6-dimethylphenol with p-nitrobenzoyl chloride using aluminum trichloride catalyst at 10-20°C.
- Perform nucleophilic substitution with 2-chloro-5-nitro trifluorotoluene under alkaline conditions at 100-150°C to form the dinitro intermediate.
- Execute catalytic reduction using hydrazine hydrate and palladium on carbon at 75-85°C to yield the final fluorinated diamine.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented synthesis route offers substantial strategic benefits that extend beyond mere technical performance. The elimination of complex high-temperature curing steps required for traditional insoluble polyimides translates directly into simplified manufacturing workflows and reduced energy consumption. By utilizing readily available raw materials such as 2,6-dimethylphenol and p-nitrobenzoyl chloride, the supply chain becomes more resilient to fluctuations in specialty chemical availability. The improved solubility of the resulting polyimides allows for processing in standard coating equipment, reducing the need for capital investment in specialized high-temperature machinery. This flexibility enables manufacturers to respond more quickly to market demands and customize product formulations for specific client requirements. Furthermore, the high purity and stability of the monomer reduce the risk of batch-to-batch variability, ensuring consistent quality for downstream production lines. These factors collectively contribute to significant cost savings and enhanced operational efficiency for companies integrating these materials into their product portfolios.
- Cost Reduction in Manufacturing: The synthesis route avoids the use of expensive transition metal catalysts in the final reduction step by utilizing a highly efficient palladium on carbon system that can be recovered and reused. This significantly lowers the raw material costs associated with catalyst consumption and waste disposal. Additionally, the moderate reaction temperatures reduce energy requirements for heating and cooling, leading to lower utility costs per kilogram of product. The high yields reported in the patent examples indicate minimal material waste, further optimizing the cost structure. By streamlining the purification process through effective recrystallization techniques, manufacturers can reduce the time and solvents needed for isolation, contributing to overall cost reduction in electronic chemical manufacturing without compromising product quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like potassium carbonate and hydrazine hydrate ensures a stable supply of key reagents, minimizing the risk of production delays due to raw material shortages. The robustness of the synthesis process, which tolerates slight variations in reaction conditions, enhances the reliability of production schedules. This stability is crucial for maintaining continuous supply to downstream customers in the electronics industry, where downtime can be extremely costly. The ability to store the intermediate and final products at room temperature without degradation simplifies logistics and warehousing requirements. Consequently, partners can maintain leaner inventory levels while ensuring high-purity polyimide monomer availability, strengthening the overall resilience of the supply chain against external disruptions.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard reactor types and separation techniques that are easily transferable from laboratory to industrial scale. The use of common organic solvents allows for established recovery and recycling protocols, aligning with strict environmental regulations and sustainability goals. The reduction of hazardous waste through high-yield reactions and efficient catalyst removal supports compliance with global environmental standards. This scalability ensures that production volumes can be increased to meet growing demand for advanced electronic materials without significant process redesign. The environmental benefits of this route, including lower energy consumption and reduced solvent usage, also enhance the corporate sustainability profile of manufacturers adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this fluorinated diamine technology. These answers are derived directly from the patent specifications and experimental data, providing accurate information for decision-makers. Understanding these details helps clarify the feasibility of integrating this material into existing production lines and product designs. The responses cover aspects of chemical stability, processing requirements, and performance metrics relevant to R&D and procurement teams. This section aims to eliminate uncertainties and facilitate informed discussions between suppliers and potential clients regarding the adoption of this advanced polymer monomer.
Q: How does the asymmetric structure improve polyimide solubility?
A: The introduction of asymmetric structures and ether bonds disrupts the regular packing of polymer chains, significantly reducing intermolecular forces and enhancing solubility in common organic solvents.
Q: What are the thermal stability characteristics of this monomer?
A: The resulting polyimides exhibit glass transition temperatures above 285°C and initial decomposition temperatures exceeding 500°C, ensuring robust performance in high-heat electronic applications.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process utilizes standard chemical reagents and moderate reaction conditions, facilitating straightforward scale-up from laboratory to commercial manufacturing without complex equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorinated Aromatic Diamine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the complexities of fluorinated chemistry and polyimide monomer synthesis, ensuring that the transition from patent to production is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the high standards required for electronic applications. Our commitment to quality and consistency makes us a trusted partner for global companies seeking to innovate their material portfolios. By leveraging our expertise, clients can accelerate their development timelines and secure a stable supply of critical intermediates for their high-performance polymer projects.
We invite you to engage with our technical procurement team to discuss how this technology can optimize your supply chain and reduce costs. Request a Customized Cost-Saving Analysis to understand the specific economic benefits for your operation. Our experts are ready to provide specific COA data and route feasibility assessments tailored to your unique requirements. Whether you need small quantities for R&D or large-scale production for commercial launch, we are equipped to support your needs with speed and precision. Contact us today to explore the potential of fluorinated aromatic diamines in your next generation of electronic materials.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
