Advanced Polyalkyl-Substituted Aromatic Diamine Monomers for Next-Generation Soluble Polyimide Manufacturing
Advanced Polyalkyl-Substituted Aromatic Diamine Monomers for Next-Generation Soluble Polyimide Manufacturing
The rapid evolution of the microelectronics and optoelectronics industries demands polymer materials that can withstand extreme thermal environments while offering superior optical clarity and processability. Traditional polyimides, while thermally robust, often suffer from poor solubility and deep coloration due to strong intermolecular charge transfer interactions. Addressing these critical limitations, the technology disclosed in patent CN101514164A introduces a novel class of polyalkyl-substituted aromatic diamine monomers. These specialized monomers serve as the foundational building blocks for creating soluble, high-temperature resistant, and low-dielectric constant polyimide films. By strategically incorporating bulky alkyl substituents into the aromatic backbone, this innovation effectively disrupts chain packing without sacrificing thermal integrity. For R&D directors and procurement specialists seeking a reliable polyimide monomer supplier, understanding the mechanistic advantages and commercial scalability of this synthesis route is paramount for securing a competitive edge in advanced material manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of soluble polyimides has relied heavily on the incorporation of flexible ether linkages or fluorinated groups into the polymer backbone. While these modifications improve solubility, they often come at a significant cost to thermal performance. For instance, fluorinated polyimides, despite their excellent optical properties, frequently exhibit glass transition temperatures limited to the 200-300 °C range, which is insufficient for high-end aerospace or semiconductor applications. Furthermore, the synthesis of fluorinated monomers involves complex, multi-step reactions with expensive reagents, leading to prohibitive production costs. Conventional rigid polyimides, on the other hand, possess excellent thermal stability but are notoriously difficult to process; they are often insoluble in common organic solvents and require hazardous concentrated sulfuric acid for dissolution. This insolubility creates severe bottlenecks in coating and film-forming processes, limiting their utility in modern precision electronics where solution processing is essential for uniform thin-film deposition.
The Novel Approach
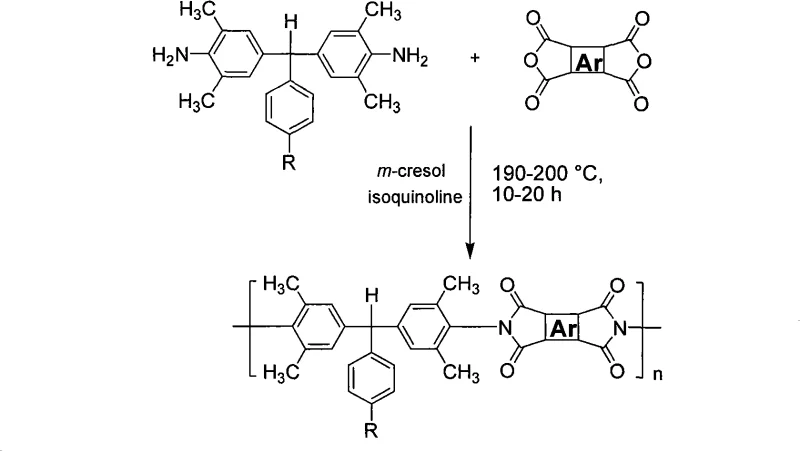
The methodology outlined in the patent offers a transformative alternative by utilizing sterically hindered aromatic diamines that maintain rigidity while preventing dense molecular stacking. The core innovation lies in the synthesis of α,α-(4-amino-3,5-dimethylphenyl)-4'-isopropyltoluene or its tert-butyl analog. These monomers introduce large, bulky side groups that force the polymer chains into a non-coplanar conformation. This structural distortion effectively inhibits the formation of charge-transfer complexes, resulting in polymers that are not only highly soluble in common solvents like NMP and DMAc but also exhibit exceptional optical transparency with cutoff wavelengths between 320-355 nm. The polymerization process itself is streamlined, utilizing a one-step high-temperature thermal imidization in m-cresol. As illustrated in the reaction scheme below, this approach allows for the direct formation of high molecular weight polyimides (Mw 40,000-65,000) without the need for separate chemical imidization steps.
Mechanistic Insights into Acid-Catalyzed Condensation and Steric Engineering
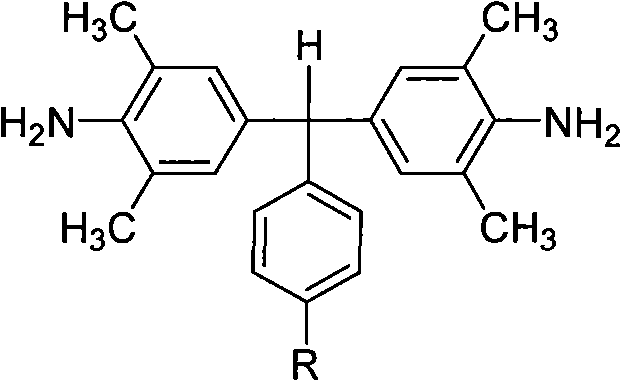
The synthesis of the core diamine monomer relies on a precise acid-catalyzed condensation reaction between 2,6-dimethylaniline and aromatic aldehydes (specifically 4-isopropylbenzaldehyde or 4-tert-butylbenzaldehyde). The reaction mechanism is driven by the electrophilic activation of the aldehyde carbonyl group by hydrochloric acid, facilitating nucleophilic attack by the electron-rich aniline rings. Crucially, the reaction is conducted under rigorous temperature control, initially heating the aniline to 100-120 °C before the dropwise addition of the aldehyde-acid mixture. This controlled addition prevents localized exotherms that could lead to polymerization byproducts or tar formation. Following the addition, the mixture is refluxed at 150-160 °C for 10-15 hours. This extended thermal treatment ensures the complete dehydration and formation of the central methine bridge, locking the bulky alkyl groups into the final structure. The resulting molecule, as depicted in the general structural formula, possesses a distinct three-dimensional geometry that is key to its performance.
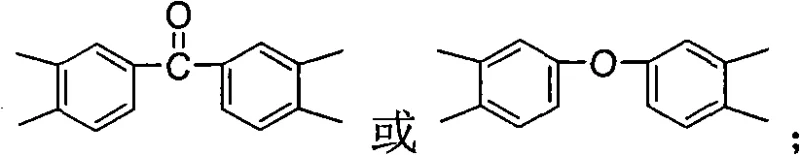
Beyond the monomer synthesis, the subsequent polymerization mechanism leverages the unique reactivity of these sterically hindered diamines with various aromatic dianhydrides. The patent specifies the use of dianhydrides such as pyromellitic dianhydride, biphenyltetracarboxylic dianhydride, or diphenyl ether tetracarboxylic dianhydride. The choice of the 'Ar' group in the dianhydride component allows for fine-tuning of the final polymer's dielectric and mechanical properties. For example, incorporating a ketone or ether linkage in the dianhydride (as shown in the variable Ar structures) can further enhance solubility or adjust the refractive index. The use of isoquinoline as a catalyst during the imidization step at 190-200 °C facilitates the cyclodehydration of the polyamic acid intermediate directly into the polyimide. This high-temperature pathway is energetically efficient and eliminates the need for chemical dehydrating agents like acetic anhydride, thereby simplifying the purification process and reducing the risk of residual impurities that could compromise the dielectric strength of the final film.
How to Synthesize Polyalkyl-Substituted Aromatic Diamine Efficiently
The preparation of these high-value monomers is designed for operational simplicity and robustness, making it highly attractive for scale-up. The process begins with the careful preparation of the reaction vessel under an inert nitrogen atmosphere to prevent oxidative degradation of the amine starting materials. The stoichiometry is critical, typically employing a molar ratio where 2,6-dimethylaniline is in excess relative to the aldehyde to drive the equilibrium towards the desired diamine product. The detailed standardized synthesis steps, including specific workup procedures and purification protocols required to achieve pharmaceutical-grade purity, are outlined in the technical guide below.
- Heat 2,6-dimethylaniline to 100-120°C under nitrogen protection and slowly add a mixture of aromatic aldehyde and hydrochloric acid.
- Reflux the mixture at 150-160°C for 10-15 hours to ensure complete condensation and formation of the diamine backbone.
- Neutralize with sodium hydroxide, precipitate with ethanol, wash with methanol, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel monomer technology represents a significant opportunity for cost reduction in electronic chemical manufacturing. The primary economic driver is the simplicity of the synthetic route. Unlike fluorinated monomers that require hazardous and expensive fluorination reagents, this process utilizes commodity chemicals like 2,6-dimethylaniline and substituted benzaldehydes, which are readily available in the global market. The absence of transition metal catalysts eliminates the need for costly and time-consuming heavy metal removal steps, which are often a bottleneck in API and fine chemical production. Furthermore, the reaction does not require specialized high-pressure equipment or cryogenic conditions; standard stainless steel reactors capable of handling temperatures up to 160 °C are sufficient. This compatibility with existing infrastructure drastically lowers the barrier to entry for manufacturers looking to diversify their polyimide portfolios.
- Cost Reduction in Manufacturing: The elimination of complex multi-step synthesis sequences significantly lowers the overall cost of goods sold. By avoiding the use of expensive fluorinated precursors and simplifying the purification workflow to standard silica gel chromatography and solvent washing, the process achieves substantial cost savings. The high yield reported in the patent examples (60-80%) further enhances economic viability, ensuring that raw material utilization is optimized and waste generation is minimized compared to lower-yielding conventional routes.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term production contracts. Since the key raw materials are bulk petrochemical derivatives rather than niche specialty intermediates, the supply chain is inherently more resilient to market fluctuations. The robust nature of the synthesis, which tolerates standard industrial conditions without requiring ultra-high purity inert gases beyond standard nitrogen protection, ensures consistent batch-to-batch quality. This reliability reduces the risk of production delays caused by raw material shortages or stringent storage requirements associated with more sensitive chemical intermediates.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial production, with no special equipment requirements noted in the patent documentation. The use of m-cresol and isoquinoline in the polymerization step, while requiring proper handling, allows for solvent recovery and recycling, aligning with modern green chemistry principles. The ability to produce polymers with high solid content (5-20%) improves reactor throughput, allowing manufacturers to maximize output per batch. Additionally, the resulting polyimides exhibit excellent solubility in low-boiling solvents like chloroform, which are easier to recover and recycle than high-boiling polar aprotic solvents, further reducing environmental impact and disposal costs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this polyalkyl-substituted diamine technology. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of the material's capabilities and limitations for potential adopters.
Q: What are the key structural advantages of this polyalkyl-substituted diamine?
A: The monomer features bulky isopropyl or tert-butyl side groups that create a rigid non-coplanar structure. This disrupts molecular packing, significantly improving solubility and optical transparency while maintaining high thermal stability (Tg > 320°C).
Q: How does this synthesis method reduce production costs compared to fluorinated alternatives?
A: The process utilizes readily available raw materials like 2,6-dimethylaniline and avoids expensive fluorinated reagents. The one-step thermal imidization eliminates complex multi-stage polymerization procedures, simplifying equipment requirements and reducing operational overhead.
Q: Is this monomer suitable for large-scale industrial polyimide production?
A: Yes, the patent explicitly states the method has no special equipment requirements and is suitable for industrial production. The reaction conditions (150-160°C reflux) are compatible with standard chemical reactors, ensuring scalable manufacturing from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyalkyl-Substituted Aromatic Diamine Supplier
The commercialization of advanced polyimide materials requires a partner who understands both the intricacies of monomer synthesis and the demands of large-scale polymer production. NINGBO INNO PHARMCHEM stands ready to support your development goals with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific thermal and atmospheric requirements of this condensation chemistry, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs, we guarantee that the diamine monomers supplied possess the precise structural integrity necessary to achieve the reported dielectric constants of 2.25-2.95 and thermal stability exceeding 320 °C.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating these monomers into your specific application workflows. Whether you are developing flexible display substrates or high-frequency circuit boards, our experts can provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your transition to next-generation soluble polyimide solutions.
