Advanced Sacubitril Manufacturing: Overcoming Chiral Synthesis Challenges for Commercial Scale
Advanced Sacubitril Manufacturing: Overcoming Chiral Synthesis Challenges for Commercial Scale
The global demand for effective heart failure treatments has placed immense pressure on the supply chains of key active pharmaceutical ingredients (APIs), particularly Sacubitril, a critical component of the blockbuster drug Entresto. As the industry seeks more robust and economically viable manufacturing pathways, Patent CN114436877A emerges as a pivotal technological advancement, offering a refined synthesis process for this cardiovascular therapeutic. This patent discloses a novel route that strategically bypasses the limitations of traditional methods by employing a clever sequence of esterification, Michael addition, and hydrolysis. For R&D directors and procurement specialists alike, understanding the nuances of this process is essential for securing a reliable Sacubitril intermediate supplier capable of delivering high-purity materials consistently. The disclosed method not only streamlines the chemical transformations but also addresses critical safety and environmental concerns associated with earlier generations of synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
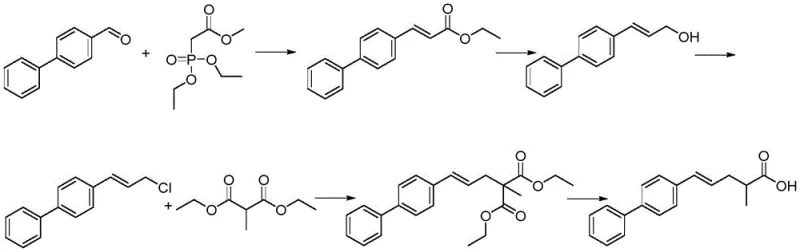
Historically, the industrial preparation of Sacubitril has relied heavily on routes described in patents such as US5217996 and WO2008031567. These conventional pathways typically utilize chiral amino alcohols as starting materials, necessitating a series of complex transformations including oxidation to aldehydes, Wittig reactions, and chiral hydrogenation. From a process chemistry perspective, these steps introduce significant vulnerabilities; oxidation reactions often require harsh conditions and generate substantial waste, while chiral hydrogenation demands expensive noble metal catalysts that are difficult to remove completely from the final API. Furthermore, the reliance on high-risk reagents creates potential safety hazards in a manufacturing plant, and the multi-step nature of these routes inevitably leads to cumulative yield losses. The presence of genotoxic impurities, such as epichlorohydrin in certain legacy routes, poses a severe regulatory hurdle, requiring extensive and costly purification protocols to ensure patient safety.
The Novel Approach
In stark contrast, the methodology outlined in CN114436877A presents a paradigm shift by utilizing a Michael addition strategy to construct the core skeleton of the molecule. This innovative approach begins with readily available starting materials and proceeds through a sequence that avoids the aforementioned high-risk oxidation and Grignard reactions. By focusing on the transformation of Compound V through an ethyl ester intermediate (Compound VI) and subsequently reacting it with succinimide, the process achieves the target structure with remarkable efficiency. The use of R-phenylethylamine as a chiral auxiliary in the precursor stage is particularly noteworthy; it is not only cost-effective but also recyclable, which drastically reduces raw material costs. This route effectively mitigates the risk of genotoxic impurities and simplifies the downstream purification process, making it an ideal candidate for cost reduction in cardiovascular drug manufacturing. The simplicity of the operation, characterized by standard unit operations like reflux and recrystallization, ensures that the process is inherently safer and more amenable to automation.
Mechanistic Insights into the Michael Addition and Chiral Resolution Strategy
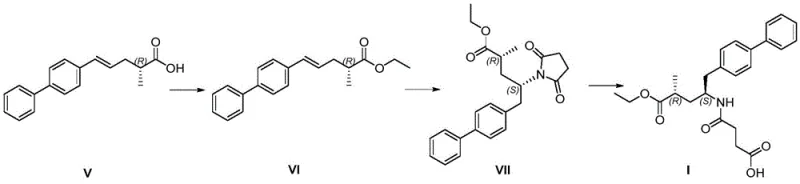
The core chemical innovation of this patent lies in the strategic application of the Michael addition reaction to install the succinimide moiety, which is later hydrolyzed to form the glutaric acid side chain of Sacubitril. In the conversion of Compound VI to Compound VII, the alpha,beta-unsaturated ester system of Compound VI acts as a Michael acceptor. Under basic conditions, provided by reagents such as potassium carbonate or sodium methoxide, the nitrogen atom of succinimide becomes nucleophilic and attacks the beta-carbon of the unsaturated ester. This conjugate addition is highly regioselective and proceeds smoothly at moderate temperatures ranging from 60°C to 80°C. The stereochemical outcome of this step is critical, as it establishes the relative configuration between the existing chiral center and the newly formed bond. The patent data indicates that this reaction yields the desired diastereomer with high selectivity, minimizing the formation of unwanted isomers that would otherwise complicate purification. Following this, the hydrolysis of the imide ring in Compound VII under mild alkaline conditions (sodium carbonate in ethanol/water) cleanly opens the ring to reveal the free carboxylic acid, yielding the final Compound I without racemization of the sensitive chiral centers.
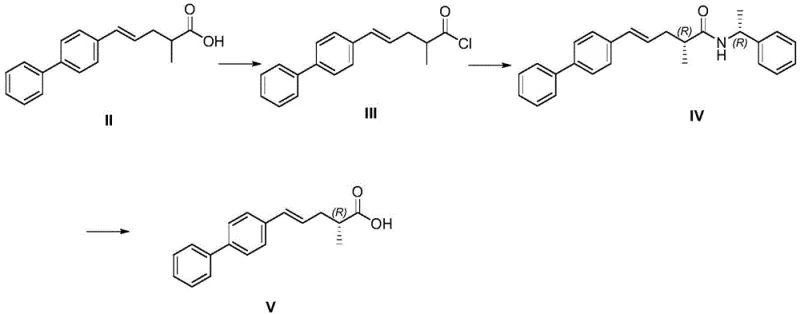
Equally important is the mechanism employed to establish the initial chirality in the precursor Compound V. The process utilizes R-phenylethylamine to resolve the racemic acid chloride (Compound III) via amide formation. This classical resolution technique leverages the diastereomeric differences between the resulting amides to separate the desired enantiomer. The patent highlights that the mother liquor from this crystallization can be racemized and recycled, a feature that significantly enhances the atom economy of the process. The subsequent hydrolysis of the chiral amide (Compound IV) to release the free acid (Compound V) is performed under controlled acidic conditions to prevent epimerization. This careful control of reaction parameters ensures that the optical purity established in the early stages is preserved throughout the synthesis, resulting in a final product with stringent purity specifications required for pharmaceutical applications. The integration of these mechanistic insights demonstrates a deep understanding of physical organic chemistry, translating directly into a more robust and scalable manufacturing process.
How to Synthesize Sacubitril Efficiently
Implementing this synthesis route requires precise control over reaction stoichiometry and thermal conditions to maximize yield and purity. The process is divided into distinct operational units, beginning with the preparation of the chiral acid precursor, followed by the key coupling and functionalization steps. Operators must adhere strictly to the specified molar ratios, such as the 1:0.05 to 1:0.1 ratio of Compound V to concentrated sulfuric acid during esterification, to ensure complete conversion without excessive degradation. The detailed standardized synthesis steps below outline the critical parameters for each stage, ensuring reproducibility from the laboratory bench to the pilot plant.
- Esterify Compound V with ethanol using concentrated sulfuric acid catalyst at 70-80°C to form Compound VI.
- Perform Michael addition of Compound VI with succinimide in the presence of alkali base at 60-80°C to generate Compound VII.
- Hydrolyze Compound VII in aqueous sodium carbonate ethanol solution at 20-50°C to yield the final Sacubitril product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical advantages of this patent translate directly into tangible business benefits, primarily centered around cost stability and supply security. The elimination of expensive chiral catalysts and the ability to recycle the chiral resolving agent fundamentally alter the cost structure of the API. By removing the dependency on precious metals and complex enzymatic processes, the manufacturing overhead is significantly lowered, allowing for more competitive pricing strategies in a volatile market. Furthermore, the simplified workflow reduces the number of unit operations, which in turn decreases the consumption of solvents and energy, aligning with modern sustainability goals and reducing the environmental footprint of production facilities.
- Cost Reduction in Manufacturing: The most significant economic driver of this process is the substitution of high-cost reagents with commodity chemicals. The use of R-phenylethylamine, which is inexpensive and recyclable, replaces costly chiral pool materials or asymmetric catalysts. Additionally, the avoidance of high-pressure hydrogenation equipment reduces capital expenditure (CAPEX) requirements for manufacturing sites. The high yields reported in the examples, particularly the near-quantitative conversion in the esterification step, minimize material loss, ensuring that every kilogram of raw material contributes maximally to the final output. This efficiency creates a buffer against raw material price fluctuations, providing long-term cost predictability for buyers.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available starting materials such as biphenyl aldehyde and succinimide. Unlike specialized chiral building blocks that may have single-source suppliers, the precursors for this route are produced by multiple chemical manufacturers globally, reducing the risk of supply disruption. The robustness of the chemical steps, which tolerate minor variations in conditions without catastrophic failure, ensures consistent batch-to-batch quality. This reliability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by downstream pharmaceutical customers.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The reaction conditions are mild, mostly operating below 80°C and at atmospheric pressure, which simplifies the engineering requirements for large-scale reactors. The workup procedures involve standard techniques like filtration and pulping, which are easily automated and scaled. Moreover, the reduction in hazardous waste generation, due to the absence of heavy metals and toxic oxidants, simplifies waste treatment compliance. This environmental advantage not only lowers disposal costs but also facilitates regulatory approvals in regions with strict environmental protection laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a clear picture of the process capabilities and limitations. Understanding these details is vital for technical teams evaluating the feasibility of adopting this route for their specific production needs.
Q: How does this new process improve upon conventional Sacubitril synthesis routes?
A: Unlike prior art (e.g., US5217996) which relies on expensive chiral amino alcohols and high-risk oxidation/hydrogenation steps, this patent utilizes a Michael addition strategy. This eliminates genotoxic impurity risks like epichlorohydrin and avoids costly chiral auxiliaries for the second center, significantly simplifying purification.
Q: What is the role of R-phenylethylamine in the precursor synthesis?
A: R-phenylethylamine acts as a chiral resolving agent during the formation of Compound IV. It allows for the construction of the chiral center with high diastereoselectivity. Crucially, the patent notes that this reagent is low-cost and can be recycled, enhancing the overall economic viability of the process.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scale-up. It features simple operations, mild reaction conditions (mostly below 80°C), and easy separation methods like pulping and recrystallization. The avoidance of hazardous reagents and the use of common solvents like ethanol and THF facilitate safe commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sacubitril Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields seen in the lab are replicated faithfully on an industrial scale. We are committed to delivering high-purity Sacubitril intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex chiral syntheses with the utmost care, guaranteeing the optical integrity of every batch we produce.
We invite you to collaborate with us to optimize your supply chain for this critical heart failure medication. By leveraging our technical proficiency, you can achieve significant efficiencies in your production planning. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing excellence can support your long-term business goals.
