Scalable Synthesis of Ethyl 1-Methyl-4-Nitroimidazole-2-Carboxylate for Pharmaceutical Applications
Introduction to Advanced Imidazole Derivative Synthesis
The pharmaceutical industry constantly seeks robust and scalable pathways for critical heterocyclic intermediates, particularly those serving as building blocks for antitumor and antiviral agents. A significant breakthrough in this domain is detailed in patent CN116120238A, which discloses a novel preparation method for ethyl 1-methyl-4-nitro-1H-imidazole-2-carboxylate. This compound is a pivotal intermediate in the synthesis of neuropathic pain drugs and various oncology therapeutics. The disclosed technology addresses long-standing challenges in the field, specifically targeting the high costs, safety hazards, and purification difficulties associated with legacy synthetic routes. By shifting the synthetic strategy from direct nitration to a sequential methylation-oxidation-esterification pathway, the invention offers a transformative approach to manufacturing high-purity pharmaceutical intermediates. This report analyzes the technical merits of this innovation, providing actionable insights for R&D directors seeking process optimization and procurement managers focused on supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-methyl-4-nitro-1H-imidazole derivatives has been plagued by inefficient and hazardous methodologies that hinder industrial scalability. One prevalent prior art route involves the use of benzyl protecting groups, requiring a multi-step sequence that includes hydrodebenzylation using palladium on carbon (Pd/C) catalysts under hydrogen pressure. This approach not only incurs high operational costs due to the use of precious metal catalysts but also demands stringent equipment specifications to handle high-pressure hydrogenation safely. Furthermore, the removal of trace heavy metals from the final API intermediate adds significant downstream processing burdens. Another conventional pathway attempts direct nitration of imidazole precursors using mixed acid systems; however, this method suffers from abysmal regioselectivity, often yielding less than 20% of the desired product while generating over 30% of the unwanted 5-nitroisomer, making separation economically unviable.
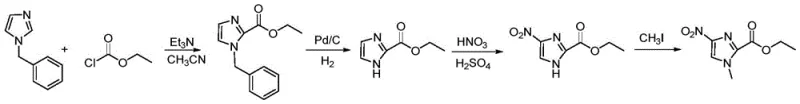
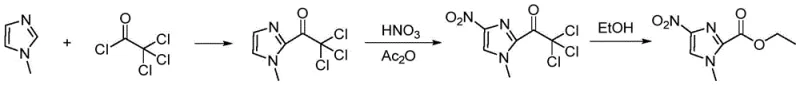
Perhaps most critically, certain established nitration protocols utilize nitric acid in combination with acetic anhydride. While chemically feasible on a small scale, this mixture poses severe safety risks on an industrial level due to the potential formation of tetranitromethane, a highly unstable and explosive byproduct. Additionally, these reactions often require extended reaction times, such as 30 hours, which drastically reduces throughput and increases energy consumption. The cumulative effect of these factors—low yield, high isomer contamination, expensive catalysts, and extreme safety hazards—renders many traditional methods unsuitable for modern, compliant pharmaceutical manufacturing environments.
The Novel Approach
In stark contrast to the problematic legacy methods, the innovative process described in CN116120238A introduces a streamlined three-step sequence that fundamentally alters the reaction logic to enhance safety and yield. The new strategy begins with the N-methylation of readily available 2-methyl-4-nitroimidazole, followed by the selective oxidation of the 2-position methyl group to a carboxylic acid, and concludes with esterification. This order of operations is chemically counter-intuitive yet highly effective; research indicated that attempting oxidation prior to methylation lowers the electron cloud density of the imidazole ring, thereby inhibiting the subsequent N-methylation and leading to unwanted methyl ester byproducts. By prioritizing N-methylation, the process leverages the electronic properties of the ring to facilitate high-selectivity oxidation in the second step. This logical inversion eliminates the need for hazardous nitration reagents entirely, replacing them with standard oxidants like potassium permanganate, and removes the requirement for expensive hydrogenation infrastructure.
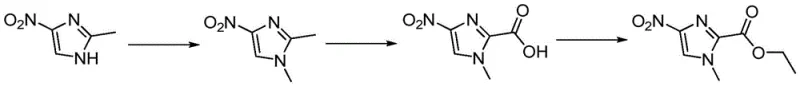
Mechanistic Insights into Sequential Methylation and Oxidation
The core mechanistic advantage of this synthesis lies in the precise control of electronic effects on the imidazole ring during the functionalization stages. The initial N-methylation step utilizes reagents such as methyl iodide or dimethyl sulfate in the presence of a base like potassium carbonate. Conducting this reaction at mild temperatures of 40-45°C ensures complete conversion to 1,2-dimethyl-4-nitroimidazole without degrading the sensitive nitro group. Crucially, introducing the N-methyl group first preserves the electron density necessary for the subsequent transformation. If the 2-methyl group were oxidized to a carboxylic acid first, the resulting electron-withdrawing effect would deactivate the nitrogen at the 1-position, making alkylation difficult and promoting side reactions where the methylating agent attacks the carboxyl group itself. This mechanistic understanding allows for a clean progression to the intermediate 1,2-dimethyl-4-nitroimidazole with yields exceeding 90%.
Following methylation, the oxidation of the 2-methyl group to the carboxylic acid is achieved using strong oxidants such as potassium permanganate in an aqueous medium. The presence of the 4-nitro group and the 1-methyl group creates a specific electronic environment that directs the oxidation selectively to the 2-position methyl group, minimizing over-oxidation or ring degradation. The final esterification step converts the carboxylic acid to the ethyl ester using thionyl chloride and ethanol. This activation via the acid chloride intermediate ensures rapid and high-yielding conversion to the final ethyl 1-methyl-4-nitro-1H-imidazole-2-carboxylate. The entire sequence is designed to minimize impurity profiles, specifically avoiding the formation of the troublesome 5-nitroisomers that plague direct nitration routes, thereby simplifying downstream purification and ensuring the high purity required for GMP manufacturing.
How to Synthesize Ethyl 1-Methyl-4-Nitroimidazole-2-Carboxylate Efficiently
Implementing this novel synthetic route requires careful attention to reaction stoichiometry and temperature control to maximize the benefits of the new methodology. The process is divided into three distinct operational units: alkylation, oxidation, and esterification. Each step utilizes common industrial solvents and reagents, facilitating easy technology transfer from the laboratory to pilot and commercial scales. The following guide outlines the critical parameters derived from the patent examples to ensure optimal performance and reproducibility. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide below.
- Perform N-methylation of 2-methyl-4-nitroimidazole using methyl iodide and potassium carbonate in acetone at 40-45°C.
- Oxidize the resulting 1,2-dimethyl-4-nitroimidazole using potassium permanganate in water under reflux conditions.
- Convert the intermediate carboxylic acid to the ethyl ester using thionyl chloride and ethanol at 0-5°C followed by reflux.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for benzyl chloride, palladium catalysts, and high-pressure hydrogen gas, manufacturers can source reagents from a broader, more stable vendor base, reducing the risk of supply disruptions. Furthermore, the avoidance of hazardous nitration mixtures significantly lowers the regulatory burden and insurance costs associated with handling explosive precursors. This shift not only enhances workplace safety but also streamlines the logistics of transporting chemicals, as the reagents involved are standard commodities rather than controlled hazardous substances. Consequently, the overall cost structure of the intermediate is optimized through both material savings and reduced operational overhead.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts represents a direct and significant reduction in variable costs. Traditional routes relying on Pd/C require not only the purchase of expensive catalysts but also the implementation of costly metal scavenging processes to meet strict residual metal limits in pharmaceutical products. By replacing this with a base-mediated alkylation and permanganate oxidation, the process removes these financial burdens entirely. Additionally, the high selectivity of the new route minimizes the loss of valuable starting materials to isomeric byproducts, effectively increasing the mass balance efficiency and reducing the cost per kilogram of the final active intermediate.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as methyl iodide, potassium carbonate, and ethanol ensures a robust and resilient supply chain. Unlike specialized nitrating agents or protected imidazole derivatives which may have limited suppliers, the reagents for this novel process are produced globally in massive volumes. This abundance mitigates the risk of price volatility and allocation issues that frequently impact the pharmaceutical supply chain. Moreover, the mild reaction conditions (40-45°C) reduce the dependency on specialized high-energy infrastructure, allowing for production in a wider range of facilities, thereby diversifying the manufacturing footprint and enhancing continuity of supply.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is superior to legacy methods. The removal of acetic anhydride and concentrated nitric acid from the workflow eliminates the generation of acidic waste streams that require complex neutralization and treatment. The aqueous workup in the oxidation step is straightforward and generates manageable solid waste (manganese dioxide) that can be filtered and disposed of according to standard protocols. The simplicity of the unit operations—stirring, heating, filtration, and distillation—makes the process inherently scalable from 100 kgs to multi-ton production without the need for bespoke reactor designs, ensuring that capacity can be ramped up quickly to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of adopting this route for your specific project requirements.
Q: Why is the new synthesis route safer than traditional nitration methods?
A: Traditional routes often utilize nitric acid and acetic anhydride, which can generate unstable tetranitromethane and pose severe explosion risks. The new method avoids direct nitration on the imidazole ring, instead using a safer oxidation of a methyl group, significantly reducing potential safety hazards.
Q: What are the yield advantages of this process compared to prior art?
A: Conventional direct nitration routes often suffer from low yields (around 20%) and high isomer formation (>30% 5-nitroisomer). The novel methylation-first approach achieves yields exceeding 90% in the initial step and maintains high purity throughout, drastically improving overall process efficiency.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common reagents like potassium permanganate and thionyl chloride under mild conditions (40-45°C for methylation). It eliminates the need for expensive palladium catalysts and high-pressure hydrogenation equipment, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ethyl 1-Methyl-4-Nitroimidazole-2-Carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from development to market. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ethyl 1-methyl-4-nitroimidazole-2-carboxylate meets the highest global standards for pharmaceutical intermediates. We are committed to delivering consistent quality and reliability for your critical supply chain needs.
We invite you to leverage our technical proficiency to optimize your sourcing strategy for this vital intermediate. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this novel route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to secure a sustainable and cost-effective supply of high-purity imidazole derivatives for your next-generation therapeutic programs.
