Advanced Synthesis of Novel Phenylethanolamine Derivatives for Respiratory Therapeutics Manufacturing
Advanced Synthesis of Novel Phenylethanolamine Derivatives for Respiratory Therapeutics Manufacturing
The pharmaceutical landscape for respiratory diseases, particularly asthma and chronic bronchitis, constantly demands novel therapeutic agents with improved efficacy and safety profiles. Patent CN1276911C introduces a significant advancement in this domain by disclosing a series of novel phenylethanolamine compounds exhibiting potent beta2-receptor excitatory effects. These compounds represent a critical class of pharmaceutical intermediates designed to overcome the limitations of existing treatments, offering a new pathway for drug development. The patent details a robust synthetic methodology that allows for the structural modification of the phenylethanolamine core, enabling the creation of diverse derivatives with tailored physicochemical properties. For R&D directors and procurement specialists in the global pharma industry, understanding the nuances of this synthesis is vital for securing a reliable supply chain of high-quality active ingredients. This report analyzes the technical depth of CN1276911C, highlighting its potential for cost reduction in API manufacturing and its feasibility for large-scale commercial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of beta2-receptor agonists has often relied on complex multi-step sequences that involve hazardous reagents or difficult purification processes. Traditional routes frequently suffer from poor regioselectivity during the functionalization of the aromatic ring, leading to challenging impurity profiles that require extensive chromatographic separation. Furthermore, many conventional methods utilize expensive transition metal catalysts that necessitate rigorous removal steps to meet stringent regulatory limits for residual metals in pharmaceutical products. The reliance on such catalysts not only inflates the cost of goods sold but also introduces supply chain vulnerabilities associated with the sourcing of precious metals. Additionally, older synthetic pathways often operate under extreme conditions of temperature and pressure, which complicates process safety and limits the ability to scale up reactions in standard glass-lined steel reactors without significant engineering modifications.
The Novel Approach
In contrast, the methodology described in CN1276911C offers a streamlined and chemically elegant solution to these challenges. The novel approach utilizes a straightforward two-stage synthesis starting from commercially available right-aminoacetophenone derivatives. By employing potassium borohydride for the initial reduction step, the process avoids the need for high-pressure hydrogenation equipment, thereby enhancing operational safety and reducing capital expenditure. The subsequent nucleophilic substitution reaction is conducted under reflux conditions in common organic solvents like ethanol or toluene, which are easily recoverable and recyclable. This strategy significantly simplifies the downstream processing, as the workup involves standard liquid-liquid extraction and crystallization techniques rather than complex distillation or chromatography. The ability to introduce diverse amine substituents at the final stage provides unparalleled flexibility for medicinal chemists to optimize the pharmacokinetic properties of the final drug candidate without redesigning the entire synthetic route.
Mechanistic Insights into Potassium Borohydride Reduction and Nucleophilic Substitution
The core of this synthetic strategy lies in the selective reduction of the alpha-brominated ketone intermediate to the corresponding alcohol, followed by a nucleophilic displacement. The first step involves the reaction of a 4-amino-3-halo-5-substituted-alpha-brominated methyl phenyl ketone with potassium borohydride in a methanol-water mixture. This reduction proceeds rapidly at room temperature, typically completing within 5 hours, to yield the bromohydrin intermediate with high efficiency. The mechanism involves the hydride transfer from the borohydride anion to the carbonyl carbon, generating an alkoxide which is subsequently protonated by the solvent system. This step is crucial as it establishes the stereochemistry and functionality required for the subsequent amination, while the mild conditions prevent the degradation of sensitive functional groups such as the trifluoromethyl or cyano moieties present on the aromatic ring.
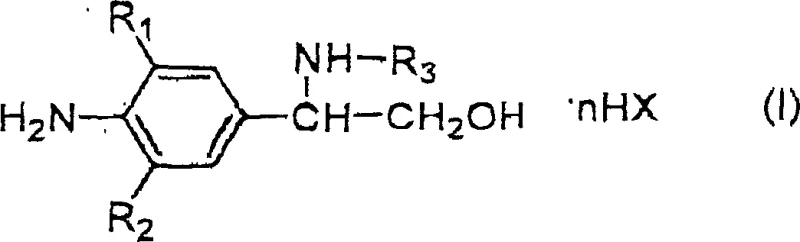
Following the reduction, the resulting alcohol intermediate undergoes a nucleophilic substitution reaction with a primary amine, such as tert-butylamine or isopropylamine. This transformation is the key step in constructing the phenylethanolamine backbone characteristic of beta2-agonists. The reaction is driven by the displacement of the bromide leaving group by the nitrogen lone pair of the amine. To facilitate this, the reaction is performed under anhydrous conditions at reflux temperature for an extended period of 10 to 15 hours. The prolonged heating ensures that the activation energy barrier for the substitution is overcome, driving the equilibrium towards the desired product. The choice of solvent, whether alcoholic or aromatic, plays a pivotal role in stabilizing the transition state and solubilizing the reactants. Impurity control is managed by carefully monitoring the reaction time and temperature, as over-heating can lead to elimination side products, while insufficient reaction time results in unreacted starting material which is removed during the subsequent acid-base extraction workup.
How to Synthesize 2-(4-amino-3-chloro-5-trifluoromethyl) Phenylethanolamine Efficiently
The practical execution of this synthesis requires precise control over stoichiometry and environmental conditions to maximize yield and purity. The process begins with the preparation of the bromohydrin intermediate, where strict temperature control during the addition of potassium borohydride is essential to manage the exotherm. Once the intermediate is isolated, it is immediately subjected to the amination step in a dry reactor to prevent hydrolysis of the reactive bromide. The detailed standardized synthetic steps, including specific molar ratios, solvent volumes, and crystallization parameters, are outlined in the technical guide below to ensure reproducibility for process chemists.
- Reduce the alpha-brominated ketone precursor using potassium borohydride in a methanol-water solvent system at room temperature for 5 hours.
- React the resulting alcohol intermediate with a primary amine (such as tert-butylamine) under anhydrous conditions in alcohol or arene solvents at reflux temperature for 10-15 hours.
- Purify the free base via extraction and crystallization, followed by conversion to the hydrochloride salt using hydrogen chloride in anhydrous diethyl ether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic route described in CN1276911C presents several compelling economic and logistical advantages. The primary benefit stems from the use of commodity chemicals and widely available starting materials, which mitigates the risk of supply disruptions often associated with specialized reagents. The elimination of precious metal catalysts not only reduces the direct material cost but also simplifies the waste management protocol, leading to substantial cost savings in environmental compliance and disposal. Furthermore, the robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process does not require specialized high-pressure or cryogenic equipment that might create bottlenecks in a multi-purpose production facility.
- Cost Reduction in Manufacturing: The synthetic pathway leverages inexpensive reducing agents like potassium borohydride and common amines, which are produced on a massive industrial scale globally. By avoiding the use of expensive chiral catalysts or protecting group strategies often seen in alternative routes, the overall cost of goods is significantly lowered. The simplified workup procedure, which relies on pH-controlled extractions and crystallization, minimizes the consumption of organic solvents and reduces the energy load required for solvent recovery. This efficiency translates directly into a more competitive pricing structure for the final pharmaceutical intermediate, allowing manufacturers to maintain healthy margins even in a price-sensitive market.
- Enhanced Supply Chain Reliability: The reliance on commercially available right-aminoacetophenone derivatives as the starting point ensures a stable and continuous supply of raw materials. Unlike custom-synthesized building blocks that may have long lead times, these precursors are stocked by multiple suppliers worldwide, providing procurement teams with leverage and redundancy. The synthetic steps themselves are operationally simple and do not depend on fragile reagents that degrade upon storage, ensuring that inventory can be held safely without significant loss of potency. This reliability is critical for maintaining uninterrupted production schedules for downstream API manufacturing, preventing costly delays in drug formulation and market launch.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram scale in the laboratory to potential tonnage production in industrial reactors. The absence of heavy metals simplifies the regulatory filing process, as there is no need for extensive validation of metal clearance steps, accelerating the time to market. Additionally, the aqueous workup and the ability to recycle solvents like toluene and ethanol align with green chemistry principles, reducing the environmental footprint of the manufacturing process. This alignment with sustainability goals is increasingly important for multinational corporations aiming to meet their corporate social responsibility targets while optimizing their chemical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel phenylethanolamine compounds. The answers are derived directly from the experimental data and specifications provided in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing manufacturing portfolio.
Q: What are the critical reaction conditions for the amination step?
A: The amination reaction requires strictly anhydrous conditions using solvents like dehydrated alcohol or toluene. The reaction must be maintained at reflux temperature for a prolonged period, typically between 10 to 15 hours, to ensure complete conversion of the alcohol intermediate.
Q: How is the purity of the final hydrochloride salt ensured?
A: Purity is achieved through a rigorous recrystallization process. The crude product is dissolved in dehydrated alcohol, filtered to remove insolubles, and then precipitated by adding anhydrous diethyl ether until crystallization occurs, followed by freezing and suction filtration.
Q: Can this synthesis route be scaled for commercial API production?
A: Yes, the route utilizes commercially available starting materials like right-aminoacetophenone derivatives and standard reagents like potassium borohydride. The process avoids exotic catalysts, making it highly suitable for scale-up in industrial reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylethanolamine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving respiratory medications. Our team of expert process chemists has extensively analyzed the synthetic route disclosed in CN1276911C and is fully prepared to support your development needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of phenylethanolamine derivative we supply meets the highest international standards for pharmaceutical use.
We invite you to collaborate with us to optimize this synthesis for your specific requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our manufacturing efficiencies can lower your overall project costs. Please contact us today to request specific COA data for our available batches and to discuss route feasibility assessments for your next-generation beta2-agonist projects. Let us be your partner in bringing innovative respiratory therapies to patients worldwide.
