Scalable Synthesis of Chiral Statin Intermediates for Global Pharmaceutical Manufacturing
The pharmaceutical industry's relentless pursuit of efficient lipid-lowering therapies has placed significant emphasis on the supply chain integrity of HMG-CoA reductase inhibitor intermediates. Patent CN1923792A introduces a transformative synthetic methodology for producing (S)-6-arylmethyl-5-hydroxy-3-oxo-hexanoic acid tert-butyl ester, a pivotal chiral building block in the manufacture of statins. This technology addresses critical bottlenecks in traditional manufacturing by replacing hazardous cryogenic processes with mild, Lewis acid-catalyzed reactions that utilize readily available chiral starting materials. For R&D directors and procurement strategists, this patent represents a shift towards more robust, cost-effective, and scalable chemical engineering solutions. By leveraging (R)-epichlorohydrin as a chiral source, the process ensures high optical purity while drastically simplifying the operational complexity typically associated with beta-keto ester synthesis. The following analysis details how this innovation aligns with modern GMP standards and supply chain resilience requirements.
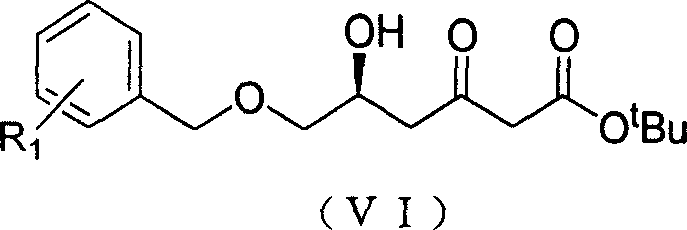
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral beta-keto esters for statin production has been plagued by severe operational constraints and safety hazards. Prior art, such as methods described in Synthesis 1995 and Angewandte Chemie, often relies on the use of ethyl chloroacetoacetate followed by chiral asymmetric reduction, necessitating the use of large quantities of butyllithium at extremely low temperatures ranging from -30°C to -45°C. These cryogenic conditions impose a heavy burden on infrastructure, requiring specialized reactors capable of maintaining sub-zero environments, which significantly increases capital expenditure (CAPEX) and energy consumption. Furthermore, the reliance on pyrophoric reagents like butyllithium introduces substantial safety risks during handling and quenching, complicating waste management protocols. Alternative routes utilizing cobalt carbonyl catalysts suffer from similar drawbacks, including the high cost of precious metal catalysts and the difficulty in removing trace metal impurities to meet stringent pharmaceutical specifications. These factors collectively hinder the ability to achieve cost reduction in pharmaceutical intermediate manufacturing at a commercial scale.
The Novel Approach
In stark contrast, the methodology disclosed in CN1923792A offers a streamlined pathway that eliminates the need for extreme thermal conditions and expensive transition metal catalysts. By initiating the synthesis with the ring-opening of (R)-epichlorohydrin using arylmethanol under Lewis acid catalysis, the process establishes the chiral framework early using a cheap, commercially abundant chiral pool material. The subsequent nucleophilic substitution with sodium cyanide proceeds efficiently in alcohol-water mixtures at moderate temperatures between 0°C and 80°C, avoiding the hazards of cryogenic operations. The final construction of the beta-keto ester skeleton is achieved through a zinc-mediated coupling reaction with tert-butyl bromoacetate, a transformation that is both chemoselective and operationally simple. This approach not only reduces the dependency on specialized equipment but also minimizes the generation of hazardous waste streams, thereby facilitating a more sustainable and economically viable production model for high-purity pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid-Catalyzed Epoxide Opening and Zinc-Mediated Coupling
The core of this synthetic strategy lies in the precise stereochemical control exerted during the initial epoxide ring-opening and the subsequent carbon-carbon bond formation. The reaction begins with the activation of (R)-epichlorohydrin by a Lewis acid catalyst, such as tin tetrachloride, iron trichloride, or boron trifluoride etherate. This coordination increases the electrophilicity of the epoxide ring, facilitating a regioselective attack by the arylmethanol nucleophile. The mechanism ensures that the chirality inherent in the starting epichlorohydrin is preserved and transferred to the resulting 1-(arylmethoxy)-3-chloro-2-propanol intermediate with high fidelity. Following this, the displacement of the chloride atom by cyanide ion proceeds via an SN2 mechanism, resulting in an inversion of configuration that is crucial for establishing the desired (S)-stereochemistry in the final product. This predictable stereochemical outcome is vital for R&D teams aiming to minimize the formation of diastereomeric impurities that could complicate downstream purification.
Furthermore, the final step involving the coupling of the protected nitrile with tert-butyl bromoacetate utilizes activated zinc to generate an organozinc species in situ. This Reformatsky-type reaction is particularly advantageous because organozinc reagents are generally more tolerant of functional groups compared to their Grignard or organolithium counterparts. The reaction conditions, typically maintained between 10°C and 100°C in solvents like tetrahydrofuran or toluene, allow for the formation of the beta-keto ester moiety without compromising the integrity of the silyl-protected hydroxyl group. The subsequent acidic workup simultaneously removes the protecting group and hydrolyzes the imine intermediate to yield the target ketone. This mechanistic elegance ensures that the impurity profile remains clean, with minimal side reactions such as self-condensation or over-alkylation, thereby supporting the production of high-purity OLED material or pharmaceutical grade intermediates with consistent quality.
How to Synthesize (S)-6-Arylmethyl-5-hydroxy-3-oxo-hexanoic Acid Tert-Butyl Ester Efficiently
The practical implementation of this synthesis route involves a sequence of four distinct chemical transformations that can be seamlessly integrated into existing multipurpose reactor trains. The process begins with the Lewis acid-catalyzed etherification, followed by cyanation, protection, and finally the zinc-mediated coupling. Each step has been optimized to maximize yield and minimize processing time, making it an ideal candidate for technology transfer. The detailed standardized synthesis steps are outlined below to guide process engineers in replicating these results.
- React (R)-epichlorohydrin with arylmethanol using a Lewis acid catalyst at 20-100°C to form (R)-1-(arylmethoxy)-3-chloro-2-propanol.
- Perform nucleophilic substitution with sodium cyanide in an alcohol-water mixture at 0-80°C to generate the chiral nitrile intermediate with inversion of configuration.
- Protect the hydroxyl group of the nitrile intermediate using silyl chlorides (e.g., TMSCl) in an organic solvent at -15 to 50°C.
- Conduct a zinc-mediated coupling reaction with tert-butyl bromoacetate at 10-100°C, followed by acidic workup to yield the final beta-keto ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers tangible strategic benefits beyond mere technical feasibility. The shift away from cryogenic conditions and pyrophoric reagents directly translates to a reduction in operational risk and insurance costs, while the use of commodity chemicals ensures a stable supply of raw materials. This stability is crucial for maintaining continuous production schedules and meeting the rigorous delivery timelines demanded by global pharmaceutical clients. By simplifying the process flow, manufacturers can achieve significant cost savings in pharmaceutical intermediate manufacturing without compromising on quality or regulatory compliance.
- Cost Reduction in Manufacturing: The elimination of expensive catalysts like cobalt carbonyl and the avoidance of energy-intensive cryogenic cooling systems result in substantial operational expenditure (OPEX) reductions. The use of zinc powder and common Lewis acids represents a fraction of the cost associated with traditional organometallic reagents, while the simplified workup procedures reduce solvent consumption and waste disposal fees. This economic efficiency allows for more competitive pricing structures in the global market for complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Sourcing (R)-epichlorohydrin and arylmethanols is far more reliable than securing specialized chiral catalysts or maintaining stocks of hazardous butyllithium solutions. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. This reliability ensures reducing lead time for high-purity pharmaceutical intermediates, enabling suppliers to respond more agilely to market demand spikes.
- Scalability and Environmental Compliance: The mild reaction temperatures and the absence of heavy metal contaminants simplify the scale-up process from pilot plant to commercial production. The aqueous workup steps and the use of recyclable solvents align with green chemistry principles, facilitating easier permitting and environmental compliance. This scalability supports the commercial scale-up of complex polymer additives or drug substances, ensuring that supply can grow in tandem with market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a transparent view of the process capabilities and limitations for potential partners.
Q: How does this synthesis route improve upon conventional methods for statin intermediates?
A: Unlike conventional methods requiring cryogenic conditions (-30°C to -45°C) and expensive catalysts like cobalt carbonyl or large amounts of butyllithium, this patented route operates at mild temperatures (20-100°C) using inexpensive Lewis acids and zinc, significantly reducing equipment costs and safety risks associated with pyrophoric reagents.
Q: What is the optical purity achievable with this manufacturing process?
A: The process utilizes (R)-epichlorohydrin as a chiral pool starting material, ensuring high stereochemical control. Experimental data in the patent demonstrates optical purity (e.e.) reaching 99.0% in key intermediates, which is critical for the efficacy and safety profile of downstream HMG-CoA reductase inhibitors.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the route is specifically designed for scalability. It avoids low-temperature steps that require specialized cryogenic reactors and uses common solvents like ethanol, THF, and toluene. The elimination of sensitive organometallic reagents simplifies waste treatment and enhances operational safety for multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-6-Arylmethyl-5-hydroxy-3-oxo-hexanoic Acid Tert-Butyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory synthesis to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of (S)-6-arylmethyl-5-hydroxy-3-oxo-hexanoic acid tert-butyl ester meets the highest international standards.
We invite global partners to collaborate with us to leverage this innovative synthesis route for your statin production needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can enhance your supply chain efficiency and product quality.
