Advanced Synthesis of Simvastatin Methyl Analogs for Commercial Pharmaceutical Manufacturing
The pharmaceutical landscape for lipid-lowering agents continues to evolve with the introduction of novel structural modifications designed to enhance efficacy and metabolic stability. Patent CN1535145A introduces a groundbreaking approach to synthesizing methyl analogs of simvastatin, specifically targeting the 3-position of the beta-hydroxypyrone ring. This innovation addresses critical limitations in existing HMG-CoA reductase inhibitors by leveraging precise stereoselective methylation techniques. For R&D directors and procurement specialists, understanding the nuances of this synthetic pathway is essential for evaluating next-generation API intermediates. The patent details a robust method that avoids complex protection-deprotection sequences, thereby streamlining the manufacturing process. By focusing on the direct methylation of simvastatin derivatives using bulky bases like Lithium Hexamethyldisilazane (Li-HMDS), the technology ensures high stereochemical purity. This report analyzes the technical merits and commercial implications of adopting this advanced synthetic route for large-scale production of high-purity statin intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for statin analogs often involve cumbersome multi-step sequences that negatively impact overall yield and cost efficiency. Conventional methods typically require the protection of sensitive hydroxyl groups on the pyrone ring before any alkylation can occur, introducing additional reagents, reaction time, and waste generation. Furthermore, achieving high stereoselectivity at the alpha-methylene position has historically been challenging, often resulting in mixtures of (3R) and (3S) isomers that require expensive and time-consuming chromatographic separation. The use of smaller bases in older methodologies frequently leads to non-selective enolization, allowing access to both faces of the pyrone ring and compromising the optical purity of the final active pharmaceutical ingredient. These inefficiencies create significant bottlenecks in the supply chain, increasing the lead time for high-purity pharmaceutical intermediates and elevating the cost of goods sold. Additionally, the reliance on harsh conditions or toxic reagents in conventional routes poses environmental compliance challenges that modern manufacturers strive to avoid.
The Novel Approach
The patented methodology described in CN1535145A represents a paradigm shift by enabling direct methylation without the need for protecting groups on the hydroxyl functionalities. This streamlined approach utilizes specific reaction conditions, including low temperatures ranging from -10°C to -60°C, to control the reactivity of the enolate intermediate. By employing sterically hindered bases such as Li-HMDS, the process achieves remarkable stereospecificity, favoring the formation of the therapeutically relevant (3R) methyl isomer. This eliminates the need for downstream purification steps to remove unwanted stereoisomers, drastically simplifying the workflow. The ability to react simvastatin directly with methylating agents like methyl iodide in a controlled nitrogen atmosphere ensures consistent quality and reproducibility. 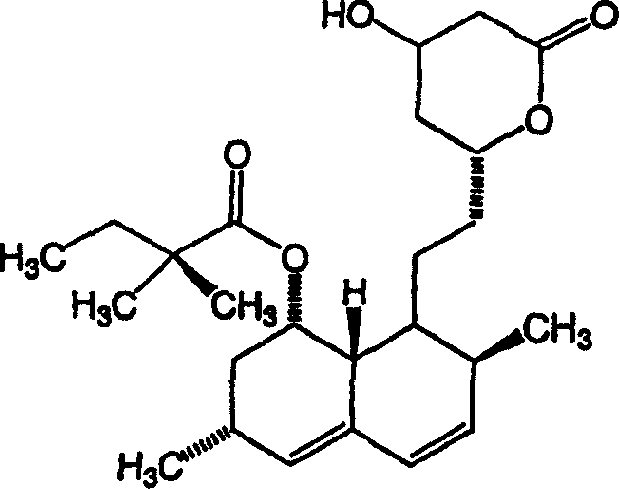 This structural clarity allows process chemists to visualize the transformation from the known simvastatin scaffold to the novel methylated derivative, highlighting the precision of the alpha-methylene functionalization.
This structural clarity allows process chemists to visualize the transformation from the known simvastatin scaffold to the novel methylated derivative, highlighting the precision of the alpha-methylene functionalization.
Mechanistic Insights into Li-HMDS Catalyzed Stereoselective Methylation
The core of this technological advancement lies in the mechanistic interaction between the bulky base and the substrate's pyrone ring. When Li-HMDS is introduced to the simvastatin solution, it deprotonates the alpha-methylene site to form a kinetic enolate. The significant steric bulk of the hexamethyldisilazane group dictates the geometry of this enolate, forcing it to adopt a conformation that shields one face of the planar system. Consequently, when the methylating agent approaches, it is directed exclusively to the less hindered face, resulting in the stereospecific formation of the (3R) configuration. This level of control is unattainable with smaller bases like lithium pyrrolidide, which allow free rotation and access to both faces, yielding a racemic or diastereomeric mixture. The preservation of the homochiral beta-hydroxypyrone ring integrity during this harsh basic treatment is also noteworthy, demonstrating the robustness of the substrate under these optimized conditions. Understanding this mechanism is vital for scaling the process, as slight deviations in temperature or base stoichiometry could alter the enolate equilibrium and compromise stereochemical outcomes.
Impurity control is inherently built into this mechanistic design, as the high stereoselectivity minimizes the generation of diastereomeric byproducts that are difficult to separate. The reaction conditions, specifically the low temperature range of -25°C to -55°C, further suppress side reactions such as over-alkylation or degradation of the sensitive lactone moiety. Post-reaction work-up involves careful quenching with water and extraction using environmentally friendlier solvents like ethyl acetate or methyl acetate, avoiding chlorinated solvents where possible. The subsequent crystallization step, utilizing polar solvents and anti-solvents, leverages the distinct solubility profile of the target methyl analog to exclude remaining impurities. 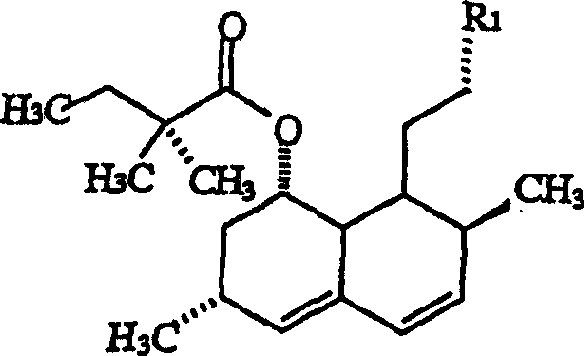 As illustrated in the general structure, the R1 group represents the modified beta-hydroxypyrone ring, which is the key pharmacophore responsible for the enhanced HMG-CoA reductase inhibitory activity. This structural modification enhances hydrophobic bonding within the enzyme's active site, potentially leading to superior clinical efficacy compared to unmethylated predecessors.
As illustrated in the general structure, the R1 group represents the modified beta-hydroxypyrone ring, which is the key pharmacophore responsible for the enhanced HMG-CoA reductase inhibitory activity. This structural modification enhances hydrophobic bonding within the enzyme's active site, potentially leading to superior clinical efficacy compared to unmethylated predecessors.
How to Synthesize Simvastatin Methyl Analog Efficiently
Implementing this synthesis requires strict adherence to the patented parameters to ensure safety and product quality. The process begins with the preparation of anhydrous tetrahydrofuran solutions and the establishment of an inert nitrogen atmosphere to prevent moisture ingress which could quench the reactive base. Operators must maintain precise temperature control during the addition of Li-HMDS and methyl iodide to manage the exothermic nature of the enolization and alkylation steps. Detailed standardized synthetic steps see the guide below.
- Prepare simvastatin solution in THF under nitrogen atmosphere and cool to -40°C to -45°C.
- Add Lithium Hexamethyldisilazane (Li-HMDS) slowly to form the enolate intermediate while maintaining strict temperature control.
- Introduce methyl iodide for methylation, followed by aqueous work-up and crystallization to isolate the high-purity 3R-methyl isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers substantial strategic benefits beyond mere technical novelty. The elimination of protection and deprotection steps significantly reduces the number of unit operations required, leading to a drastic simplification of the manufacturing workflow. This reduction in process complexity translates directly into lower operational expenditures and reduced consumption of raw materials and solvents. Furthermore, the high stereoselectivity achieved through the use of Li-HMDS minimizes the need for costly chromatographic purification, which is often a bottleneck in API production. By streamlining the synthesis, manufacturers can achieve faster batch turnover times, enhancing the overall responsiveness of the supply chain to market demands. The robustness of the reaction conditions also suggests a high degree of scalability, allowing for seamless transition from pilot plant to commercial scale production without significant re-engineering.
- Cost Reduction in Manufacturing: The removal of protecting group chemistry eliminates the cost of additional reagents and the associated waste disposal fees, resulting in significant cost savings in API intermediate manufacturing. The high yield and purity achieved in the crystallization step reduce the loss of valuable starting materials, optimizing the overall material balance. Additionally, the ability to use common organic solvents for extraction and crystallization avoids the need for specialized or expensive solvent systems. These factors collectively contribute to a more economical production model that can withstand market price fluctuations.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route reduces the dependency on a wide variety of specialized reagents, thereby mitigating supply risks associated with raw material availability. The robustness of the process ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification results. This reliability is crucial for maintaining continuous supply to downstream formulation partners and meeting regulatory commitments. A streamlined process also facilitates easier technology transfer between manufacturing sites, ensuring global supply continuity.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are manageable in large-scale reactors without requiring exotic equipment. The reduction in solvent usage and the avoidance of chlorinated solvents align with green chemistry principles, facilitating easier environmental permitting and compliance. Minimizing waste generation through high selectivity reduces the burden on waste treatment facilities, lowering the environmental footprint of the manufacturing operation. This alignment with sustainability goals enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel methyl analogs. The answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy. Understanding these details helps stakeholders make informed decisions regarding the integration of this technology into their portfolios.
Q: What is the primary advantage of using Li-HMDS over lithium pyrrolidide in this synthesis?
A: Li-HMDS offers superior steric bulk, enabling stereospecific formation of the (3R) methyl isomer by approaching the pyrone ring from the less hindered side, whereas lithium pyrrolidide yields a mixture of isomers due to its smaller size.
Q: Does this methylation process require protection of the hydroxyl groups on the pyrone ring?
A: No, the patented method specifically allows methylation at the alpha-methylene site without protecting the two hydroxyl groups of the opened pyrone ring, significantly simplifying the workflow and reducing reagent costs.
Q: How does this novel analog contribute to cholesterol management compared to existing statins?
A: The methyl substitution at the 3-position of the beta-hydroxypyrone ring enhances hydrophobic bonding energy with the drug receptor, potentially offering improved inhibitory activity against HMG-CoA reductase for treating hypercholesterolemia.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Simvastatin Methyl Analog Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent technologies into commercially viable realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the Li-HMDS mediated methylation are executed with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity pharmaceutical intermediates meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence ensures that the structural integrity and stereochemical purity of the simvastatin methyl analogs are preserved throughout the manufacturing process.
We invite you to collaborate with us to leverage this advanced technology for your next-generation lipid-lowering therapies. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals. Partner with us to secure a reliable source of high-quality API intermediates that drive innovation and efficiency in your drug development pipeline.
