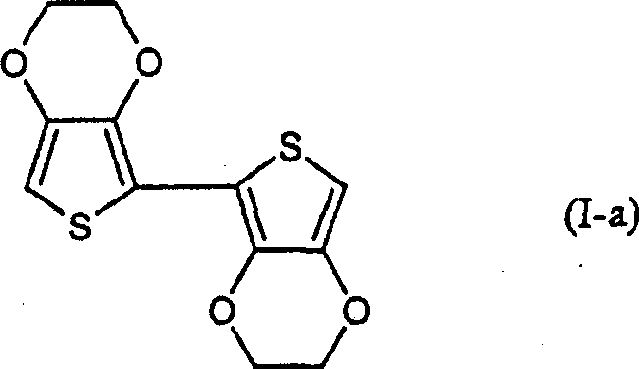
Advanced Synthesis of 2,2'-Bis(3,4-ethylenedioxythiophene) for High-Performance Conductive Polymers
The landscape of conductive polymer precursors is undergoing a significant transformation driven by the innovations disclosed in patent CN1506366A. This pivotal intellectual property introduces a robust and scalable methodology for the preparation of 2,2'-bis(3,4-alkylenedioxythiophene) derivatives, specifically targeting the critical intermediate known as the EDOT dimer. Traditionally, the synthesis of these conjugated systems has been plagued by complex organometallic requirements, but this new approach leverages a sophisticated dehydrogenation strategy that bypasses the limitations of cryogenic lithiation. For R&D directors and procurement specialists in the electronic chemicals sector, this represents a paradigm shift towards more sustainable and cost-efficient manufacturing of materials essential for OLEDs, solar cells, and antistatic coatings. The core innovation lies in the conversion of dihydro-thiophene intermediates into fully aromatic bi-thiophene structures using mild yet effective oxidizing agents, thereby unlocking higher purity and yield profiles previously unattainable.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2,2'-bis(3,4-ethylenedioxythiophene) has relied heavily on the Ullmann coupling reaction or similar organometallic pathways involving n-Butyl Lithium. These conventional routes impose severe operational constraints, primarily the necessity for extreme cryogenic conditions, often requiring cooling mixtures to maintain temperatures as low as -78°C. Such conditions not only escalate energy consumption and equipment costs but also introduce significant safety hazards associated with handling pyrophoric reagents like n-Butyl Lithium, which are hypersensitive to moisture and air. Furthermore, literature data indicates that these traditional methods suffer from mediocre yields, often hovering around 27.7% to 50%, and frequently result in products with only medium purity, necessitating extensive and costly downstream purification processes. The reliance on inert atmospheres (nitrogen or argon) throughout the entire reaction sequence further complicates the engineering controls required for commercial scale-up, making the supply chain fragile and prone to disruptions.
The Novel Approach
In stark contrast, the methodology outlined in CN1506366A offers a streamlined alternative that operates under much more forgiving conditions. By utilizing a two-step sequence involving acid-catalyzed dimerization followed by oxidative dehydrogenation, the process eliminates the need for cryogenic cooling and inert gas shielding. The reaction can be conducted at temperatures ranging from 20°C to 200°C, with preferred embodiments utilizing reflux conditions in common organic solvents like xylene or dichloromethane. This shift allows for the use of stable, non-pyrophoric reagents such as boron trifluoride etherate or trifluoroacetic acid for the initial dimerization, and quinones like chloranil for the final aromatization. The result is a dramatic improvement in process robustness, with reported yields reaching up to 69% for the target EDOT dimer, alongside a simplified workflow that is inherently safer and more amenable to large-scale continuous manufacturing.
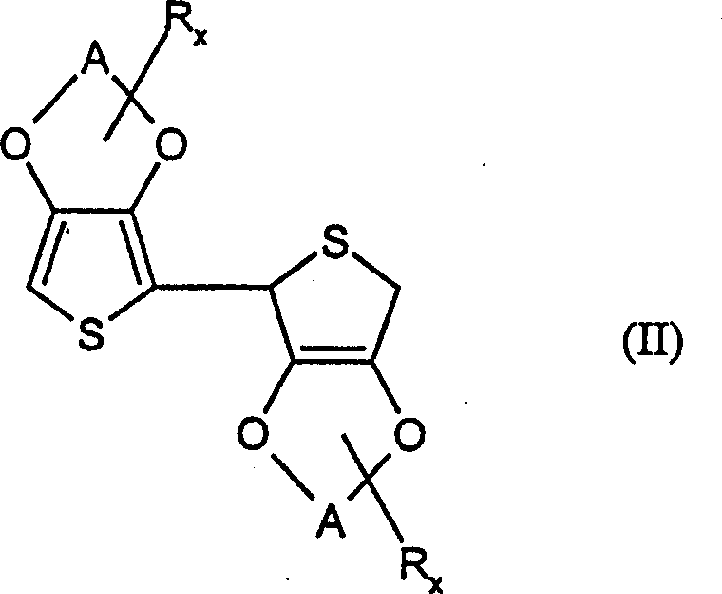
Mechanistic Insights into Acid-Catalyzed Dimerization and Oxidative Aromatization
The mechanistic elegance of this synthesis lies in its bifurcated pathway, beginning with the activation of the thiophene monomer. In the first stage, a Lewis acid or protonic acid catalyst facilitates the electrophilic attack of one 3,4-alkylenedioxythiophene molecule onto another, forming a 2,5-dihydro-intermediate. This step is crucial as it establishes the carbon-carbon bond between the two heterocyclic rings without destroying the sensitive alkylenedioxy functionality. Unlike lithiation, which can indiscriminately deprotonate various sites leading to regio-isomeric impurities, the acid-catalyzed route offers superior regioselectivity. The subsequent dehydrogenation step is equally critical; it involves the removal of hydrogen atoms from the dihydro-intermediate to restore aromaticity. This is achieved through hydride transfer mechanisms when using quinones, or via halogenation-elimination sequences when using agents like bromine or N-bromosuccinimide. The choice of dehydrogenating agent is paramount, as it must be strong enough to drive aromatization but mild enough to prevent over-oxidation of the thiophene ring into polymeric species or sulfones.
Impurity control is intrinsically built into this mechanism through the careful selection of reaction stoichiometry and temperature. The patent emphasizes the use of equimolar or slightly excess amounts of the dehydrogenating agent (1.0 to 1.2 equivalents) to minimize side reactions. Furthermore, the workup procedure involves washing with alkaline solutions, such as 4% potassium hydroxide, which effectively neutralizes any residual acid catalyst and removes acidic byproducts generated during the oxidation. This ensures that the final crude product is of high quality, often requiring only simple recrystallization or a brief column chromatography pass to achieve electronic-grade purity. The ability to tolerate functional groups on the alkylene bridge, such as hydroxyl or alkyl substituents, without interference further underscores the chemoselectivity of this catalytic system, providing a versatile platform for synthesizing a wide array of specialized monomers.
How to Synthesize 2,2'-Bis(3,4-ethylenedioxythiophene) Efficiently
The practical execution of this synthesis is designed for reproducibility and safety in a pilot or plant setting. The process begins with the preparation of the dihydro-dimer intermediate, which can be isolated or used in situ, followed by the critical dehydrogenation step in a high-boiling solvent. Detailed standard operating procedures for temperature control, reagent addition rates, and quenching protocols are essential to maximize the 69% yield potential demonstrated in the patent examples. For a comprehensive guide on the specific molar ratios, solvent choices, and purification techniques validated by experimental data, please refer to the standardized synthesis protocol below.
- Perform acid-catalyzed dimerization of 3,4-alkylenedioxythiophene monomers using Lewis acids (e.g., BF3) or protonic acids (e.g., TFA) at 0-40°C to form the dihydro-intermediate.
- React the resulting 2-(3,4-alkylenedioxy-2,5-dihydro-2-thienyl)-3,4-alkylenedioxythiophene with a dehydrogenating agent such as chloranil or DDQ under reflux conditions.
- Purify the final aromatic product via filtration, alkaline washing to remove acidic byproducts, and column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of cryogenic infrastructure and hazardous organolithium reagents translates directly into a significant reduction in capital expenditure (CAPEX) and operational expenditure (OPEX). Facilities no longer require specialized low-temperature reactors or extensive safety systems for pyrophoric material handling, thereby lowering the barrier to entry for production and reducing the overall cost of goods sold. Moreover, the use of commodity chemicals like chloranil and common organic solvents ensures a stable and resilient supply chain, mitigating the risks associated with sourcing specialized, single-source reagents. This stability is crucial for maintaining consistent delivery schedules to downstream customers in the volatile electronics market.
- Cost Reduction in Manufacturing: The removal of expensive and dangerous reagents like n-Butyl Lithium drastically simplifies the bill of materials. Additionally, the ability to run reactions at ambient or reflux temperatures rather than -78°C leads to substantial energy savings. The higher yields achieved (nearly doubling the efficiency of some prior art methods) mean less raw material waste and lower disposal costs for hazardous byproducts, contributing to a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: By relying on robust chemistry that tolerates air and moisture better than organometallic alternatives, the risk of batch failure due to environmental excursions is minimized. This reliability ensures a consistent flow of high-purity intermediates, preventing bottlenecks in the production of finished conductive polymers. The scalability of the reflux-based process allows for seamless transition from kilogram-scale R&D batches to multi-ton commercial production without the need for process re-engineering.
- Scalability and Environmental Compliance: The process generates fewer hazardous wastes compared to lithium-based routes, simplifying effluent treatment and regulatory compliance. The solvents used are standard and easily recoverable, supporting green chemistry initiatives. Furthermore, the simplicity of the workup—often involving simple filtration and washing—reduces processing time and labor intensity, enabling faster turnaround times and improved responsiveness to market demand fluctuations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. They are derived from the specific advantages and procedural details outlined in the patent documentation to assist decision-makers in evaluating the feasibility of adoption. Understanding these nuances is key to leveraging the full potential of this synthetic route for your specific application needs.
Q: Why is the new dehydrogenation method superior to traditional Ullmann coupling for EDOT dimer production?
A: The new method eliminates the need for cryogenic conditions (-78°C) and hazardous organolithium reagents like n-Butyl Lithium. It operates at atmospheric pressure and higher temperatures (reflux), significantly improving safety, scalability, and yield (up to 69% vs 27-50% traditionally).
Q: What dehydrogenating agents are compatible with this synthesis route?
A: The patent specifies several effective agents including quinones (specifically 2,3,5,6-tetrachloro-1,4-benzoquinone/chloranil and DDQ), sulfur, bromine, N-bromosuccinimide, and hydrogen peroxide. Chloranil is highlighted as particularly preferred for high yields.
Q: Can this method produce substituted derivatives beyond the standard ethylene bridge?
A: Yes, the method demonstrates broad applicability. It allows for the synthesis of derivatives where the ethylene bridge is replaced by other alkylidene groups (C2-C4) or functionalized with hydroxyalkyl groups, which was difficult or impossible with previous lithiation-based methods due to side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Bis(3,4-ethylenedioxythiophene) Supplier
As the global demand for high-performance conductive polymers continues to surge, securing a reliable source of high-purity precursors is more critical than ever. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN1506366A to deliver superior quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on our stringent purity specifications. With our rigorous QC labs and state-of-the-art manufacturing facilities, we guarantee that every batch of 2,2'-bis(3,4-ethylenedioxythiophene) meets the exacting standards required for next-generation electronic applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your product performance and bottom line. Let us be your partner in innovation, delivering the chemical building blocks that power the future of electronics.
