Advanced Purification Technology for Commercial Scale Ranitidine Bismuth Citrate Production
Introduction to Advanced Impurity Control in Anti-Ulcer API Manufacturing
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity profiles of critical therapeutic agents, particularly for complex salts like Ranitidine Bismuth Citrate. Patent CN103896888A introduces a groundbreaking preparation method that addresses long-standing challenges in impurity management during the synthesis of this potent anti-ulcer medication. The core innovation lies not merely in the reaction conditions but in the rigorous pre-treatment of the crystallization solvent, specifically absolute ethanol, which is traditionally a hidden source of contamination. By implementing a specialized scavenging protocol using sodium salts, manufacturers can drastically reduce the formation of specific nitro-containing degradation products that have historically plagued batch consistency. This technical advancement ensures that the final active pharmaceutical ingredient meets stringent pharmacopoeial standards with a significant safety margin, positioning it as a superior choice for high-quality generic and branded drug formulations globally.
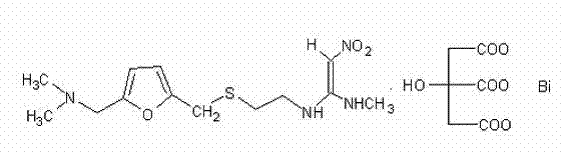
Furthermore, the disclosed process optimizes the upstream synthesis by eliminating the need to isolate the unstable Ranitidine free base as a discrete solid intermediate. Instead, the methodology utilizes an in-situ generation approach where Ranitidine Hydrochloride is converted directly in the aqueous phase before reacting with bismuth citrate. This strategic modification simplifies the workflow, reduces the consumption of hazardous organic extraction solvents, and minimizes the environmental footprint associated with solvent recovery systems. For procurement and supply chain leaders, this translates to a more streamlined production cycle that is less susceptible to bottlenecks caused by solvent handling regulations. The combination of solvent engineering and process intensification represents a holistic upgrade to the manufacturing landscape of gastrointestinal therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
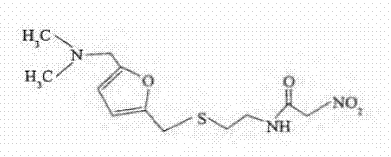
Traditional manufacturing routes for Ranitidine Bismuth Citrate have historically relied on the use of pre-isolated Ranitidine free base or standard commercial grades of absolute ethanol without further purification. A significant drawback of using the isolated free base is its inherent instability and the high cost associated with its preparation, which typically involves multiple extraction steps using volatile organic compounds. These extra unit operations not only inflate the raw material costs but also introduce variability in the physical form of the intermediate, which can affect the subsequent salt formation kinetics. Moreover, conventional recrystallization processes often utilize untreated industrial ethanol, which contains trace amounts of aldehydes and ketones as impurities. These trace contaminants are highly reactive towards the nitro-ethylene diamine moiety of the ranitidine molecule, leading to the formation of N-[2-[5-[(dimethylamino)methyl]-2-furyl]methylthio]ethyl]-2-nitroacetamide, a critical impurity that is difficult to remove once formed.
The Novel Approach
The novel approach detailed in the patent data fundamentally shifts the paradigm by focusing on the quality of the recrystallization medium and the efficiency of the salt formation step. By treating commercial absolute ethanol with specific sodium salts such as sodium metabisulfite or sodium bisulfite, the process effectively scavenges the reactive carbonyl impurities before they can interact with the drug substance. This pre-emptive purification step ensures that the crystallization environment is chemically inert regarding the formation of the problematic acetamide derivative. Additionally, the process bypasses the isolation of the free base by reacting Ranitidine Hydrochloride with a weak base like ammonia water directly in the presence of bismuth citrate. This one-pot style modification reduces the number of filtration and drying steps, thereby lowering energy consumption and labor costs while simultaneously improving the overall yield and purity profile of the final bismuth salt complex.
Mechanistic Insights into Solvent Scavenging and Impurity Suppression
The chemical mechanism underpinning this invention is rooted in nucleophilic addition and scavenging reactions within the solvent matrix. Trace impurities in industrial ethanol, primarily acetaldehyde and other aldehydes, act as electrophiles that can react with the secondary amine groups or facilitate oxidative degradation pathways involving the nitro group of the ranitidine structure. When sodium metabisulfite or similar sulfite salts are introduced into the ethanol, they react rapidly with these aldehydes to form stable bisulfite addition compounds. This reaction effectively removes the electrophilic species from the solution, rendering the solvent safe for the sensitive nitro-ethylene diamine pharmacophore. Consequently, the pathway leading to the formation of the N-nitroacetamide impurity is blocked at the source, preventing the contamination of the crystal lattice during the critical precipitation phase.
Beyond solvent treatment, the reaction mechanism for the salt formation itself is optimized through pH control and temperature management. By adjusting the pH of the Ranitidine Hydrochloride solution to a weakly alkaline range using agents like ammonium hydroxide, the free base is generated in situ without precipitating as a solid. This soluble free base is then immediately available to coordinate with the bismuth citrate complex at elevated temperatures of 90 to 95°C. The elevated temperature ensures complete dissolution and reaction kinetics favoring the formation of the thermodynamically stable bismuth salt. Following the reaction, controlled cooling to 50 to 60°C combined with activated carbon treatment allows for the adsorption of any colored by-products or residual organic impurities. The subsequent addition of the treated ethanol induces supersaturation in a controlled manner, promoting the growth of large, pure crystals that exclude the remaining trace impurities from the solid phase, resulting in a product with exceptional chemical purity.
How to Synthesize Ranitidine Bismuth Citrate Efficiently
Implementing this synthesis route requires precise control over solvent quality and reaction parameters to achieve the reported purity levels. The process begins with the preparation of the scavenged ethanol, followed by the aqueous conversion of the hydrochloride salt, and concludes with a carefully managed crystallization sequence. Operators must ensure that the sodium salt treatment of the ethanol is verified using a colorimetric detection reagent to confirm the absence of reactive aldehydes before proceeding to the main reaction. This attention to detail in the preparatory stages is what differentiates a standard batch from a high-purity pharmaceutical grade batch. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- Pre-treat industrial grade absolute ethanol with sodium salts such as sodium metabisulfite to scavenge trace aldehyde and ketone impurities.
- React Ranitidine Hydrochloride with a weak base in aqueous solution to generate the free base in situ, avoiding organic extraction.
- Combine the in-situ generated base with bismuth citrate at 90-95°C, followed by decolorization and crystallization in the treated ethanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers substantial strategic advantages beyond mere technical compliance. The elimination of the Ranitidine free base isolation step represents a significant reduction in processing time and resource allocation. By removing the need for organic solvent extraction and the subsequent recovery of those solvents, the manufacturing facility can operate with a smaller equipment footprint and reduced utility consumption. This streamlining of the process flow directly contributes to a more resilient supply chain, as there are fewer unit operations that can potentially fail or cause delays. Furthermore, the reliance on treated industrial ethanol rather than expensive high-purity specialty solvents helps in stabilizing raw material costs, providing a buffer against market volatility in the solvent sector.
- Cost Reduction in Manufacturing: The process achieves cost efficiency primarily by consolidating reaction steps and minimizing solvent usage. By avoiding the isolation of the intermediate free base, the manufacturer saves on the capital and operational expenses associated with extraction columns, evaporators, and drying ovens. The ability to use treated industrial-grade ethanol instead of premium analytical-grade solvents further drives down the bill of materials without compromising product quality. Additionally, the reduced generation of waste solvent streams lowers the costs associated with environmental compliance and hazardous waste disposal, creating a leaner and more economically viable production model.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently reduces the risk of supply chain disruptions. With fewer intermediate handling steps, there is less opportunity for material loss or cross-contamination, leading to more predictable batch yields. The use of readily available industrial chemicals like sodium metabisulfite and ammonia water ensures that the supply of critical reagents remains stable and unaffected by the shortages that often plague specialized fine chemical intermediates. This robustness allows for more accurate production planning and shorter lead times, enabling the supplier to respond more agilely to fluctuations in market demand for anti-ulcer medications.
- Scalability and Environmental Compliance: The methodology is designed with industrial scalability in mind, utilizing standard unit operations that are easily transferred from pilot plant to full commercial scale. The reduction in organic solvent usage aligns perfectly with modern green chemistry principles and increasingly strict environmental regulations. By minimizing the volume of volatile organic compounds released into the atmosphere or requiring incineration, the process facilitates easier permitting and ongoing regulatory compliance. This environmental stewardship not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key criterion for selection by major multinational pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the method's capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing networks or for procurement teams assessing supplier qualifications.
Q: How does the new method control the maximum single impurity in Ranitidine Bismuth Citrate?
A: The method treats absolute ethanol with sodium salts to remove trace impurities that react with the drug substance, reducing the maximum single impurity content from over 1.0% to below 0.5% or even 0.3%.
Q: Why is avoiding the isolation of Ranitidine free base beneficial for manufacturing?
A: Skipping the isolation step eliminates the need for organic solvent extraction and subsequent solvent recovery, significantly reducing operational complexity, environmental pollution, and production costs.
Q: What specific reagents are used to purify the crystallization solvent?
A: Sodium salts such as sodium metabisulfite, sodium bisulfite, or sulfothiorine are added to commercial industrial-grade ethanol, stirred, and filtered to create a qualified low-impurity solvent.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ranitidine Bismuth Citrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate solvent treatment and in-situ reaction protocols are executed with precision at every scale. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities at the parts-per-million level, guaranteeing that every batch of Ranitidine Bismuth Citrate meets or exceeds the highest international pharmacopoeial standards. Our commitment to quality assurance means that the benefits of this patented impurity control strategy are fully realized in the final product delivered to your facility.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from recent batches and comprehensive route feasibility assessments to demonstrate how our optimized process can enhance your product quality while reducing overall manufacturing costs. Let us be your trusted partner in delivering high-purity gastrointestinal therapeutics to the global market.
