Advanced Synthesis of Carbazole-Thiadiazole Derivatives for Commercial Pharmaceutical Applications
Advanced Synthesis of Carbazole-Thiadiazole Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for heterocyclic compounds that combine structural rigidity with biological activity. A recent breakthrough detailed in patent CN111217805B introduces a highly efficient method for preparing (E)2-(2-(9-alkyl)carbazole-3-)vinyl-5-amino-1,3,4-thiadiazole derivatives. These molecules represent a sophisticated fusion of the electron-rich carbazole system and the bioactive 1,3,4-thiadiazole ring, creating a large conjugated system with significant potential in medicinal chemistry. The structural versatility of this scaffold allows for extensive modification at the 9-position of the carbazole ring, enabling the tuning of physicochemical properties for specific drug delivery or material science applications. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such high-value scaffolds is critical for meeting the evolving demands of R&D teams globally.
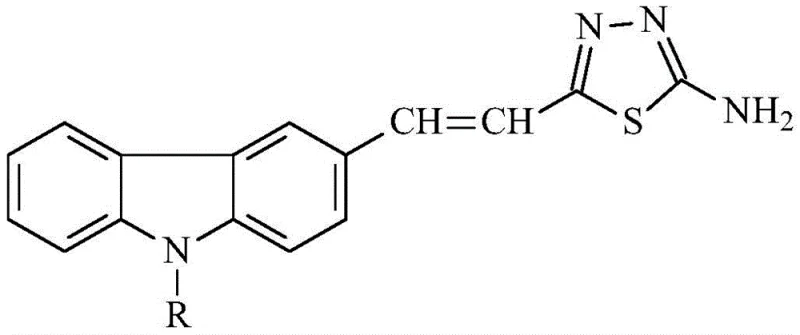
This specific class of compounds is not merely an academic curiosity; it addresses a tangible need for novel antibacterial agents. The integration of the thiadiazole moiety into the carbazole backbone enhances the molecule's ability to interact with biological targets, offering a promising avenue for combating resistant bacterial strains. The patent outlines a streamlined approach that bypasses the multi-step complexities often associated with constructing such fused heterocyclic systems. By focusing on a direct cyclization strategy, the methodology significantly reduces the operational burden on manufacturing facilities, aligning perfectly with the goals of cost reduction in API manufacturing. For procurement managers and supply chain heads, the implication is clear: a route that is both chemically elegant and operationally simple translates to better supply security and lower overall production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex thiadiazole-carbazole hybrids has been plagued by inefficient methodologies that rely on harsh conditions and poor atom economy. Conventional routes often require the pre-functionalization of the carbazole ring followed by separate coupling steps to introduce the thiadiazole unit, leading to cumulative yield losses at each stage. Furthermore, many historical methods utilize expensive transition metal catalysts or toxic solvents that necessitate rigorous purification protocols to meet pharmaceutical grade standards. These legacy processes frequently suffer from long reaction times and difficult work-up procedures, where the separation of the desired product from tarry byproducts becomes a major bottleneck. Such inefficiencies not only drive up the cost of goods sold (COGS) but also create significant environmental liabilities due to the generation of hazardous waste streams. For a commercial scale-up of complex pharmaceutical intermediates, these factors render traditional methods economically unviable and environmentally unsustainable.
The Novel Approach
In stark contrast, the methodology disclosed in CN111217805B offers a transformative solution by utilizing a direct condensation-cyclization strategy. The process leverages readily available starting materials, specifically (E)1-(9-alkyl-carbazole-3-)-acrylic acid and thiosemicarbazide, to construct the target scaffold in a single pot. The use of phosphorus oxychloride (POCl3) or concentrated hydrochloric acid acts dually as the solvent and the dehydrating catalyst, eliminating the need for additional reagents and simplifying the reaction matrix. This approach ensures that the reaction proceeds with high conversion rates, consistently delivering yields above 90% regardless of the alkyl chain length at the 9-position. The simplicity of the work-up, involving a straightforward pH adjustment and filtration, drastically reduces the time required for downstream processing. This represents a paradigm shift towards greener chemistry, facilitating high-purity pharmaceutical intermediates production with minimal environmental footprint.

Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this synthetic innovation lies in the acid-mediated cyclization mechanism that efficiently closes the thiadiazole ring onto the vinyl-carbazole framework. The reaction initiates with the activation of the carboxylic acid group on the carbazole derivative by the strong acid medium (POCl3 or HCl), forming a highly reactive acyl chloride or protonated species in situ. This activated intermediate then undergoes nucleophilic attack by the terminal amino group of thiosemicarbazide. Following the initial amide bond formation, an intramolecular cyclization occurs, driven by the dehydration capability of the reaction medium, which removes water to aromatize the 1,3,4-thiadiazole ring. The preservation of the (E)-configuration of the vinyl linker throughout this vigorous acidic treatment is particularly noteworthy, demonstrating the robustness of the conjugated system under the specified reflux conditions. This mechanistic pathway avoids the formation of unstable intermediates that often degrade in conventional thermal processes.
From an impurity control perspective, the protocol is designed to minimize side reactions that could compromise the purity profile of the final active pharmaceutical ingredient (API) intermediate. The use of Thin Layer Chromatography (TLC) for real-time monitoring ensures that the reaction is quenched precisely upon completion, preventing over-reaction or decomposition of the product. The subsequent neutralization step using ammonia water is critical; by adjusting the pH to the 8-9 range, the product precipitates selectively while soluble inorganic salts and unreacted starting materials remain in the aqueous phase. This crystallization-induced purification effectively strips away polar impurities and residual acids without the need for column chromatography. Such inherent purity is essential for reducing lead time for high-purity pharmaceutical intermediates, as it simplifies the quality control (QC) release testing and ensures compliance with stringent regulatory specifications.
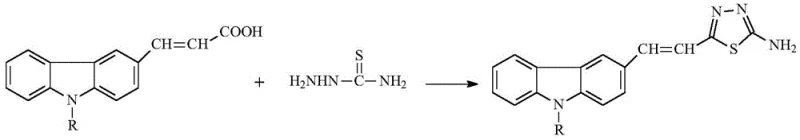
How to Synthesize (E)2-(2-(9-alkyl)carbazole-3-)vinyl-5-amino-1,3,4-thiadiazole Efficiently
Implementing this synthesis on an industrial scale requires careful attention to reagent stoichiometry and thermal management to maximize safety and yield. The process begins with the precise charging of dry equipment to prevent premature hydrolysis of the dehydrating agent, followed by the controlled addition of reactants to manage exotherms. The reaction mixture is then subjected to reflux, a standard unit operation that ensures uniform heating and efficient mass transfer throughout the bulk solution. Once the conversion is confirmed via analytical monitoring, the cooling and neutralization phases must be executed with precision to ensure the formation of a filterable solid with the desired crystal habit. For a detailed breakdown of the specific molar ratios, temperatures, and safety precautions required for this transformation, please refer to the standardized operating procedure outlined below.
- Charge a dry reactor with (E)1-(9-alkyl-carbazole-3-)-acrylic acid, thiosemicarbazide, and phosphorus oxychloride (POCl3) or concentrated HCl.
- Heat the mixture to reflux conditions and monitor reaction progress via TLC until the starting material is fully consumed.
- Cool the reaction mixture in an ice bath, adjust pH to 8-9 using ammonia water to precipitate the solid product, then filter and dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers substantial strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials; both the carbazole-acrylic acid precursors and thiosemicarbazide are commodity chemicals available from multiple global sources, mitigating the risk of single-source dependency. Furthermore, the elimination of expensive transition metal catalysts and exotic ligands removes a significant cost driver from the bill of materials, directly contributing to cost reduction in pharmaceutical intermediate manufacturing. The high atom economy of the cyclization step means that less raw material is wasted as byproduct, optimizing the input-output ratio and reducing the volume of waste that requires costly disposal. These factors combined create a resilient and cost-effective sourcing strategy for long-term production contracts.
- Cost Reduction in Manufacturing: The process utilizes inexpensive mineral acids like concentrated HCl or POCl3 as dual-purpose solvents and catalysts, removing the need for costly organic solvents or noble metal catalysts. This substitution significantly lowers the variable cost per kilogram of the final product. Additionally, the high yield (>90%) minimizes the loss of valuable carbazole starting materials, ensuring that the majority of input costs are converted into saleable product rather than waste. The simplified work-up procedure, which relies on precipitation and filtration rather than energy-intensive distillation or chromatography, further reduces utility consumption and labor hours per batch.
- Enhanced Supply Chain Reliability: By relying on a robust chemical transformation that tolerates a wide range of alkyl substituents, manufacturers can maintain production continuity even if specific grades of starting materials fluctuate in availability. The use of standard industrial equipment (three-neck flasks, reflux condensers, filtration units) means that the process can be easily transferred between different manufacturing sites without requiring specialized hardware. This flexibility ensures that supply chains remain agile and responsive to market demands, preventing bottlenecks that often occur with proprietary or highly specialized synthetic technologies.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-ton commercial production without significant re-engineering. The ability to use aqueous work-ups and the absence of heavy metal residues simplify the wastewater treatment process, aiding in compliance with increasingly strict environmental regulations. This 'green' aspect of the synthesis not only reduces the environmental liability of the manufacturing site but also aligns with the sustainability goals of major pharmaceutical buyers who prioritize eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole-thiadiazole derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this chemistry into your existing product pipeline or research portfolio.
Q: What is the typical yield for this carbazole-thiadiazole cyclization?
A: According to patent CN111217805B, the process consistently achieves isolated yields exceeding 90% across various alkyl substituents, indicating high efficiency and minimal byproduct formation.
Q: Can concentrated HCl replace POCl3 in this synthesis?
A: Yes, the patent explicitly states that concentrated hydrochloric acid can serve as an effective alternative to phosphorus oxychloride, acting as both solvent and catalyst while potentially reducing corrosive waste handling costs.
Q: What is the biological activity profile of these derivatives?
A: The synthesized compounds demonstrate significant bacteriostatic activity against both Gram-positive and Gram-negative bacteria, making them valuable candidates for developing novel antimicrobial agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)2-(2-(9-alkyl)carbazole-3-)vinyl-5-amino-1,3,4-thiadiazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating innovative patent chemistry into reliable commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields reported in the laboratory are maintained at an industrial scale. We operate with stringent purity specifications and utilize rigorous QC labs to verify that every batch of (E)2-(2-(9-alkyl)carbazole-3-)vinyl-5-amino-1,3,4-thiadiazole meets the exacting standards required for downstream drug development. Our commitment to quality assurance means that you receive a consistent, high-performance intermediate that accelerates your timeline from discovery to clinical trials.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce your overall project costs. Let us be your partner in bringing these potent antibacterial agents to the global market.
