Advanced Asymmetric Oxidation Strategy for High-Purity R-Type Chiral Sulfoxide Commercialization
Advanced Asymmetric Oxidation Strategy for High-Purity R-Type Chiral Sulfoxide Commercialization
The pharmaceutical landscape for anticoagulants has been significantly reshaped by the introduction of Ticagrelor, a potent P2Y12 receptor antagonist. However, the rigorous quality standards required for such active pharmaceutical ingredients demand precise control over impurity profiles, particularly chiral impurities which can exhibit distinct biological activities or toxicities. Patent CN110746428A, published in early 2020, addresses a critical gap in the synthesis of Ticagrelor-related substances by disclosing a robust preparation method for R-type chiral sulfoxide compounds. This technology represents a substantial leap forward in stereoselective synthesis, moving away from traditional racemic production towards highly specific asymmetric catalysis. For R&D directors and process chemists, this patent offers a validated pathway to generate reference standards and intermediates with exceptional optical purity, facilitating deeper biological profiling and regulatory compliance. The methodology leverages a sophisticated metal-organic catalytic system that operates under mild conditions, ensuring that the delicate chiral centers remain intact throughout the multi-step transformation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfoxide derivatives related to Ticagrelor has been plagued by the formation of racemic mixtures, presenting a formidable challenge for purification and characterization. Conventional oxidation methods often lack the necessary stereocontrol, resulting in a 1:1 mixture of R and S enantiomers that are notoriously difficult to separate using standard chromatographic techniques on a large scale. This lack of selectivity not only depresses the overall yield of the desired isomer by nearly half but also introduces significant complexity into the downstream processing workflow. Furthermore, existing literature, such as CN105237540A, describes routes that fail to distinguish between the S-type and R-type peaks in HPLC detection, rendering them inadequate for producing high-purity reference materials needed for rigorous safety testing. The inability to access pure single enantiomers hampers the ability to fully understand the pharmacological profile of potential impurities, creating a bottleneck in the drug development lifecycle.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN110746428A utilizes a meticulously engineered reaction sequence that prioritizes stereochemical integrity from the outset. By strategically ordering the synthetic steps—specifically performing nucleophilic substitution prior to the critical oxidation step—the process avoids the high-temperature conditions that typically induce racemization. The core innovation lies in the deployment of a chiral catalytic oxidation system comprising a metal organic catalyst, a chiral reagent, and water in precise molar ratios. This system enables the direct conversion of the sulfide precursor into the R-type sulfoxide with remarkable efficiency. 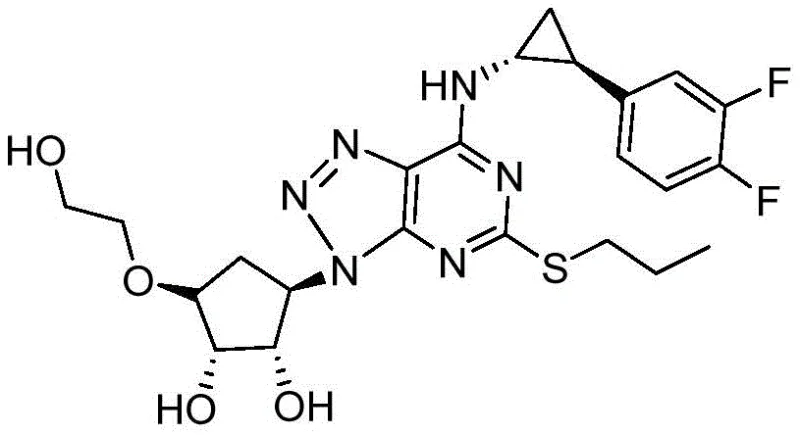 As illustrated in the reaction scheme, the pathway flows logically from cyclization to substitution, followed by the asymmetric oxidation and final deprotection. This streamlined route eliminates the need for post-synthesis chiral resolution, thereby drastically simplifying the manufacturing process and enhancing the overall atom economy of the synthesis.
As illustrated in the reaction scheme, the pathway flows logically from cyclization to substitution, followed by the asymmetric oxidation and final deprotection. This streamlined route eliminates the need for post-synthesis chiral resolution, thereby drastically simplifying the manufacturing process and enhancing the overall atom economy of the synthesis.
Mechanistic Insights into Vanadium-Catalyzed Asymmetric Oxidation
The heart of this technological breakthrough is the asymmetric oxidation step, which employs a vanadium-based catalytic system to achieve high enantioselectivity. The patent data reveals that among various metal organic catalysts tested, including molybdenum, titanium, and iron complexes, vanadyl acetylacetonate (VO(acac)2) demonstrated superior performance. When paired with (S,S)-diethyl tartrate as the chiral ligand and water as a co-catalyst, this system creates a highly organized transition state that favors the formation of the R-enantiomer. The reaction is conducted at cryogenic temperatures, specifically between 0°C and 4°C, which is crucial for suppressing thermal racemization and maintaining the kinetic control necessary for high optical purity. The molar ratio of the catalyst components is equally critical, with an optimal ratio of 1:2:10 (catalyst:chiral reagent:water) yielding the best results. This precise tuning of the catalytic environment ensures that the oxygen transfer to the sulfur atom occurs with strict facial selectivity, a feat that is difficult to achieve with non-chiral oxidants.
Beyond mere yield optimization, this mechanistic approach provides robust control over the impurity profile, a key concern for regulatory approval. The mild reaction conditions prevent the degradation of sensitive functional groups, such as the triazole ring and the cyclopropylamine moiety, which are prone to decomposition under harsher oxidative conditions. By avoiding strong acids or bases during the oxidation phase, the process minimizes the formation of side products that could complicate purification. The subsequent hydrolysis step is also carefully managed, using dilute inorganic acid in an alcohol solvent at moderate temperatures (20-30°C) to remove protecting groups without compromising the newly formed chiral sulfoxide bond. This holistic control over reaction parameters ensures that the final product meets stringent purity specifications, often exceeding 99.7%, which is essential for its use as a reference standard or a high-value intermediate in the synthesis of next-generation anticoagulants.
How to Synthesize R-Type Chiral Sulfoxide Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing this high-value intermediate. The process begins with the diazotization and cyclization of an amino-purine derivative to form the triazole core, followed by the installation of the chiral cyclopropylamine side chain via nucleophilic substitution. The pivotal oxidation step requires careful attention to temperature and stoichiometry to maximize enantiomeric excess. Finally, the removal of the acetonide protecting group yields the target diol sulfoxide. While the general workflow is straightforward, the success of the operation hinges on the precise execution of the catalytic oxidation parameters described in the technical disclosure. For laboratory and pilot plant teams looking to replicate this success, adhering to the specific molar ratios and temperature gradients is non-negotiable.
- Perform diazotization and cyclization of the amine precursor in glacial acetic acid with sodium nitrite at controlled low temperatures to form the triazole core.
- Execute nucleophilic substitution using a chiral cyclopropylamine derivative in toluene with potassium carbonate to install the chiral side chain.
- Conduct the critical asymmetric oxidation using a vanadium-based catalyst system with a chiral tartrate ligand at 0°C to ensure high enantioselectivity.
- Finalize the synthesis by hydrolyzing the protecting groups under acidic conditions in an alcohol solvent to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this novel synthesis route offers compelling economic and operational advantages over traditional racemic methods. The primary value driver is the elimination of chiral resolution steps, which are notoriously expensive and yield-limiting in pharmaceutical manufacturing. By directly synthesizing the desired R-isomer, the process effectively doubles the theoretical yield compared to racemic synthesis followed by separation, leading to substantial cost savings in raw material consumption. Furthermore, the use of commercially available and relatively inexpensive catalysts like vanadyl acetylacetonate reduces the dependency on exotic, high-cost proprietary reagents. This shift not only lowers the bill of materials but also mitigates supply risk associated with single-source specialty chemicals. The mild reaction conditions further contribute to cost reduction by lowering energy consumption for heating and cooling, making the process more sustainable and economically viable for large-scale production.
- Cost Reduction in Manufacturing: The streamlined synthetic route significantly reduces manufacturing costs by removing the need for complex chiral chromatography or crystallization resolutions. Traditional methods often suffer from a maximum theoretical yield of 50% for the desired enantiomer, whereas this asymmetric catalytic approach pushes yields much higher, optimizing resource utilization. Additionally, the avoidance of harsh reaction conditions reduces equipment wear and tear and lowers utility costs associated with extreme temperature control. The high purity of the crude product also minimizes the number of purification cycles required, saving both time and solvent costs. These cumulative efficiencies translate into a more competitive pricing structure for the final intermediate, allowing for better margin management in the supply chain.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of robust, scalable chemistry that relies on stable and widely available reagents. Unlike processes dependent on sensitive biocatalysts or unstable organometallics, this vanadium-catalyzed system is tolerant of minor variations in operating conditions, ensuring consistent batch-to-batch quality. The ability to produce high-purity material without complex separation steps shortens the overall production cycle time, enabling faster response to market demand fluctuations. This agility is crucial for maintaining continuous supply lines for critical API intermediates, reducing the risk of stockouts that can disrupt downstream drug manufacturing. Moreover, the simplified process flow reduces the number of unit operations, decreasing the potential points of failure in the production line.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process aligns well with green chemistry principles. The high atom economy and reduced solvent usage for purification lower the overall environmental footprint of the manufacturing process. The mild conditions reduce the generation of hazardous waste streams associated with aggressive oxidants or extreme pH adjustments. Scaling this process from kilogram to tonnage levels is facilitated by the straightforward nature of the reaction steps, which do not require specialized high-pressure or cryogenic equipment beyond standard industrial capabilities. This ease of scale-up ensures that supply can be rapidly expanded to meet commercial needs without extensive re-engineering of the production facility, supporting long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance.
Q: Why is the specific order of substitution before oxidation critical in this synthesis?
A: Performing substitution before oxidation prevents racemization. If oxidation occurs first, the subsequent high-temperature substitution step can cause conformational transformation, leading to a racemic mixture and significantly lower optical purity.
Q: What is the optimal catalyst system for achieving high enantiomeric excess?
A: The patent identifies vanadyl acetylacetonate (VO(acac)2) combined with (S,S)-diethyl tartrate and water in a specific molar ratio (1:2:10) as the superior system, yielding over 67% with >99% purity.
Q: How does this method improve upon previous Ticagrelor impurity synthesis routes?
A: Previous methods produced racemic sulfoxide mixtures that were difficult to separate. This novel route directly synthesizes the specific R-type isomer with high stereo-selectivity, eliminating the need for complex chiral resolution steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-Type Chiral Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of safe and effective cardiovascular therapies. Our team of expert process chemists has extensively analyzed the technology disclosed in CN110746428A and is fully equipped to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of the vanadium catalytic system is maintained even at large volumes. Our state-of-the-art facilities are designed to handle sensitive asymmetric reactions with precision, supported by rigorous QC labs that enforce stringent purity specifications for every batch released. We understand that consistency is key in the pharmaceutical supply chain, and our robust quality management systems guarantee that every gram of R-type chiral sulfoxide we deliver meets the highest industry standards.
We invite pharmaceutical partners and research institutions to collaborate with us to leverage this advanced synthesis capability. Whether you require custom synthesis for clinical trials or bulk supply for commercial manufacturing, our technical procurement team is ready to assist. Contact us today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to see how our optimized process can enhance your supply chain efficiency and reduce your overall cost of goods sold. Let us be your trusted partner in delivering high-quality chiral solutions for the next generation of anticoagulant medicines.
