Advanced One-Pot Synthesis of Peramivir Key Intermediate for Commercial Scale-Up
Advanced One-Pot Synthesis of Peramivir Key Intermediate for Commercial Scale-Up
The global demand for effective antiviral therapeutics has placed significant scrutiny on the efficiency and scalability of synthetic routes for key pharmaceutical ingredients. In this context, the technological breakthroughs detailed in patent CN102757365B represent a pivotal advancement in the manufacturing of Peramivir, a potent neuraminidase inhibitor. This specific intellectual property discloses a novel method for preparing the critical intermediate (1S,2S,3S,4R,1'S)-3-(1-acetamido-2-ethylbutyl)-4-(Boc)-amino-2-hydroxy-cyclopentane-1-carboxylate. Unlike traditional methodologies that suffer from fragmented operational steps, this innovation utilizes a streamlined one-pot strategy that combines reductive ring opening with immediate acetylation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patent is essential for optimizing supply chains. The structural complexity of Peramivir, as illustrated below, demands precise stereochemical control which this new method addresses effectively.
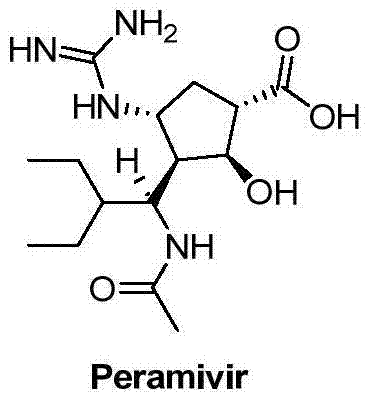
The molecular architecture of Peramivir involves multiple chiral centers and sensitive functional groups that require meticulous handling during synthesis. The ability to construct the cyclopentane core with high fidelity is the cornerstone of producing a high-purity API that meets stringent regulatory standards. By leveraging the insights from this patent, manufacturers can transition from legacy processes to more agile and cost-effective production models. This shift not only enhances the economic viability of the final drug product but also ensures a more resilient supply chain capable of meeting sudden surges in demand during viral outbreaks. The following analysis dissects the technical merits and commercial implications of this superior synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing Peramivir intermediates have been plagued by inefficiencies that hinder large-scale commercialization. Traditional routes often involve a multi-step sequence where the reductive ring opening of the isoxazoline precursor is performed as a discrete unit operation, necessitating the isolation and purification of the resulting amine intermediate before proceeding to acetylation. This separation is not merely a logistical burden; it introduces significant risks of product degradation and yield loss due to the instability of the free amine species. Furthermore, the requirement for extensive work-up procedures between steps increases solvent consumption, waste generation, and overall processing time. For a procurement manager focused on cost reduction in API manufacturing, these redundant operations translate directly into inflated production costs and extended lead times. The cumulative yield of such fragmented processes often falls short of economic targets, making the final drug substance prohibitively expensive to produce at the volumes required for global health initiatives.
The Novel Approach
In stark contrast, the methodology outlined in the patent data introduces a paradigm shift by integrating the reduction and acetylation steps into a seamless one-pot operation. By utilizing intermediate (II) as the starting material, the process employs a combined reducing agent system to effectuate the ring opening, followed immediately by the addition of an acetylating agent without any intermediate isolation. This telescoping of reactions eliminates the need for complicated post-processing of the reaction system after the reduction phase. The direct quenching and acetylating strategy not only simplifies the operational workflow but also protects the sensitive amine functionality in situ, thereby preventing decomposition. As depicted in the reaction scheme below, this approach transforms a cumbersome multi-stage synthesis into a concise and robust protocol. For supply chain heads, this simplification means fewer unit operations, reduced equipment occupancy time, and a drastically simplified material flow that enhances overall plant throughput.
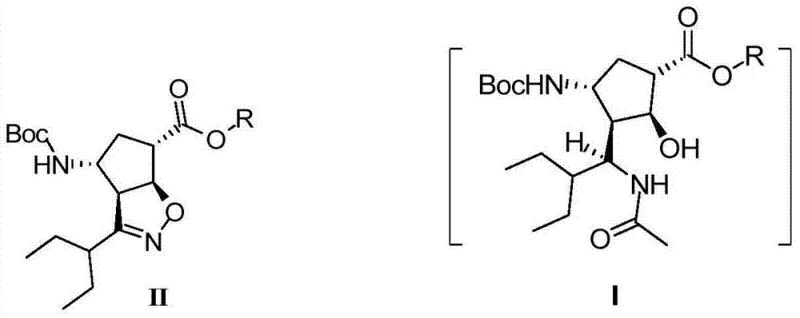
The visual representation of this transformation highlights the elegance of the chemical design, where the structural integrity of the cyclopentane ring is maintained while the necessary functional group manipulations occur in a single vessel. This level of process intensification is critical for modern pharmaceutical manufacturing, where efficiency and sustainability are paramount. The ability to bypass the isolation of unstable intermediates reduces the exposure of the material to potential contaminants and environmental factors that could compromise quality. Consequently, this novel approach offers a compelling value proposition for stakeholders looking to optimize their production portfolios. It stands as a testament to how innovative process chemistry can drive substantial improvements in both technical performance and commercial competitiveness within the fine chemical sector.
Mechanistic Insights into NaBH4/NiCl2 Catalyzed Reductive Ring Opening
The core of this technological advancement lies in the specific choice of the reducing system, which utilizes a combination of metal borohydride and metal chloride, specifically sodium borohydride (NaBH4) and nickel chloride hexahydrate (NiCl2·6H2O). This dual-component system generates an active catalytic species in situ that facilitates the cleavage of the nitrogen-oxygen bond in the isoxazoline ring under remarkably mild conditions. The reaction is conducted in an anhydrous solution, typically a mixture of methanol and tetrahydrofuran, at temperatures ranging from -50°C to room temperature, with a preferred window between -15°C and 0°C. Maintaining this precise thermal control is vital for ensuring the stereoselectivity of the reduction, as it dictates the facial selectivity of the hydride attack on the cyclic substrate. For R&D teams, understanding these parameters is crucial for replicating the high yields reported in the patent, which consistently exceed 80% across multiple batches. The neutral nature of the reaction medium further contributes to the compatibility of the process with other acid- or base-sensitive protecting groups present on the molecule, such as the Boc group.
Beyond the primary reduction event, the mechanism inherently supports superior impurity control, which is a key concern for regulatory compliance and downstream processing. By avoiding the isolation of the free amine, the process minimizes the formation of oxidation by-products and dimerization impurities that often arise when reactive amines are exposed to air or prolonged storage. The immediate subsequent acetylation with acetic anhydride serves as an effective capping step, locking the stereochemistry and stabilizing the molecule for subsequent purification. This integrated approach to impurity management reduces the burden on downstream purification techniques like column chromatography or recrystallization, leading to a cleaner crude profile. For quality assurance professionals, this means a more predictable and consistent impurity spectrum, facilitating easier validation and release testing. The robustness of this mechanistic pathway ensures that the critical quality attributes of the intermediate are preserved throughout the synthesis, aligning perfectly with the rigorous standards expected of a high-purity pharmaceutical intermediate.
How to Synthesize Peramivir Intermediate Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions described in the patent embodiments to ensure reproducibility and safety. The process begins with the dissolution of the isoxazoline starting material in a cooled solvent system, followed by the controlled addition of the nickel catalyst and reducing agent. Monitoring the reaction progress via thin-layer chromatography is essential to determine the exact endpoint before introducing the acetylating agent. Once the reduction is complete, the direct addition of acetic anhydride allows for the immediate conversion of the amine to the acetamide. The detailed standardized synthesis steps, including specific molar ratios, stirring rates, and work-up procedures, are provided in the technical guide below for your process engineering team to review.
- Dissolve the starting isoxazoline intermediate in a mixed solvent system of methanol and THF, then cool the solution to a controlled low temperature range between -15°C and -10°C.
- Introduce the combined reducing agent system comprising sodium borohydride and nickel chloride hexahydrate slowly to maintain reaction stability and monitor progress via TLC until raw material consumption is complete.
- Without isolating the intermediate, directly add acetic anhydride to the reaction mixture for in-situ acetylation, followed by pH adjustment, extraction, and purification via column chromatography or recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this one-pot synthetic route offers profound benefits that extend well beyond the laboratory bench. For procurement managers tasked with optimizing the cost structure of API manufacturing, the elimination of intermediate isolation steps translates directly into significant operational savings. By removing the need for separate filtration, drying, and re-dissolution stages, the process reduces the consumption of solvents and energy, which are major cost drivers in chemical production. Furthermore, the simplified workflow decreases the labor hours required per batch, allowing existing personnel to manage higher production volumes without proportional increases in headcount. These efficiencies compound to create a leaner manufacturing model that is better equipped to withstand market volatility and pricing pressure. The ability to source this intermediate from a supplier utilizing such advanced technology ensures a more competitive cost basis for the final drug product.
- Cost Reduction in Manufacturing: The telescoped nature of the reaction sequence fundamentally alters the economics of production by consolidating multiple chemical transformations into a single vessel. This consolidation eliminates the capital expenditure associated with additional reactors and isolation equipment, while also reducing the footprint required for manufacturing. The high yield achieved through this method, consistently greater than 80%, ensures that raw material utilization is maximized, minimizing waste and the cost of goods sold. Additionally, the use of commodity reagents like sodium borohydride and acetic anhydride avoids the reliance on exotic or prohibitively expensive catalysts, further stabilizing the input cost profile. These factors collectively contribute to a substantial reduction in the overall manufacturing cost, providing a distinct margin advantage.
- Enhanced Supply Chain Reliability: Supply chain continuity is often jeopardized by complex processes that have multiple points of failure; this streamlined approach mitigates those risks significantly. By reducing the number of unit operations, the probability of operational delays or batch failures due to handling errors is drastically lowered. The robustness of the chemistry allows for more flexible scheduling and faster turnaround times between batches, enabling suppliers to respond more敏捷 ly to fluctuations in demand. For supply chain heads, this means a more dependable source of critical materials that can support just-in-time manufacturing strategies. The simplified logistics of moving materials through fewer processing stages also reduces the risk of cross-contamination and mix-ups, ensuring a smoother flow of goods from synthesis to packaging.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden bottlenecks, but this one-pot method is inherently designed for scalability. The reaction conditions are mild and do not require extreme pressures or temperatures, making them easier to manage in large-scale reactors. From an environmental standpoint, the reduction in solvent usage and waste generation aligns with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The decreased volume of waste streams reduces the burden on effluent treatment plants and lowers the costs associated with waste disposal. This sustainability profile not only meets corporate social responsibility goals but also future-proofs the supply chain against regulatory changes that might penalize less efficient manufacturing methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this technology into their existing supply networks. The responses cover aspects of yield optimization, impurity control, and the practicalities of scaling the process for industrial applications.
Q: What are the primary advantages of the one-pot reductive acetylation method over traditional multi-step processes?
A: The one-pot method eliminates the need for complex post-processing and isolation of the unstable reduced intermediate. By directly quenching and acetylating in the same vessel, the process significantly reduces operational time, minimizes material loss during transfer, and simplifies the overall workflow, leading to higher overall yields exceeding 80%.
Q: How does the NaBH4/NiCl2 system impact the stereochemical integrity of the Peramivir intermediate?
A: The combined reducing agent system operates under mild neutral conditions, typically between -50°C and room temperature. This controlled environment ensures high stereoselectivity during the reductive ring-opening of the isoxazoline ring, preserving the critical chiral centers required for the biological activity of the final antiviral agent.
Q: Is this synthetic route suitable for large-scale commercial production of antiviral intermediates?
A: Yes, the process utilizes common reagents like sodium borohydride and acetic anhydride, which are readily available on an industrial scale. The elimination of sensitive isolation steps and the robustness of the one-pot protocol make it highly amenable to scale-up from kilogram to multi-ton production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Peramivir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent concept to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to market launch. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, guaranteeing that every batch of Peramivir intermediate meets the highest international standards. We understand the critical nature of antiviral supply chains and are committed to delivering consistent quality and reliability. Our team of experts is ready to collaborate with you to optimize this specific one-pot route for your unique production requirements, leveraging our infrastructure to maximize efficiency and output.
We invite you to engage with our technical procurement team to discuss how we can tailor this advanced synthesis to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this streamlined process. We encourage you to contact us to obtain specific COA data and route feasibility assessments that will demonstrate our capability to serve as your long-term strategic partner. Let us help you secure a stable and cost-effective supply of high-quality pharmaceutical intermediates for your global operations.
