Advanced Synthesis of Cefozopran Side-Chain Intermediate for Commercial Scale-Up
The pharmaceutical landscape for fourth-generation cephalosporins continues to evolve, driven by the critical need for effective antibiotics against resistant bacterial strains. Patent CN102093266B introduces a transformative synthetic methodology for producing O-tosyl-2-carbamoyl-2-methoxyl-imido-acetamido-oxime, a pivotal intermediate in the manufacture of Cefozopran. This specific chemical entity serves as the essential side-chain acid precursor, dictating the efficacy and stability of the final antibiotic drug. The disclosed technology represents a significant departure from legacy processes by optimizing reaction pathways to enhance both economic viability and operational safety. By leveraging a cascade of nitrosation and esterification reactions starting from readily available cyanoacetamide, the patent outlines a robust framework for high-yield production. For R&D directors and procurement specialists alike, understanding this pathway is crucial for securing a stable supply of high-purity pharmaceutical intermediates. The innovation lies not just in the chemistry, but in the strategic simplification of the manufacturing workflow, addressing long-standing bottlenecks in cephalosporin side-chain synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefozopran side-chain intermediates has been plagued by reliance on expensive and difficult-to-source starting materials such as 3-amino isoxazole or malononitrile. These conventional routes often involve multi-step sequences with rigorous purification requirements between each stage, leading to substantial material loss and inflated production costs. The use of malononitrile, in particular, introduces safety concerns due to its toxicity and handling requirements, while 3-amino isoxazole suffers from supply chain volatility and high market pricing. Furthermore, traditional methods frequently necessitate harsh reaction conditions, including extreme temperatures or the use of hazardous solvents, which complicate waste management and environmental compliance. The cumulative effect of these inefficiencies is a final product with a prohibitive cost structure, limiting the accessibility of the resulting antibiotic in competitive global markets. Additionally, the need for multiple isolation and drying steps increases the overall lead time, making it difficult for manufacturers to respond agilely to surges in demand for critical care medicines.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes cyanoacetamide as the foundational building block, a commodity chemical that offers superior availability and cost stability. This strategy fundamentally reengineers the synthesis by employing a telescoped "one-pot" technique that combines nitrosation and esterification into a seamless operation. By eliminating the need to isolate the unstable nitroso-intermediate, the process minimizes exposure to air and moisture, thereby reducing degradation and improving overall yield. The subsequent conversion involves a mild hydrolysis followed by direct tosylation, maintaining the momentum of the synthesis without unnecessary interruptions. This streamlined workflow not only accelerates the production timeline but also significantly reduces the volume of organic solvents required, aligning with modern green chemistry principles. The result is a highly efficient manufacturing protocol that delivers the target oxime intermediate with exceptional purity, ready for downstream coupling reactions without extensive additional purification.
Mechanistic Insights into Nitrosation and Esterification Cascade
The core of this synthetic breakthrough lies in the precise control of the nitrosation mechanism, where the active methylene group of cyanoacetamide undergoes electrophilic substitution. Under acidic conditions facilitated by sulfuric acid, sodium nitrite generates the reactive nitrosonium ion in situ, which attacks the enol form of the amide. This step is critical, as the temperature must be strictly maintained between 40°C and 50°C to prevent the formation of dinitroso byproducts or the hydrolysis of the nitrile group. Following nitrosation, the reaction mixture is carefully neutralized to a weakly alkaline pH using sodium carbonate, creating the optimal environment for the subsequent nucleophilic attack by dimethyl sulfate. This esterification step converts the unstable oxime hydroxyl into a stable methoxyimino ether, locking in the desired Z-configuration which is essential for the biological activity of the final cephalosporin. The ability to perform these transformations sequentially in the same vessel demonstrates a sophisticated understanding of reaction kinetics and thermodynamics.
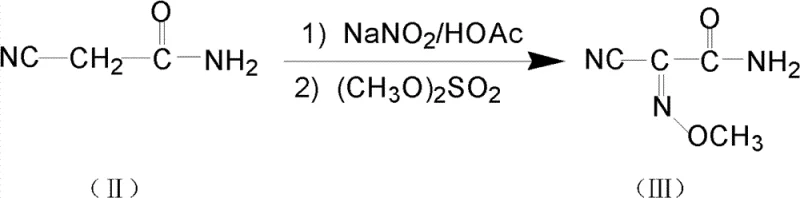
Following the formation of the methoxyimino intermediate, the synthesis proceeds through a chemoselective hydrolysis of the nitrile group to an amidoxime. This transformation is mediated by hydroxylamine hydrochloride in the presence of triethylamine, which acts as a base to scavenge the generated acid and drive the equilibrium forward. The reaction is conducted at mild temperatures, typically between 15°C and 30°C, ensuring that the sensitive methoxyimino linkage remains intact while the nitrile is converted. The resulting amidoxime is then immediately subjected to tosylation using p-toluenesulfonyl chloride. This final step protects the oxime oxygen, preventing unwanted cyclization or degradation during storage and subsequent coupling with the cephalosporin nucleus. The entire sequence is designed to minimize the generation of regioisomers and geometric isomers, which are common impurities in oxime chemistry that can be difficult to separate.
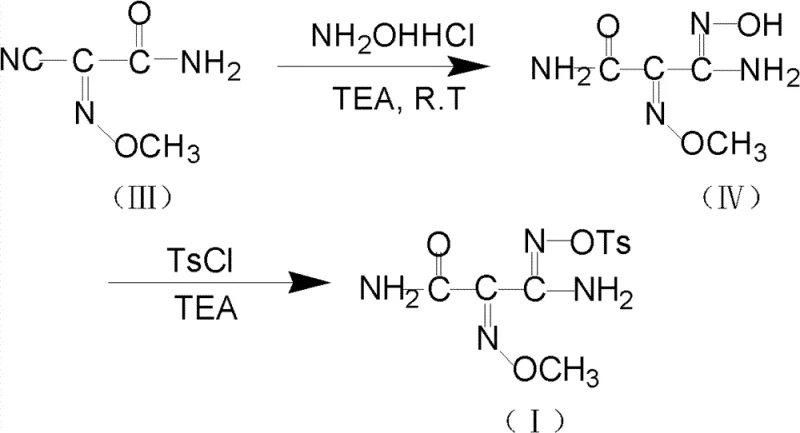
How to Synthesize O-tosyl-2-carbamoyl-2-methoxyl-imido-acetamido-oxime Efficiently
To implement this synthesis effectively in a pilot or production setting, operators must adhere to strict procedural controls regarding reagent addition rates and thermal management. The process begins with the suspension of cyanoacetamide in water, followed by the controlled addition of sodium nitrite and acid to initiate nitrosation. Once the nitrosation is complete, verified by TLC or HPLC, the pH is adjusted, and dimethyl sulfate is added dropwise to manage the exotherm. After isolating the intermediate 2-cyano-2-methoxyl-imido-acetamide, it is redissolved in methanol for the hydroxylamine treatment. The detailed standardized synthesis steps, including specific molar ratios and stirring times, are outlined below to ensure reproducibility and safety.
- Perform nitrosation of cyanoacetamide with sodium nitrite and acid at 40-50°C, followed by direct esterification with dimethyl sulfate under weakly alkaline conditions to form 2-cyano-2-methoxyl-imido-acetamide.
- Hydrolyze the nitrile group of the intermediate using hydroxylamine hydrochloride and triethylamine in methanol at 15-30°C to generate the amidoxime derivative.
- React the crude amidoxime directly with p-toluenesulfonyl chloride (TsCl) and triethylamine at 0-10°C to yield the final O-tosylated product without intermediate isolation.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift to cyanoacetamide as a starting material offers a profound strategic advantage by decoupling production costs from the volatile pricing of specialized heterocycles. This raw material is produced on a massive global scale for various industrial applications, ensuring a consistent supply stream that is less susceptible to the shortages often seen with niche pharmaceutical precursors. The simplification of the process flow directly translates to reduced capital expenditure on equipment, as fewer reactors and separation units are required to achieve the same output volume. Moreover, the reduction in unit operations lowers the labor intensity of the manufacturing campaign, allowing facilities to allocate human resources more efficiently across other high-value projects. The overall effect is a significant compression of the cost of goods sold (COGS), providing a competitive buffer against market fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like 3-amino isoxazole removes a major cost driver from the bill of materials. Additionally, the telescoped nature of the reactions means that solvents and reagents are utilized more efficiently, with less waste generated per kilogram of product. By avoiding the isolation of unstable intermediates, the process saves on filtration, drying, and packaging costs associated with those transient species. The use of common reagents such as sodium nitrite and dimethyl sulfate further ensures that procurement teams can leverage existing vendor relationships and bulk purchasing power to negotiate favorable terms.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain resilience by reducing the number of potential failure points. Traditional multi-step syntheses are vulnerable to delays at any single isolation stage; however, this continuous flow-like approach minimizes such risks. The mild reaction conditions reduce the likelihood of batch failures due to thermal runaways or equipment corrosion, ensuring higher on-time delivery rates for customers. Furthermore, the scalability of the process allows manufacturers to ramp up production quickly in response to public health emergencies or seasonal demand spikes for antibiotics without requiring extensive process re-validation.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing aqueous and alcoholic solvent systems that are easier to recover and recycle compared to chlorinated or aromatic solvents used in older methods. The reduction in hazardous waste generation simplifies effluent treatment protocols, lowering the environmental compliance burden and associated disposal fees. This alignment with green chemistry standards not only improves the corporate sustainability profile but also future-proofs the manufacturing site against tightening environmental regulations. The ability to run larger batch sizes without compromising safety or quality makes this an ideal candidate for multi-ton annual production campaigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new methodology for their own supply chains.
Q: What are the primary cost advantages of this synthesis route compared to conventional methods?
A: This route utilizes cyanoacetamide as a starting material, which is significantly more accessible and cost-effective than the traditional 3-amino isoxazole or malononitrile. Furthermore, the telescoped 'one-pot' methodology eliminates multiple isolation steps, drastically reducing solvent consumption, labor hours, and overall processing time.
Q: How does the process ensure high purity and minimize impurities during the nitrosation step?
A: High purity is achieved through precise temperature control during the nitrosation phase (maintained between 40-50°C) and the immediate neutralization of residual acid prior to esterification. This prevents side reactions and decomposition, ensuring the intermediate 2-cyano-2-methoxyl-imido-acetamide is formed with minimal byproduct generation before proceeding to the next stage.
Q: Is this synthetic method suitable for large-scale industrial manufacturing?
A: Yes, the method is explicitly designed for industrial suitability. It avoids harsh reaction conditions and expensive catalysts, relying instead on common reagents like sodium nitrite and dimethyl sulfate. The mild temperature requirements (0-50°C range) and the ability to telescope reactions make it highly scalable for multi-ton production campaigns.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-tosyl-2-carbamoyl-2-methoxyl-imido-acetamido-oxime Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global fight against bacterial infections. Our team of expert chemists has extensively analyzed the pathway described in CN102093266B and possesses the technical capability to execute this synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of O-tosyl-2-carbamoyl-2-methoxyl-imido-acetamido-oxime meets the highest pharmacopeial standards.
We invite you to collaborate with us to optimize your Cefozopran supply chain. By leveraging our manufacturing expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to deliver a secure, cost-effective, and high-quality supply of this vital pharmaceutical intermediate.
