Advanced Manufacturing of Arbidol Mesylate: A Scalable Route for Global API Production
Advanced Manufacturing of Arbidol Mesylate: A Scalable Route for Global API Production
The pharmaceutical industry is constantly seeking more efficient and safer pathways for producing critical antiviral agents, and the synthesis of Arbidol Mesylate represents a significant leap forward in this domain. As detailed in patent CN102260205A, this innovative methodology addresses the longstanding solubility and toxicity issues associated with traditional Arbidol Hydrochloride. By shifting to a mesylate salt form, manufacturers can achieve a compound with vastly superior water solubility—dissolving 1g in merely 27mL of water compared to the near-insolubility of the HCl salt. This technical breakthrough not only enhances the therapeutic efficacy of the final drug but also streamlines the formulation process for oral solid dosages. For R&D directors and procurement specialists, understanding this specific chemical architecture is vital for securing a reliable supply chain of high-quality antiviral intermediates.
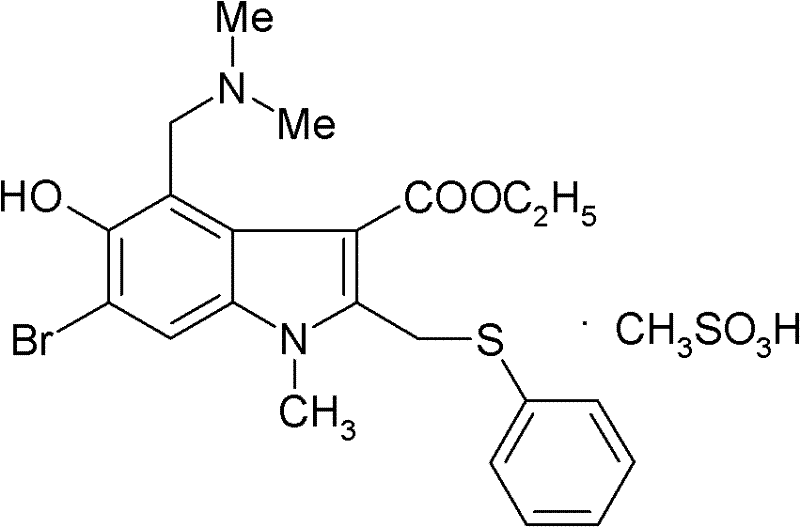
The structural integrity of Arbidol Mesylate, as depicted above, relies on a precise arrangement of functional groups including the bromine atom, the phenylthiomethyl moiety, and the dimethylaminomethyl side chain. Achieving this structure efficiently requires a departure from legacy methods that often rely on hazardous reagents and cumbersome protection strategies. The patent outlines a robust sequence starting from common bulk chemicals like ethyl acetoacetate, transforming them through a series of well-controlled reactions including amination, Nenitzescu condensation, and bromination. This approach ensures that the final product meets stringent purity specifications required for clinical applications, making it an ideal candidate for large-scale commercial production by experienced chemical manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Arbidol base has been plagued by operational hazards and environmental inefficiencies that hinder scalable manufacturing. Traditional routes typically necessitate the use of methylamine gas for the initial amination step, which presents significant safety challenges regarding storage, handling, and reaction control due to its gaseous state and high reactivity. Furthermore, legacy processes heavily rely on toxic chlorinated solvents such as 1,2-dichloroethane and tetrachloroethane, which are increasingly restricted by global environmental regulations due to their carcinogenic potential and persistence in the environment. Another major bottleneck in conventional synthesis is the requirement for protecting the active hydroxyl group on the indole ring prior to bromination, followed by a subsequent deprotection step. This adds unnecessary complexity, extends the production cycle, increases raw material consumption, and generates additional waste streams that complicate downstream processing and increase overall operational costs.
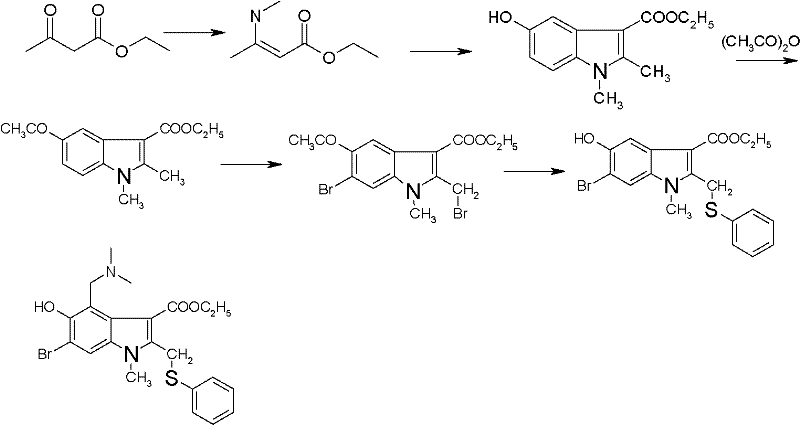
The Novel Approach
In stark contrast to these outdated methodologies, the novel synthesis route described in the patent introduces a streamlined and environmentally conscious strategy that eliminates these critical bottlenecks. By utilizing a 40wt% aqueous methylamine solution instead of gaseous methylamine, the process becomes inherently safer and easier to control, allowing for precise stoichiometric addition without the need for specialized gas handling infrastructure. The replacement of highly toxic solvents with acetone and chloroform significantly reduces the environmental footprint of the manufacturing process, aligning with modern green chemistry principles and facilitating easier regulatory approval for industrial facilities. Most notably, this innovative route bypasses the need for hydroxyl protection entirely, allowing for direct bromination of the indole core. This strategic simplification reduces the total number of reaction steps, thereby shortening the lead time for production and substantially lowering the cost of goods sold (COGS) while maintaining high yields and product purity.
Mechanistic Insights into Nenitzescu Condensation and Direct Bromination
The core of this synthetic success lies in the meticulous optimization of the Nenitzescu reaction and the subsequent bromination strategy, which together construct the complex indole scaffold with high fidelity. The process begins with the formation of an enamine intermediate from ethyl acetoacetate and aqueous methylamine, which then undergoes a cycloaddition with para-benzoquinone in an acetone medium. This solvent choice is critical; acetone provides the necessary polarity to dissolve the quinone while facilitating the precipitation of the indole product upon concentration and cooling, simplifying isolation. Following the formation of the 5-hydroxyindole core, the direct bromination step is executed in chloroform under reflux conditions. Unlike traditional methods that might require mild conditions to preserve a protecting group, this robust protocol allows for vigorous reaction conditions that ensure complete conversion to the 6-bromo derivative, setting the stage for the subsequent nucleophilic substitution with thiophenol.
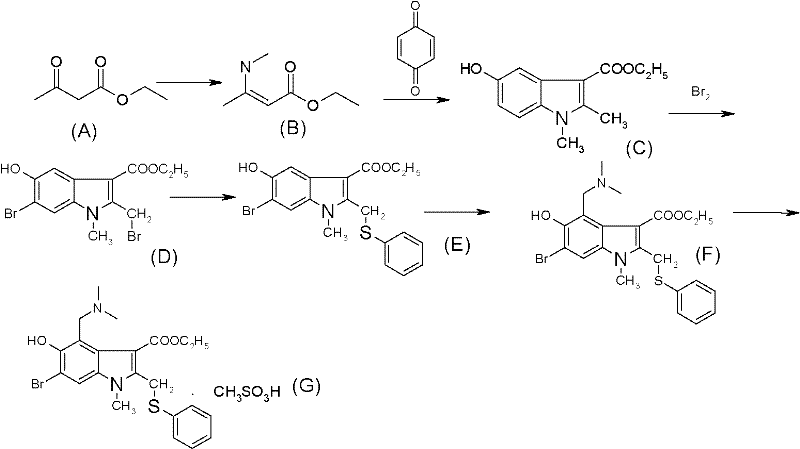
Following the installation of the bromine and phenylthio groups, the synthesis proceeds through a Mannich reaction to introduce the dimethylaminomethyl functionality at the 4-position of the indole ring. This step is carefully controlled at temperatures between 60-70°C in glacial acetic acid to prevent over-reaction or degradation of the sensitive indole nucleus. The final conversion to the mesylate salt is achieved by reacting the free base with methanesulfonic acid in acetone, a process that leverages the low solubility of the salt in this solvent to drive crystallization. This mechanistic pathway ensures that impurities are effectively excluded during the crystallization phase, resulting in a final product with purity levels exceeding 98%, as verified by HPLC analysis in the patent examples. Such rigorous control over reaction parameters is essential for producing pharmaceutical intermediates that meet the exacting standards of global regulatory bodies.
How to Synthesize Arbidol Mesylate Efficiently
Implementing this synthesis route requires a disciplined approach to reaction conditions and workup procedures to maximize yield and safety. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates and reducing solvent usage. For instance, the amination step yields an organic layer that can be directly utilized or minimally processed before entering the condensation phase. The patent emphasizes the importance of temperature control during the exothermic amination and the precise stoichiometry during the salt formation to ensure consistent crystal morphology and particle size distribution. These factors are critical for downstream formulation scientists who rely on uniform physical properties for tablet compression or capsule filling. Detailed standardized operating procedures for each unit operation are essential for technology transfer from the laboratory to the pilot plant and eventually to full commercial scale.
- Perform amination of ethyl acetoacetate using 40wt% aqueous methylamine solution at 20-50°C to form 3-methylamino-butenoic acid ethyl ester.
- Conduct a Nenitzescu reaction with para-benzoquinone in acetone solvent to generate the indole core structure (1,2-dimethyl-5-oxyindole-3-carboxyl ethyl ester).
- Execute direct bromination in chloroform followed by thiophenol substitution and a Mannich reaction to install the dimethylaminomethyl group, finishing with methanesulfonic acid salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route translates into tangible strategic advantages that go beyond mere chemical elegance. The elimination of hazardous gases and restricted solvents mitigates the risk of supply disruptions caused by tightening environmental regulations or transportation restrictions on dangerous goods. By relying on commodity chemicals like acetone and aqueous amines, manufacturers can secure a more stable and cost-effective raw material supply base, insulating the production schedule from the volatility of specialty chemical markets. Furthermore, the reduction in synthetic steps directly correlates to a shorter manufacturing cycle time, enabling faster response to market demand surges, such as those seen during influenza outbreaks. This agility is a crucial component of a resilient supply chain, ensuring that critical antiviral medications remain available when they are needed most.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the synthetic sequence and the substitution of expensive or regulated reagents. By removing the protection and deprotection steps, the process saves on the cost of protecting group reagents, the solvents required for those steps, and the labor hours associated with additional unit operations. Additionally, the switch to aqueous methylamine eliminates the need for specialized gas scrubbing systems and high-pressure reactors, reducing capital expenditure (CAPEX) and maintenance costs. The use of acetone, a low-cost and widely available solvent, further drives down variable costs compared to the specialized chlorinated solvents used in legacy routes. These cumulative efficiencies result in a significantly lower cost of production, allowing for more competitive pricing in the global API market without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of readily available starting materials that are produced on a massive industrial scale globally. Ethyl acetoacetate, para-benzoquinone, and methanesulfonic acid are commodity chemicals with multiple qualified suppliers, reducing the risk of single-source dependency. The avoidance of methylamine gas removes a significant logistical hurdle, as transporting compressed gases requires specialized containers and regulatory compliance that can delay shipments. By utilizing liquid reagents and standard solvents, the logistics of raw material inbound become simpler and more flexible. This reliability ensures that production schedules can be maintained consistently, preventing stockouts and ensuring a steady flow of intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is designed for scalability with minimal ecological impact. The replacement of carcinogenic solvents like 1,2-dichloroethane with acetone and chloroform simplifies waste treatment protocols and reduces the burden on effluent treatment plants. The process generates less hazardous waste per kilogram of product, aligning with corporate sustainability goals and reducing disposal costs. Furthermore, the simplified workflow with fewer isolation steps reduces the potential for operator exposure to intermediates, enhancing workplace safety. This environmental compatibility makes the process easier to permit and operate in various jurisdictions, facilitating global manufacturing capabilities and ensuring long-term operational viability in a regulatory landscape that is increasingly focused on green chemistry metrics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of Arbidol Mesylate. These insights are derived directly from the patented methodology and are intended to clarify the advantages of this specific synthetic route for stakeholders evaluating potential partnerships. Understanding these nuances is key to making informed decisions about sourcing strategies and technology adoption.
Q: Why is Arbidol Mesylate preferred over Arbidol Hydrochloride?
A: Arbidol Mesylate exhibits significantly improved water solubility (1g dissolves in 27mL water) compared to the hydrochloride salt (1g does not dissolve in 10000mL water), leading to better bioavailability and faster absorption in the body.
Q: How does this synthesis route improve environmental safety?
A: The process eliminates the use of highly toxic solvents like 1,2-dichloroethane and tetrachloroethane, replacing them with acetone and chloroform. Additionally, it avoids the handling of hazardous methylamine gas by using aqueous solutions.
Q: What are the key efficiency gains in this manufacturing method?
A: The route removes the need for protecting and deprotecting the hydroxyl group on the indole ring. This reduction in synthetic steps shortens the production cycle, lowers material costs, and simplifies the purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arbidol Mesylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of complex pharmaceutical intermediates like Arbidol Mesylate. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the clinical trial phase or full-scale commercialization. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this synthesis, guaranteeing a consistent and high-quality supply that adheres to international GMP standards.
We invite you to collaborate with us to optimize your supply chain for antiviral therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume needs, demonstrating how our efficient manufacturing processes can reduce your overall procurement costs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and cost-effective source of Arbidol Mesylate, ensuring your projects move forward without interruption.
