Revolutionizing Bromfenac Intermediate Production: A Deep Dive into Nickel-Catalyzed Synthesis
Revolutionizing Bromfenac Intermediate Production: A Deep Dive into Nickel-Catalyzed Synthesis
The pharmaceutical industry constantly seeks more efficient pathways to produce critical active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in Chinese Patent CN109608345B, which details a novel catalytic method for synthesizing 2-amino-4'-bromo-benzophenone. This compound serves as a pivotal intermediate in the manufacturing of Bromfenac Sodium, a potent nonsteroidal anti-inflammatory drug (NSAID) widely used in ophthalmic solutions for treating postoperative inflammation and conjunctivitis. The patent addresses long-standing synthetic challenges, offering a route that bypasses the harsh conditions and low yields associated with traditional methodologies. By leveraging a specialized nickel-catalyzed system promoted by Bronsted acids, this technology enables the direct coupling of o-aminobenzonitrile with p-bromobenzoic acid derivatives. For global procurement teams and R&D directors, this represents a shift towards more sustainable and economically viable manufacturing processes for high-value ophthalmic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
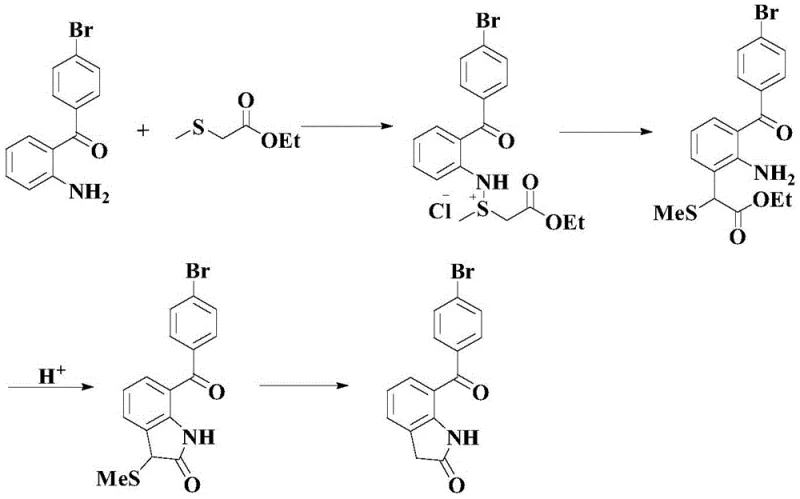
Historically, the synthesis of 2-amino-4'-bromo-benzophenone has been fraught with inefficiencies and safety hazards. Traditional routes often rely on multi-step sequences starting from phthalic anhydride and halogenated benzenes. As illustrated in prior art, these methods typically involve a Friedel-Crafts acylation followed by acyl chlorination, amidation, and finally a Hoffman degradation. These processes are not only labor-intensive but also require the use of stoichiometric amounts of aluminum trichloride and liquid bromine, reagents that pose severe environmental and handling risks. Furthermore, alternative pathways attempting to construct the indolone core directly often suffer from abysmal overall yields, frequently falling below 30 percent due to the accumulation of losses across four distinct reaction steps. The reliance on such cumbersome chemistry creates bottlenecks in the supply chain, driving up costs and complicating waste management protocols for manufacturers.
The Novel Approach
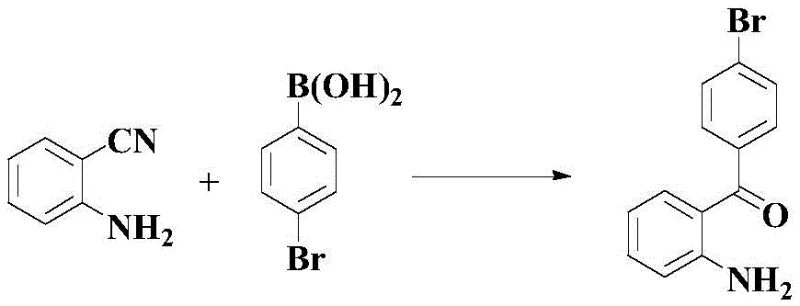
In stark contrast to these legacy methods, the technology disclosed in Patent CN109608345B introduces a streamlined, direct catalytic approach. The core innovation lies in the ability to react o-aminobenzonitrile directly with p-bromobenzoic acid (or its boronic acid derivative) in a single pot. This eliminates the need for pre-functionalization steps and avoids the generation of hazardous byproducts associated with Friedel-Crafts chemistry. The reaction proceeds efficiently in a mixed solvent system of 1,4-dioxane and water, utilizing a nickel catalyst and a Bronsted acid promoter. This methodology not only simplifies the operational workflow but also significantly enhances the atom economy of the process. By collapsing multiple synthetic steps into a single catalytic transformation, the new approach drastically reduces the time and resources required to produce this critical pharmaceutical building block.
Mechanistic Insights into Nickel-Catalyzed Addition of Aryl Acids to Nitriles
The success of this synthesis hinges on overcoming specific mechanistic hurdles that have plagued previous attempts at similar transformations. Typically, the presence of a free amino group on the aromatic ring poses a significant challenge in transition metal catalysis. The amino group acts as a strong coordinating ligand, which can bind tightly to the nickel center, effectively poisoning the catalyst and preventing the desired oxidative addition or transmetallation steps. Additionally, the electron-donating nature of the amino group increases the electron density of the benzene ring, making the cyano carbon less electrophilic and thus less susceptible to nucleophilic attack by organometallic species. Previous literature, such as Documents 7 and 8 cited in the patent background, failed to produce the target molecule when using o-aminobenzonitrile, yielding no detectable product due to these deactivation pathways.
The patented method resolves these issues through the strategic use of a Bronsted acid, specifically trifluoroacetic acid (TFA). The acid plays a dual role: firstly, it likely protonates the amino group, thereby masking its coordinating ability and preventing catalyst deactivation. Secondly, the acidic environment may activate the nitrile group towards nucleophilic attack or facilitate the hydrolysis of intermediate imine species. The choice of ligand on the nickel catalyst is also critical; the patent identifies Ni(dppe)Cl2 as the optimal catalyst, suggesting that the specific steric and electronic properties of the dppe ligand create a coordination sphere that remains active even in the presence of the challenging substrate. This precise tuning of the catalytic system allows for the successful formation of the carbon-carbon bond between the aryl acid and the nitrile, a transformation that was previously deemed unfeasible for this specific substrate class.
How to Synthesize 2-Amino-4'-Bromo-Benzophenone Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize yield and purity. The process involves mixing the starting materials in a specific solvent ratio and maintaining a controlled temperature profile. The use of a mixed solvent system containing water is particularly notable, as it facilitates the solubility of the acid promoter while maintaining the organic phase for the catalytic cycle. Following the reaction, a standard aqueous workup involving ethyl acetate extraction and washing with saturated sodium bicarbonate effectively removes acidic residues and inorganic salts. The final purification is achieved through flash column chromatography, yielding the product as a pale yellow solid with high purity suitable for downstream pharmaceutical applications.
- Combine o-aminobenzonitrile, p-bromobenzoic acid (or boronic acid), Ni(dppe)Cl2 catalyst, and trifluoroacetic acid in a 1,4-dioxane/water mixed solvent.
- Heat the reaction mixture to a temperature between 90°C and 120°C and maintain stirring for 12 to 24 hours to ensure complete conversion.
- Perform standard workup involving evaporation, ethyl acetate extraction, washing with bicarbonate/brine, drying, and flash column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology offers profound strategic advantages beyond mere technical novelty. The shift from multi-step, hazard-heavy syntheses to a direct catalytic coupling fundamentally alters the cost structure and risk profile of producing Bromfenac intermediates. By eliminating the need for corrosive reagents like aluminum trichloride and liquid bromine, manufacturers can significantly reduce their expenditure on specialized containment equipment, personal protective equipment, and hazardous waste disposal services. Furthermore, the simplification of the process flow reduces the manpower and reactor time required per batch, leading to substantial operational cost savings. This efficiency gain allows suppliers to offer more competitive pricing while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of multiple synthetic steps directly translates to lower variable costs. Traditional routes involve isolation and purification of intermediates at each stage, consuming significant amounts of solvents and energy. By consolidating the synthesis into a single catalytic step, the new method minimizes solvent usage and energy consumption for heating and cooling cycles. Additionally, the avoidance of expensive and hazardous reagents reduces the raw material bill of materials. The high yield reported in the patent examples indicates that less starting material is wasted, further enhancing the overall economic efficiency of the production process. These cumulative savings allow for a more robust cost structure that can withstand fluctuations in raw material markets.
- Enhanced Supply Chain Reliability: The starting materials for this new method, o-aminobenzonitrile and p-bromobenzoic acid (or boronic acid), are commodity chemicals that are readily available from multiple global suppliers. This contrasts sharply with older methods that might require custom-synthesized precursors or reagents with limited supply chains, such as specific acyl chlorides or sulfur-containing intermediates. By relying on widely available feedstocks, manufacturers can mitigate the risk of supply disruptions caused by vendor shortages or logistical bottlenecks. The robustness of the reaction conditions also implies a lower rate of batch failures, ensuring a consistent and reliable output of intermediates to meet the demanding schedules of API production.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process represents a significant improvement. The removal of heavy metal waste (from stoichiometric aluminum salts) and halogenated waste streams simplifies the effluent treatment process. This makes the technology highly scalable, as regulatory hurdles for expanding production capacity are lower when the process generates less hazardous waste. The use of a nickel catalyst, while requiring recovery, is generally more manageable than the disposal of large quantities of aluminum sludge. Consequently, facilities can scale up production to meet commercial demand without incurring prohibitive costs for environmental remediation or facing strict regulatory pushback, ensuring long-term business continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nickel-catalyzed synthesis. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for quality assurance teams establishing control strategies. The answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, ensuring accuracy and relevance for industrial application.
Q: Why do conventional nickel-catalyzed methods fail for synthesizing 2-amino-4'-bromo-benzophenone?
A: Conventional methods often fail due to three main issues: side reactions caused by unprotected free amino groups coupling with aryl halides, catalyst deactivation via coordination between the amino group and the transition metal, and electronic effects where the electron-rich amino group hinders nucleophilic attack on the cyano carbon.
Q: What represents the key innovation in Patent CN109608345B compared to prior art?
A: The key innovation is the specific combination of a nickel catalyst (preferably Ni(dppe)Cl2) with a strong Bronsted acid (preferably trifluoroacetic acid) in a mixed solvent system. This unique system overcomes the deactivation issues and allows the direct reaction of o-aminobenzonitrile with p-bromobenzoic acid derivatives, which was previously impossible.
Q: How does this new method improve supply chain stability for Bromfenac production?
A: This method utilizes readily available starting materials like o-aminobenzonitrile and p-bromobenzoic acid, eliminating the need for hazardous reagents like liquid bromine and aluminum trichloride used in traditional Friedel-Crafts routes. This simplifies procurement, reduces environmental compliance burdens, and ensures a more consistent supply of high-purity intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4'-Bromo-Benzophenone Supplier
The technological advancements described in Patent CN109608345B highlight the evolving landscape of pharmaceutical intermediate synthesis, where efficiency and sustainability are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex catalytic reactions, including the nickel-mediated couplings described herein, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-amino-4'-bromo-benzophenone meets the highest quality standards required for ophthalmic drug manufacturing.
We invite potential partners to engage with our technical team to explore how this innovative synthesis route can optimize your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and high-quality supply of this critical intermediate, enabling you to focus on delivering life-saving therapies to patients worldwide.
