Scalable Synthesis of Chiral Binaphthalene Dicarboxylic Acid for Advanced Pharmaceutical Applications
Scalable Synthesis of Chiral Binaphthalene Dicarboxylic Acid for Advanced Pharmaceutical Applications
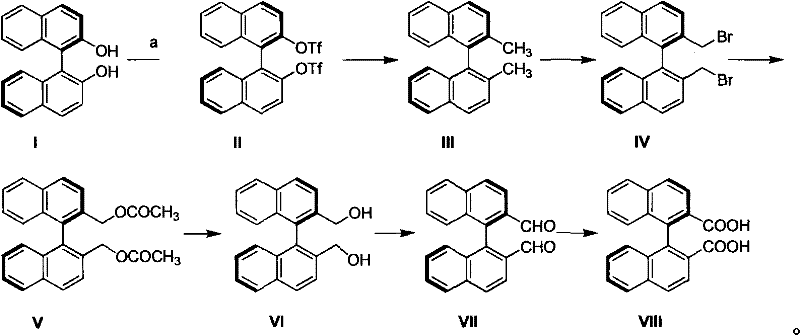
The development of efficient synthetic routes for optically pure binaphthyl derivatives remains a critical challenge in the fine chemical industry, particularly for applications in asymmetric catalysis and advanced pharmaceutical intermediates. Patent CN101781197A introduces a robust and highly efficient preparation method for chiral binaphthalene dicarboxylic acid, addressing the longstanding issues of high cost and low availability associated with this valuable compound. This innovative approach utilizes chiral binaphthol as the starting material, navigating through a series of well-controlled transformations including triflation, methylation, bromination, and sequential oxidations to achieve the target dicarboxylic acid. The significance of this technology lies in its ability to produce the final product with an optical purity exceeding 99% and a remarkable total yield of 74.8%, all while avoiding the need for expensive preparative chromatography. For R&D directors and procurement specialists seeking a reliable chiral binaphthalene dicarboxylic acid supplier, this patent represents a paradigm shift towards more sustainable and cost-effective manufacturing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure binaphthalene dicarboxylic acid has been plagued by significant technical and economic hurdles that hinder widespread commercial adoption. Traditional methods often rely on the chiral resolution of racemic mixtures, a process that is inherently inefficient as it theoretically discards 50% of the synthesized material, leading to substantial waste and inflated costs. Furthermore, the resolving agents required for these processes are frequently expensive, difficult to recover, and in some cases, possess toxicity profiles that complicate regulatory compliance and environmental safety. Alternative synthetic routes involving Ullmann coupling reactions or carbonylation with carbon monoxide gas present their own set of dangers, requiring stringent control of reaction conditions and posing severe safety risks due to the use of highly toxic CO gas. These conventional pathways often result in complex crude mixtures that necessitate labor-intensive purification via preparative HPLC or column chromatography, creating bottlenecks that make commercial scale-up of complex pharmaceutical intermediates nearly impossible for many manufacturers.
The Novel Approach
In stark contrast to these legacy methods, the novel synthetic route disclosed in the patent offers a streamlined, linear progression that maximizes atom economy and operational safety. By initiating the synthesis from readily available chiral binaphthol, the process leverages the inherent chirality of the starting material, thereby eliminating the need for kinetic resolution and ensuring that the stereochemical integrity is maintained throughout the transformation. The strategy employs a sequence of robust organic reactions, beginning with the protection of hydroxyl groups as triflates, followed by a nickel-catalyzed methylation and a radical bromination to functionalize the benzylic positions. This is succeeded by esterification, hydrolysis to the diol, and a controlled two-step oxidation sequence via the aldehyde to the final carboxylic acid. Crucially, every intermediate in this pathway, as well as the final product, can be purified simply through recrystallization, a unit operation that is far more amenable to industrial scaling than chromatographic techniques. This methodological breakthrough not only simplifies the workflow but also drastically reduces the consumption of solvents and silica gel, directly translating to lower production costs and a smaller environmental footprint.
Mechanistic Insights into the Multi-Step Functionalization Strategy
The success of this synthesis relies heavily on the precise control of reactivity at the 2,2'-positions of the binaphthyl backbone, achieved through a clever manipulation of leaving groups and radical chemistry. The initial conversion of the phenolic hydroxyl groups into triflates serves a dual purpose: it protects the sensitive phenol functionality and activates the aromatic ring for the subsequent nickel-catalyzed cross-coupling with methyl Grignard reagents. This methylation step is critical as it installs the methyl groups that will eventually become the carboxylic acid moieties, setting the stage for the benzylic functionalization. Following methylation, the process utilizes N-bromosuccinimide (NBS) under radical conditions to selectively brominate the benzylic methyl groups. This radical bromination is highly selective for the benzylic position due to the stability of the resulting radical intermediate, ensuring that the aromatic core remains intact. The resulting dibromide is then subjected to nucleophilic substitution with acetate, which acts as a masked hydroxyl group, allowing for a gentle hydrolysis later to reveal the primary alcohol without affecting the sensitive biaryl axis.
Impurity control is intrinsically built into this mechanistic design through the physical properties of the intermediates generated at each stage. The patent highlights that each intermediate possesses distinct solubility characteristics that allow for effective purification via recrystallization, effectively filtering out side products and unreacted starting materials before they can propagate through the synthesis. For instance, the oxidation of the diol to the dialdehyde using manganese dioxide is a mild transformation that prevents over-oxidation or degradation of the binaphthyl skeleton, which is a common risk with stronger oxidants. The final oxidation to the dicarboxylic acid utilizes sodium chlorite under buffered conditions (Pinnick oxidation conditions), which is known for its high chemoselectivity towards aldehydes in the presence of other functional groups. By avoiding harsh conditions and utilizing reagents that generate easily removable byproducts, the process ensures that the optical purity remains above 99% throughout the seven steps, preventing racemization which could otherwise occur under acidic or basic extremes.
How to Synthesize Chiral Binaphthalene Dicarboxylic Acid Efficiently
The execution of this synthesis requires careful attention to stoichiometry and temperature control, particularly during the exothermic triflation and the radical bromination steps. The process begins with the dissolution of chiral binaphthol in dichloromethane at low temperatures, followed by the slow addition of triflic anhydride to manage heat generation and ensure complete conversion to the bis-triflate. Subsequent steps involve standard workup procedures such as aqueous washes and drying, but the key to success lies in the recrystallization protocols specified for each intermediate, which serve as the primary purification engine of the entire route. The detailed standardized synthesis steps, including specific solvent volumes, reaction times, and recrystallization solvents for each of the seven transformations, are outlined in the technical guide below to ensure reproducibility and high yield.
- Triflation of chiral binaphthol using Tf2O to form the bis-triflate intermediate.
- Nickel-catalyzed methylation followed by radical bromination to install bromomethyl groups.
- Esterification and hydrolysis to generate the diol, followed by oxidation to aldehyde and finally to the dicarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound advantages in terms of cost structure and supply reliability. The elimination of preparative chromatography is perhaps the most significant economic driver, as column chromatography is notoriously difficult to scale, consumes vast quantities of silica and solvents, and limits batch sizes. By replacing this with recrystallization, the manufacturing process becomes compatible with standard stainless steel reactors and filtration equipment found in any multipurpose chemical plant, significantly reducing capital expenditure requirements. Furthermore, the starting material, chiral binaphthol, is a commodity chemical available from multiple global sources, mitigating the risk of single-source dependency and ensuring a stable supply chain for high-purity pharmaceutical intermediates. The robustness of the reaction conditions, which avoid extreme pressures or toxic gases like carbon monoxide, also lowers the barrier for contract manufacturing organizations (CMOs) to adopt the process, increasing the pool of potential suppliers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high overall yield and the avoidance of expensive purification technologies. By achieving a total yield of 74.8% over seven steps, the process minimizes the loss of valuable chiral material, which is often the most expensive component of the cost of goods sold. Additionally, the use of inexpensive reagents such as potassium acetate and manganese dioxide, combined with the ability to recycle solvents like dichloromethane and ethyl acetate, contributes to a substantially lower variable cost per kilogram. The removal of the need for chiral resolving agents, which are often proprietary and costly, further enhances the cost competitiveness of the final product, making it viable for broader applications in generic drug synthesis and fine chemical production.
- Enhanced Supply Chain Reliability: From a logistics perspective, the simplicity of the raw material profile enhances supply security. Since the synthesis does not rely on exotic catalysts or hazardous gases that require special transportation permits, the sourcing of inputs is straightforward and less prone to regulatory delays. The ability to purify intermediates via recrystallization also means that the quality of the final product is less sensitive to minor fluctuations in raw material quality, as impurities are stripped away at multiple stages. This inherent robustness reduces the likelihood of batch failures and production stoppages, ensuring a consistent and reliable flow of material to downstream customers who depend on just-in-time delivery for their own API manufacturing schedules.
- Scalability and Environmental Compliance: The environmental profile of this synthesis aligns well with modern green chemistry principles, facilitating easier regulatory approval and permitting. The avoidance of heavy metal catalysts in the oxidation steps (using MnO2 and NaClO2 instead of Cr-based oxidants where possible, or managing them effectively) and the elimination of toxic CO gas reduce the burden on waste treatment facilities. The process generates solid waste primarily consisting of inorganic salts and organic byproducts that are easier to handle and dispose of compared to the massive volumes of silica waste generated by chromatographic purification. This cleaner waste stream not only reduces disposal costs but also simplifies the environmental impact assessment, accelerating the timeline for commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of chiral binaphthalene dicarboxylic acid based on the patented methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity standards, scalability, and comparative advantages over legacy methods. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own catalytic systems or drug synthesis pipelines.
Q: What is the optical purity of the chiral binaphthalene dicarboxylic acid produced by this method?
A: The patented process ensures an optical purity greater than 99% through rigorous recrystallization steps at each stage, eliminating the need for preparative chromatography.
Q: Why is this synthesis route preferred over traditional resolution methods?
A: Unlike traditional resolution which wastes 50% of the material and requires expensive resolving agents, this route starts from readily available chiral binaphthol and achieves a high total yield of 74.8%.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids toxic gases like CO and complex purification techniques, relying instead on standard unit operations like reflux and recrystallization, making it highly scalable.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Binaphthalene Dicarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral building blocks play in the development of next-generation pharmaceuticals and advanced materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, leveraging our rigorous QC labs to verify optical purity and chemical identity at every stage of the manufacturing process. Our capability to implement the advanced synthetic routes described in patent CN101781197A allows us to offer this valuable intermediate at a competitive price point without compromising on the quality required for sensitive asymmetric catalysis applications.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogues, we are ready to provide the data-driven insights you need. Contact us today to request samples and discover how our commitment to innovation and quality can accelerate your R&D timelines and optimize your production costs.
