Advanced Chiral Selenium Ligand Synthesis for Scalable Pharmaceutical Intermediate Manufacturing
Advanced Chiral Selenium Ligand Synthesis for Scalable Pharmaceutical Intermediate Manufacturing
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more efficient and robust chiral ligands, as evidenced by the groundbreaking technical disclosures in patent CN115466288B. This patent introduces a novel method for synthesizing selenium and sulfur ligands based on chiral diphosphine frameworks, specifically targeting the QuinoxP and BenzP structural motifs which are critical for high-performance transition metal catalysis. The core innovation lies in a streamlined oxidation process that converts readily available chiral diphosphine precursors into their corresponding selenium or sulfur analogues under remarkably mild conditions. For R&D directors and technical procurement leaders in the pharmaceutical and fine chemical sectors, this represents a pivotal shift away from labor-intensive, multi-step ligand modifications towards a more direct, high-yield synthetic pathway. The ability to generate these specialized ligands with high purity and excellent catalytic activity directly addresses the persistent bottlenecks in the production of optically active compounds, particularly chiral lactone ring products which serve as vital scaffolds in modern drug discovery. By leveraging this technology, manufacturers can secure a more reliable supply of high-purity pharmaceutical intermediates while simultaneously optimizing the underlying cost structures associated with complex catalytic processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral phosphine ligands modified with heavier chalcogens like selenium or sulfur has been plagued by significant operational inefficiencies and chemical constraints that hinder large-scale adoption. Conventional routes often necessitate harsh reaction conditions, including elevated temperatures and the use of highly reactive or toxic selenating agents that pose substantial safety and environmental risks in a commercial plant setting. Furthermore, existing methodologies frequently require intricate protection and deprotection strategies to preserve the stereochemical integrity of the chiral backbone during the functionalization of the phosphorus centers, leading to elongated synthetic sequences and cumulative yield losses. These complexities not only drive up the cost of goods sold but also introduce variability in the impurity profile, which is a critical concern for regulatory compliance in API intermediate manufacturing. The reliance on such cumbersome processes limits the availability of these high-value ligands, creating supply chain vulnerabilities for downstream users who depend on consistent access to specialized catalytic systems for their asymmetric transformations. Consequently, the industry has long sought a more robust and scalable alternative that can deliver these essential tools without compromising on safety or economic viability.
The Novel Approach
The methodology outlined in patent CN115466288B offers a transformative solution by employing a direct oxidation strategy using elemental selenium or sulfur powder at room temperature, effectively bypassing the need for aggressive reagents or thermal stress. This one-pot synthetic approach allows for the direct conversion of chiral QuinoxP or BenzP diphosphine ligands into their selenium or sulfur derivatives with exceptional efficiency, as demonstrated by yields consistently exceeding 86% and often reaching above 90% in optimized solvent systems. The simplicity of mixing the diphosphine precursor with selenium powder in common solvents like dichloromethane under anhydrous conditions drastically reduces the operational burden on technical teams, enabling faster turnaround times for ligand production. Moreover, the mild nature of this reaction preserves the delicate chiral information within the molecular structure, ensuring that the resulting ligands maintain the rigorous stereochemical specifications required for high-fidelity asymmetric catalysis. This technological leap not only simplifies the manufacturing workflow but also enhances the overall sustainability of the process by minimizing waste generation and energy consumption, aligning perfectly with the growing demand for green chemistry solutions in the fine chemical industry.
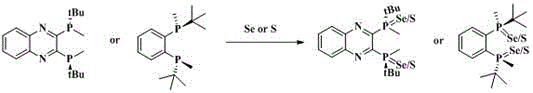
Mechanistic Insights into Chiral Diphosphine Selenium Sulfur Ligand Formation
The chemical elegance of this synthesis lies in the direct nucleophilic attack of the phosphorus centers on the elemental selenium or sulfur, facilitating the formation of robust P=Se or P=S double bonds without disrupting the chiral environment of the ligand backbone. In the case of QuinoxP and BenzP derivatives, the steric bulk provided by the tert-butyl groups adjacent to the phosphorus atoms plays a crucial role in stabilizing the transition state during the oxidation process, thereby preventing racemization and ensuring high optical purity of the final product. The reaction mechanism proceeds smoothly at room temperature, suggesting a low activation energy barrier that is highly favorable for scale-up operations where thermal control can be challenging. Detailed analysis of the reaction kinetics indicates that the molar ratio of the diphosphine ligand to the chalcogen powder is a critical parameter, with optimal results observed when using a slight excess of selenium or sulfur to drive the reaction to completion. This precise control over stoichiometry allows manufacturers to minimize raw material waste while maximizing the conversion efficiency, a key factor in maintaining cost competitiveness in the production of specialty chemicals. The resulting ligands exhibit a rigid structural conformation that is essential for inducing high enantioselectivity in downstream catalytic applications, making them superior candidates for the synthesis of complex chiral molecules.
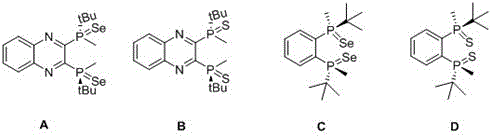
Impurity control is another critical aspect where this novel method demonstrates significant advantages over traditional synthesis routes, particularly regarding the removal of unreacted starting materials and inorganic byproducts. The use of silica gel column chromatography with specific eluent systems, such as ethyl acetate and petroleum ether mixtures, allows for the efficient separation of the target selenium or sulfur ligands from any residual phosphine oxides or elemental chalcogen. The patent data highlights that the purified ligands possess distinct nuclear magnetic resonance signatures that confirm the successful formation of the P=Se or P=S bonds without detectable degradation of the chiral framework. This high level of chemical purity is paramount for R&D directors who require reproducible catalytic performance in their process development campaigns, as even trace impurities can poison sensitive transition metal catalysts or alter the enantiomeric excess of the final API intermediate. By establishing a robust purification protocol that is compatible with standard industrial equipment, this method ensures that the supply of these advanced ligands meets the stringent quality specifications demanded by the global pharmaceutical market, thereby reducing the risk of batch failures and production delays.
How to Synthesize Chiral Diphosphine Selenium Ligand Efficiently
The implementation of this synthesis route in a commercial setting requires careful attention to solvent selection and reaction monitoring to ensure consistent product quality and safety. The patent specifies that anhydrous conditions are essential to prevent the hydrolysis of the phosphine centers, necessitating the use of ultra-dry solvents like dichloromethane or toluene which must be rigorously dried prior to use. Operators should dissolve the chiral diphosphine ligand in the solvent under an inert atmosphere, typically nitrogen or argon, before adding the selenium or sulfur powder to initiate the oxidation process. The reaction mixture is then stirred at room temperature for a period ranging from 10 to 1697 hours, depending on the specific substrate and desired conversion level, with progress monitored via thin-layer chromatography to determine the optimal endpoint. Once the reaction is complete, the mixture is filtered through diatomite to remove insoluble inorganic residues, followed by concentration and purification via silica gel chromatography to isolate the final white solid product. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating this high-yield process.
- Dissolve chiral QuinoxP or BenzP diphosphine ligand in an anhydrous solvent such as dichloromethane under inert atmosphere.
- Add selenium powder or sulfur powder to the reaction system at room temperature with a molar ratio of 1: 2 to 1:5.
- Stir the mixture for 10 to 1697 hours, then purify via silica gel column chromatography to isolate the high-purity ligand.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this ligand synthesis technology offers substantial opportunities for cost reduction and supply chain resilience in the manufacturing of high-value pharmaceutical intermediates. The elimination of complex multi-step synthetic sequences and the use of inexpensive, readily available raw materials like selenium powder significantly lower the direct material costs associated with ligand production. Furthermore, the ability to conduct the reaction at room temperature removes the need for energy-intensive heating or cooling systems, resulting in drastic simplifications of the utility requirements and a corresponding decrease in operational expenditures. For supply chain heads, the robustness of this one-pot method translates into enhanced reliability, as the reduced number of processing steps minimizes the potential for human error and equipment failure that often plague more intricate chemical manufacturing processes. The high yields reported in the patent data suggest that less starting material is required to produce the same amount of final product, effectively increasing the throughput capacity of existing facilities without the need for major capital investment. These factors collectively contribute to a more agile and cost-effective supply chain capable of responding rapidly to market demands for specialized chiral catalysts.
- Cost Reduction in Manufacturing: The streamlined nature of this synthesis route eliminates the need for expensive reagents and complex protection group chemistry, leading to substantial cost savings in the production of chiral ligands. By utilizing elemental selenium or sulfur which are commodity chemicals, the raw material cost base is significantly lower compared to specialized selenating agents used in conventional methods. Additionally, the high conversion efficiency minimizes the loss of valuable chiral starting materials, ensuring that the overall cost per kilogram of the final ligand is optimized for commercial viability. This economic efficiency allows manufacturers to offer more competitive pricing for downstream catalytic services while maintaining healthy profit margins, a critical advantage in the highly competitive fine chemical market.
- Enhanced Supply Chain Reliability: The simplicity of the reaction conditions and the use of common solvents reduce the dependency on specialized supply chains for hazardous or hard-to-source reagents. This decentralization of raw material sourcing mitigates the risk of supply disruptions caused by geopolitical issues or vendor shortages, ensuring a continuous flow of production for critical pharmaceutical intermediates. The robustness of the process also means that it can be easily transferred between different manufacturing sites or scaled up with minimal requalification effort, providing procurement managers with greater flexibility in managing their supplier base. Consequently, companies can secure a more stable supply of high-purity ligands, reducing the likelihood of production delays that could impact the launch timelines of new drug candidates.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of toxic byproducts make this method highly scalable and compliant with increasingly stringent environmental regulations. The ability to run the reaction at room temperature reduces the carbon footprint of the manufacturing process, aligning with corporate sustainability goals and reducing the burden on waste treatment facilities. Furthermore, the straightforward purification process using silica gel chromatography is well-established in industrial settings, allowing for seamless scale-up from laboratory to commercial production without significant process redesign. This environmental and operational compatibility ensures that the technology can be deployed rapidly to meet growing market demand while adhering to global standards for safety and environmental protection.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand synthesis technology, based on the detailed data provided in the patent documentation. These insights are designed to clarify the operational benefits and chemical capabilities of the method for stakeholders involved in process development and procurement. Understanding these nuances is essential for making informed decisions about integrating this technology into existing manufacturing workflows to maximize efficiency and product quality.
Q: What are the primary advantages of this selenium ligand synthesis method over conventional routes?
A: The method described in patent CN115466288B operates at room temperature and utilizes a one-pot oxidation strategy, eliminating the need for complex multi-step protection and deprotection sequences often required in traditional ligand synthesis, thereby significantly reducing operational complexity and energy consumption.
Q: How does this ligand system impact the enantioselectivity of downstream catalytic reactions?
A: The synthesized chiral diphosphine selenium ligands demonstrate excellent catalytic activity in the synthesis of chiral lactone ring products, with specific dual-ligand systems showing synergistic effects that substantially improve both yield and enantiomeric excess values compared to single ligand applications.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the process utilizes easily obtainable raw materials like selenium powder and common solvents, and avoids harsh reaction conditions, making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates with consistent quality and safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced catalytic technologies in driving innovation within the pharmaceutical and fine chemical industries. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the chiral diphosphine selenium ligand synthesis can be translated into robust industrial processes. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, providing our partners with the confidence they need to advance their drug development programs. By leveraging our deep technical expertise and state-of-the-art manufacturing facilities, we can help you overcome the challenges associated with sourcing high-performance chiral ligands and accelerate your path to market.
We invite you to collaborate with us to explore how this innovative synthesis method can optimize your specific catalytic applications and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs, demonstrating the tangible economic benefits of adopting this technology. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to evaluate the potential of these chiral ligands in your own R&D pipelines. Together, we can drive the next generation of asymmetric synthesis solutions forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
