Advanced Copper-Catalyzed Deboronation Strategy for Scalable Aryl Vinyl Trifluoromethyl Sulfide Production
Advanced Copper-Catalyzed Deboronation Strategy for Scalable Aryl Vinyl Trifluoromethyl Sulfide Production
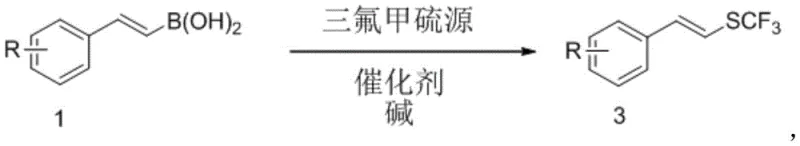
The introduction of the trifluoromethylthio (SCF3) group into organic molecules represents a pivotal strategy in modern medicinal chemistry and agrochemical design, primarily due to its profound impact on lipophilicity and metabolic stability. Patent CN112608260A discloses a groundbreaking method for synthesizing aryl vinyl trifluoromethyl sulfide compounds through a deboronative pathway, utilizing aryl vinyl boronic acids as stable and accessible substrates. This technology addresses the long-standing challenge of introducing the SCF3 motif under mild conditions, moving away from the苛刻 requirements of traditional halide substitution methods. By leveraging a copper-catalyzed system, this invention provides a robust platform for generating high-value fluorine-containing intermediates that are essential for next-generation pharmaceuticals and crop protection agents. The process demonstrates exceptional selectivity, particularly in maintaining the E/Z geometry of the vinyl group, which is critical for the biological activity of the final active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alkenyltrifluoromethylthio compounds has relied heavily on substrates such as haloarylethenes, cinnamic acids, or phenylacetylenes, which present significant logistical and chemical hurdles for large-scale manufacturing. These conventional precursors often require harsh reaction conditions, including extreme temperatures or the use of strong bases that can compromise sensitive functional groups elsewhere in the molecule. Furthermore, the availability of specific halogenated vinyl starting materials can be limited, leading to supply chain bottlenecks and increased raw material costs for procurement teams. The lack of stereocontrol in many traditional methods also results in complex mixtures of E and Z isomers, necessitating expensive and time-consuming purification steps that drastically reduce overall process efficiency. Additionally, many existing protocols rely on expensive transition metal catalysts or unstable trifluoromethylthio reagents that pose safety risks during storage and handling.
The Novel Approach
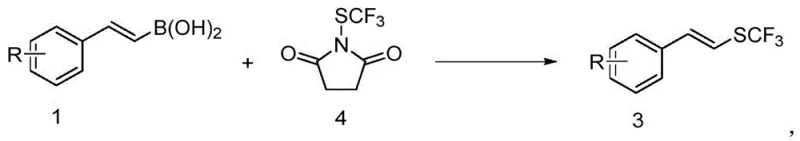
In stark contrast, the novel approach detailed in the patent utilizes aryl vinyl boronic acids, which are renowned for their commercial availability, stability, and ease of handling in industrial settings. This method employs either silver trifluoromethyl mercaptan or N-trifluoromethylthio succinimide as the sulfur source, enabling a direct deboronative trifluoromethylthiolation that proceeds with high efficiency. 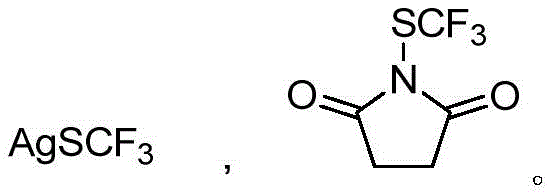 The reaction operates under remarkably mild conditions, typically between 25°C and 100°C, eliminating the need for energy-intensive heating or cryogenic cooling systems. This shift not only simplifies the operational protocol but also significantly broadens the substrate scope, allowing for the incorporation of diverse functional groups such as halogens, esters, and amides without degradation. The result is a streamlined synthetic route that offers superior atom economy and reduces the environmental footprint associated with waste generation from failed reactions or extensive purification.
The reaction operates under remarkably mild conditions, typically between 25°C and 100°C, eliminating the need for energy-intensive heating or cryogenic cooling systems. This shift not only simplifies the operational protocol but also significantly broadens the substrate scope, allowing for the incorporation of diverse functional groups such as halogens, esters, and amides without degradation. The result is a streamlined synthetic route that offers superior atom economy and reduces the environmental footprint associated with waste generation from failed reactions or extensive purification.
Mechanistic Insights into Copper-Catalyzed Deboronative Trifluoromethylthiolation
The mechanistic pathway of this transformation is sophisticated yet elegant, relying on the versatile redox chemistry of copper to facilitate the formation of the C-S bond. When using silver trifluoromethyl mercaptan (AgSCF3) as the reagent, the reaction likely proceeds via a radical mechanism initiated by an oxidant such as potassium persulfate. The oxidant promotes the generation of the trifluoromethylthio radical (.SCF3) from the silver salt, which then interacts with the copper catalyst cycle. Specifically, the radical species captures an electron from Cu(I), leading to the formation of a high-valent Cu(III)-SCF3 intermediate. This key intermediate subsequently undergoes transmetallation with the aryl vinyl boronic acid followed by reductive elimination to release the desired aryl vinyl trifluoromethyl sulfide product and regenerate the active Cu(I) catalyst. This radical-mediated pathway ensures rapid turnover and high conversion rates even at lower temperatures.
Alternatively, when N-trifluoromethylthio succinimide is employed, the mechanism shifts towards a more classical organometallic cycle involving ligand acceleration. In this scenario, the copper catalyst, typically CuI, coordinates with a bidentate ligand such as 2,2'-bipyridine to form a stable metal complex in situ. This complex activates the aryl vinyl boronic acid to generate a (bpy)Cu-alkenyl intermediate, which is crucial for the subsequent bond-forming event. 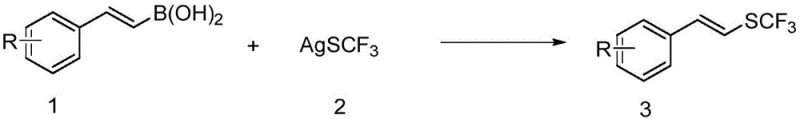
 The alkenyl-copper species then reacts with the electrophilic sulfur source, promoting metal transfer under alkaline conditions to forge the C-S bond. This dual-pathway capability highlights the versatility of the copper catalytic system, allowing chemists to tune the reaction conditions based on the specific electronic properties of the substrate and the availability of reagents, ensuring optimal yields across a wide range of molecular architectures.
The alkenyl-copper species then reacts with the electrophilic sulfur source, promoting metal transfer under alkaline conditions to forge the C-S bond. This dual-pathway capability highlights the versatility of the copper catalytic system, allowing chemists to tune the reaction conditions based on the specific electronic properties of the substrate and the availability of reagents, ensuring optimal yields across a wide range of molecular architectures.
How to Synthesize Aryl Vinyl Trifluoromethyl Sulfide Efficiently
The practical execution of this synthesis is designed for reproducibility and safety, making it highly suitable for both laboratory optimization and pilot plant operations. The general procedure involves charging a reaction vessel with the aryl vinyl boronic acid substrate, the chosen trifluoromethylthio source, a copper catalyst like cuprous iodide, and an appropriate base such as cesium carbonate or sodium carbonate. The reaction is conducted in polar aprotic solvents like dimethyl sulfoxide (DMSO) or acetonitrile, which facilitate the solubility of inorganic salts and stabilize the charged intermediates. Maintaining an inert nitrogen atmosphere is critical to prevent the oxidation of the copper catalyst and the degradation of sensitive reagents. Following the reaction period of 4 to 24 hours, the workup is straightforward, involving aqueous extraction and purification via flash column chromatography to yield the target compound with high purity. For a detailed, step-by-step standard operating procedure including specific molar ratios and safety precautions, please refer to the technical guide below.
- Prepare the reaction mixture by combining aryl vinyl boronic acid, a trifluoromethylthio source (such as AgSCF3 or N-trifluoromethylthio succinimide), a copper catalyst, and a base in a suitable solvent like DMSO or acetonitrile.
- Maintain the reaction under an inert nitrogen atmosphere at temperatures ranging from 25°C to 100°C for a duration of 4 to 24 hours to ensure complete conversion.
- Upon completion, purify the crude mixture through filtration, washing, and flash column chromatography to isolate the high-purity aryl vinyl trifluoromethyl sulfide product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers compelling advantages that directly address the pain points of cost volatility and supply chain fragility in the fine chemicals sector. By shifting the feedstock from specialized halides to commodity boronic acids, manufacturers can leverage a more robust and competitive global supply market for raw materials. The elimination of precious metal catalysts in favor of abundant copper salts drastically reduces the bill of materials cost, while also simplifying the regulatory burden associated with heavy metal residue limits in final pharmaceutical products. Furthermore, the mild reaction conditions translate to lower energy consumption and reduced wear on reactor equipment, contributing to significant operational expenditure savings over the lifecycle of the product. These factors combine to create a more resilient supply chain capable of withstanding market fluctuations and ensuring consistent delivery schedules for downstream clients.
- Cost Reduction in Manufacturing: The substitution of expensive palladium or nickel catalysts with inexpensive copper salts represents a fundamental shift in cost structure for fluorine chemistry. Since copper is orders of magnitude cheaper than precious metals and does not require complex ligand systems in all variations, the direct material cost is significantly lowered. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, reducing the volume of solvents and silica gel required for purification, which further drives down the cost of goods sold. This economic efficiency makes the production of high-value fluorinated intermediates viable for broader therapeutic areas where cost sensitivity is higher.
- Enhanced Supply Chain Reliability: Aryl vinyl boronic acids are widely produced intermediates with established supply chains, unlike many specialized trifluoromethylthio halides which may have single-source suppliers. By utilizing these stable and commercially available starting materials, procurement managers can mitigate the risk of supply disruptions caused by geopolitical issues or production outages at niche chemical plants. The stability of the boronic acid substrates also allows for longer shelf life and easier logistics, reducing waste from expired raw materials and enabling just-in-time inventory strategies that optimize working capital.
- Scalability and Environmental Compliance: The process operates at atmospheric pressure and moderate temperatures, removing the need for specialized high-pressure reactors or cryogenic infrastructure that often limits batch sizes in traditional fluorination chemistry. This inherent safety profile facilitates easier scale-up from kilogram to tonne quantities without significant engineering changes. Moreover, the use of copper and the avoidance of toxic tin or mercury reagents aligns with increasingly stringent environmental regulations, simplifying waste disposal and reducing the carbon footprint of the manufacturing process, which is a key metric for sustainable sourcing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this deboronative trifluoromethylthiolation technology. These insights are derived directly from the experimental data and scope defined in the patent literature, providing a clear understanding of the method's capabilities and limitations for potential adopters. Understanding these nuances is essential for R&D teams evaluating route selection and for supply chain partners assessing the feasibility of long-term contracts. We encourage stakeholders to review these details to fully appreciate the strategic value this chemistry brings to the production of complex fluorinated molecules.
Q: What are the primary advantages of using aryl vinyl boronic acids over haloarylethenes for SCF3 introduction?
A: Aryl vinyl boronic acids offer superior stability and ease of synthesis compared to haloarylethenes. The deboronative approach described in CN112608260A allows for milder reaction conditions (25-100°C) and avoids the harsh conditions often required for halide substitution, leading to better E/Z selectivity and simplified purification processes.
Q: Which trifluoromethylthio sources are compatible with this copper-catalyzed system?
A: The patent specifies two primary sources: silver trifluoromethyl mercaptan (AgSCF3) and N-trifluoromethylthio succinimide. AgSCF3 typically requires an oxidant like potassium persulfate to generate the radical species, while the succinimide derivative functions effectively with a ligand such as 2,2'-bipyridine under basic conditions.
Q: How does this method impact the scalability of fluorine-containing API intermediates?
A: This method significantly enhances scalability by utilizing inexpensive copper catalysts instead of precious metals like palladium. The use of stable boronic acid substrates and the ability to operate at ambient to moderate temperatures reduces energy consumption and safety risks associated with cryogenic or high-pressure reactions, facilitating easier commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Vinyl Trifluoromethyl Sulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed deboronation technology in accelerating the development of novel fluorinated therapeutics and agrochemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including dedicated containment systems and rigorous QC labs that enforce stringent purity specifications for every batch. We are committed to delivering high-quality aryl vinyl trifluoromethyl sulfide intermediates that meet the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with our technical team to explore how this innovative synthetic route can optimize your specific project requirements. By engaging with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this boronic acid-based methodology for your pipeline. Please contact our technical procurement team today to request specific COA data for our catalog compounds or to discuss custom route feasibility assessments tailored to your unique molecular targets. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
