Advanced Manufacturing of Trans-(1R,2S)-2-(3,4-difluorophenyl)cyclopropylamine: A Strategic Upgrade for API Production
Advanced Manufacturing of Trans-(1R,2S)-2-(3,4-difluorophenyl)cyclopropylamine: A Strategic Upgrade for API Production
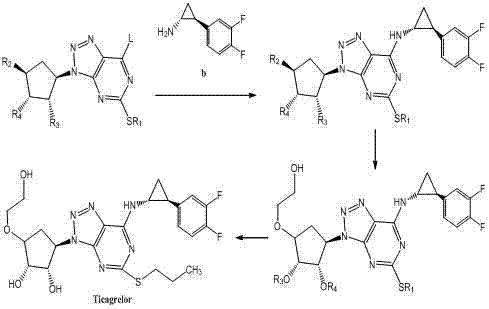
The pharmaceutical industry's relentless pursuit of efficient, safe, and scalable synthetic routes for critical anticoagulant medications has led to significant innovations in intermediate manufacturing. Patent CN102249929A introduces a transformative methodology for synthesizing trans-(1R,2S)-2-(3,4-difluorophenyl)cyclopropylamine, a pivotal chiral building block for the blockbuster antiplatelet drug Ticagrelor. This patent details a robust chemical pathway that leverages diphenylphosphoryl azide (DPPA) mediated Curtius rearrangement to bypass the severe safety hazards and operational complexities associated with historical production methods. For R&D directors and supply chain strategists, this technology represents a paradigm shift from hazardous, multi-step batch processes to a streamlined, thermally stable 'one-pot' operation. By fundamentally altering the reaction mechanism to avoid the direct handling of explosive sodium azide and corrosive thionyl chloride, this approach not only enhances workplace safety but also drastically simplifies the downstream purification landscape, ensuring a more reliable supply of high-purity intermediates for global API manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthesis routes for this critical cyclopropylamine intermediate, as documented in prior art such as CN1334816 and CN1431992A, are plagued by inherent safety risks and inefficient process controls that pose significant challenges for industrial scale-up. Traditional pathways frequently rely on the use of sodium azide, a reagent known for its extreme toxicity and potential for violent decomposition, necessitating expensive containment infrastructure and rigorous safety monitoring that drives up operational expenditures. Furthermore, these legacy processes often involve harsh reagents like thionyl chloride and require drastic thermal cycling, where reaction temperatures must be rapidly swung from 100°C down to below 0°C to manage exotherms and selectivity. Such extreme thermal gradients place immense stress on reactor vessels and utility systems, leading to increased maintenance costs and potential batch failures due to poor temperature homogeneity in large reactors. Additionally, the multi-step nature of these conventional routes, which often require the isolation of unstable acyl azides or isocyanates, results in cumulative yield losses and generates substantial volumes of hazardous waste that complicate environmental compliance.
The Novel Approach
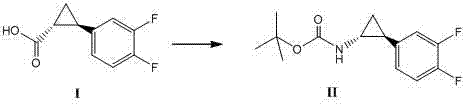
In stark contrast to these cumbersome legacy techniques, the methodology disclosed in CN102249929A utilizes a sophisticated DPPA-mediated rearrangement that operates under significantly milder and more controllable conditions, effectively eliminating the need for isolated hazardous intermediates. This innovative route allows for the direct conversion of the carboxylic acid precursor into the protected amine derivative within a single reaction vessel, maintaining a steady thermal profile between 60°C and 100°C without the need for cryogenic cooling. By integrating the rearrangement and subsequent deprotection steps into a cohesive 'one-pot' sequence, the process minimizes unit operations, reduces solvent consumption, and prevents the accumulation of dangerous azide species that characterize older technologies. The use of tert-butanol as a solvent and triethylamine as a base further enhances the reaction's stability, providing a homogeneous medium that facilitates efficient heat transfer and mass transport. This streamlined approach not only accelerates the overall production timeline but also ensures a cleaner reaction profile, which is critical for meeting the stringent impurity specifications required for cardiovascular pharmaceutical ingredients.
Mechanistic Insights into DPPA-Mediated Curtius Rearrangement
The core chemical innovation driving this process is the in situ generation and rearrangement of an acyl azide intermediate using diphenylphosphoryl azide (DPPA), a reagent that offers superior handling safety compared to hydrazoic acid or sodium azide salts. Mechanistically, the carboxylic acid substrate first reacts with DPPA in the presence of a tertiary amine base to form a mixed anhydride, which subsequently undergoes nucleophilic attack by the azide moiety to generate the acyl azide. Upon heating to the optimal range of 80°C to 90°C, this acyl azide undergoes a concerted Curtius rearrangement, extruding nitrogen gas to form a highly reactive isocyanate species. In the presence of the tert-butanol solvent, this isocyanate is immediately trapped to form the tert-butoxycarbonyl (Boc) protected amine, effectively stabilizing the reactive intermediate and preventing side reactions such as urea formation or polymerization. This mechanistic pathway is exceptionally clean because the nitrogen gas byproduct simply vents from the reaction mixture, driving the equilibrium forward and eliminating the need for complex workup procedures to remove inorganic salts typically associated with azide chemistry.
From an impurity control perspective, this mechanism provides a distinct advantage by avoiding the formation of chlorinated byproducts that are common when thionyl chloride is used in traditional activation steps. The absence of chlorine sources means that the final product is free from genotoxic alkyl chloride impurities, a critical quality attribute for regulatory approval in oncology and cardiovascular drug substances. Furthermore, the stereospecificity of the Curtius rearrangement ensures that the chiral integrity of the cyclopropane ring is preserved throughout the transformation, maintaining the crucial (1R,2S) configuration required for biological activity. The subsequent acid-mediated deprotection step is equally elegant, utilizing hydrochloric acid to cleave the Boc group under mild conditions, followed by a simple pH adjustment to liberate the free amine. This two-stage cascade—rearrangement followed by deprotection—can be executed sequentially in the same vessel, minimizing exposure to air and moisture which could otherwise degrade the sensitive amine product, thereby ensuring high optical purity and chemical stability.
How to Synthesize Trans-(1R,2S)-2-(3,4-difluorophenyl)cyclopropylamine Efficiently
The practical implementation of this synthesis involves dissolving the chiral carboxylic acid starting material alongside DPPA and triethylamine in tert-butanol, followed by heating the mixture to induce the rearrangement. Once the formation of the Boc-protected intermediate is confirmed, typically via TLC or HPLC monitoring, the reaction mixture is concentrated and treated directly with hydrochloric acid to remove the protecting group. The final step involves neutralizing the acidic solution to a pH of 8 to 10 using a base like sodium bicarbonate, which precipitates or allows for the extraction of the pure amine product. This standardized protocol eliminates the need for cryogenic equipment and hazardous reagent handling, making it ideally suited for multi-kilogram to ton-scale manufacturing campaigns. For detailed operational parameters and specific stoichiometric ratios, please refer to the structured guide below.
- Dissolve trans-(1R,2R)-2-(3,4-difluorophenyl)cyclopropanecarboxylic acid, DPPA, and triethylamine in tert-butanol and heat to 60-100°C for rearrangement.
- Remove the solvent and treat the resulting Boc-protected intermediate with hydrochloric acid to cleave the protecting group.
- Adjust the pH of the reaction system to alkaline conditions (pH 8-10) to isolate the final free amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this DPPA-based synthetic route offers profound strategic benefits that extend far beyond simple chemical yield improvements, fundamentally altering the cost structure and risk profile of the supply chain. By transitioning away from explosive sodium azide and corrosive thionyl chloride, manufacturers can significantly reduce the costs associated with specialized hazardous waste disposal, safety insurance premiums, and regulatory compliance reporting. The elimination of extreme thermal cycling requirements means that existing standard glass-lined or stainless steel reactors can be utilized without modification, avoiding the capital expenditure needed for cryogenic cooling systems or specialized low-temperature infrastructure. Furthermore, the 'one-pot' nature of the synthesis drastically reduces the total cycle time per batch, increasing the asset utilization rate of production facilities and allowing for greater throughput without expanding the physical footprint of the plant. These operational efficiencies translate directly into a more resilient supply chain capable of responding rapidly to fluctuations in global demand for Ticagrelor, ensuring continuity of supply even during periods of market volatility.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the consolidation of multiple reaction steps into a single vessel operation, which dramatically lowers solvent consumption and labor hours per kilogram of product. By avoiding the isolation of intermediates, the process eliminates the yield losses typically incurred during filtration, drying, and re-dissolution steps, leading to a substantial improvement in overall material efficiency. Additionally, the use of DPPA, while a specialized reagent, removes the need for expensive and logistically challenging reagents like liquid ammonia or specialized azide solutions, simplifying the raw material procurement portfolio. The reduction in hazardous waste generation further lowers disposal costs, contributing to a leaner and more cost-effective manufacturing model that enhances margin potential for generic API producers.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of the reaction conditions, which are tolerant to minor variations in temperature and mixing rates compared to the finicky low-temperature protocols of the past. The reliance on commercially available and stable reagents like DPPA and tert-butanol ensures that raw material sourcing is not subject to the geopolitical or logistical bottlenecks often associated with controlled precursors like sodium azide. Moreover, the simplified workflow reduces the number of quality control checkpoints required between steps, accelerating the release time for finished batches and shortening the lead time for delivering high-purity pharmaceutical intermediates to downstream API synthesis sites. This reliability is crucial for maintaining just-in-time inventory levels and preventing production stoppages in the final drug formulation stages.
- Scalability and Environmental Compliance: From an environmental and scalability standpoint, this method aligns perfectly with modern green chemistry principles by minimizing the E-factor (mass of waste per mass of product) through solvent recycling and reduced reagent usage. The absence of heavy metal catalysts or persistent organic pollutants in the waste stream simplifies wastewater treatment processes, ensuring compliance with increasingly stringent environmental regulations in major manufacturing hubs. The thermal stability of the rearrangement step allows for straightforward scale-up from pilot plant to commercial production scales of 100 MT or more without encountering the heat transfer limitations that plague exothermic azide reactions. This scalability ensures that suppliers can meet the growing global volume requirements for anticoagulant therapies without compromising on safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway, derived directly from the comparative data and experimental examples provided in the patent literature. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of this supply source. The answers reflect the specific advantages of the DPPA route over traditional methods, focusing on safety, yield, and operational simplicity.
Q: Why is the DPPA rearrangement method safer than traditional azide routes?
A: Traditional methods often utilize sodium azide, which is explosive and highly toxic, requiring stringent safety protocols. The DPPA method generates the acyl azide in situ under controlled thermal conditions, significantly mitigating the risk of explosion and simplifying hazard management in large-scale production.
Q: How does this process improve yield compared to prior art?
A: By employing a 'one-pot' strategy that combines the rearrangement and deprotection steps without isolating unstable intermediates, the process minimizes material loss during transfer and purification. This continuity leads to substantially higher overall yields compared to multi-step isolation processes found in older patents.
Q: What are the critical temperature controls for this synthesis?
A: The rearrangement reaction is optimally conducted between 60°C and 100°C, specifically around 80-90°C. This avoids the extreme thermal shocks seen in legacy methods, which required cooling from 100°C to below 0°C, thereby reducing energy consumption and equipment stress.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-(1R,2S)-2-(3,4-difluorophenyl)cyclopropylamine Supplier
As the global demand for next-generation anticoagulants continues to rise, securing a supply partner with deep technical expertise in complex chiral intermediates is paramount for maintaining competitive advantage in the pharmaceutical market. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent, high-quality intermediates. Our state-of-the-art facilities are equipped to handle the precise thermal and safety requirements of advanced rearrangement chemistries, ensuring that every batch meets stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. We understand that the integrity of your final API depends on the quality of the starting materials, which is why we adhere to the highest standards of GMP-compliant manufacturing and traceability.
We invite you to collaborate with us to optimize your supply chain for Ticagrelor and related cardiovascular therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized DPPA-based process can reduce your total cost of ownership. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines and quality expectations. Let us be your strategic partner in navigating the complexities of fine chemical manufacturing.
