Advanced Chiral ONO Tridentate Ligand and Cobalt Complex for Commercial Scale-up
Advanced Chiral ONO Tridentate Ligand and Cobalt Complex for Commercial Scale-up
The chemical industry is constantly seeking robust solutions for asymmetric synthesis and chiral separation, areas where traditional methods often face significant economic and technical hurdles. Patent CN113087640B introduces a groundbreaking chiral ONO tridentate Schiff base carboxylic acid ligand that addresses these challenges through a novel structural design and efficient preparation method. This innovation not only simplifies the synthesis of chiral coordination compounds but also opens new avenues for creating rich structural diversity in supramolecular materials. By leveraging cheap raw materials and a straightforward reaction pathway, this technology offers a compelling value proposition for manufacturers aiming to optimize their production of high-purity specialty chemicals. The strategic implementation of this ligand system can significantly enhance the efficiency of chiral catalyst preparation, making it a critical asset for R&D teams focused on next-generation pharmaceutical intermediates and functional materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral coordination materials has been hindered by the high cost and complexity associated with acquiring suitable chiral ligands. Traditional approaches often rely on expensive chiral sources that drive up the overall production cost, making large-scale application economically unfeasible for many industrial processes. Furthermore, conventional ligands frequently exhibit simple coordination modes, such as the common N2O2 pockets, which limit the structural diversity and functional tunability of the resulting metal complexes. This lack of structural richness restricts their utility in advanced applications like gas storage, fluorescence sensing, and sophisticated catalytic separations. The synthesis difficulty associated with these traditional ligands also poses a significant barrier, requiring harsh conditions or multi-step purifications that reduce overall yield and operational efficiency. Consequently, supply chains for high-performance chiral materials remain fragile and cost-prohibitive.
The Novel Approach
The technology disclosed in CN113087640B overcomes these limitations by introducing an asymmetric ONO tridentate chiral Schiff base carboxylic acid ligand that is both cost-effective and structurally versatile. This novel approach utilizes abundant and inexpensive raw materials, such as 1-adamantanol and methyl 4-hydroxybenzoate, to construct a robust ligand framework through simple organic reactions. The unique ONO chelate coordination mode allows for the formation of complexes with richer structures compared to traditional symmetric ligands, enabling the creation of diverse chiral environments essential for high-precision applications. Additionally, the presence of a carboxyl group in the ligand skeleton facilitates further functionalization and supramolecular assembly via hydrogen bonding. This method drastically simplifies the synthesis process, reducing the technical barriers to entry and allowing for more reliable and scalable manufacturing of chiral coordination materials.
Mechanistic Insights into ONO Tridentate Chelation and Cobalt Coordination
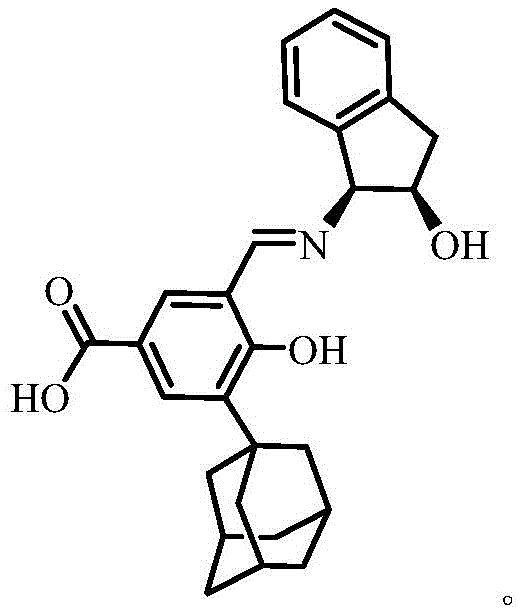
The core of this technological advancement lies in the precise molecular architecture of the chiral ONO tridentate Schiff base carboxylic acid ligand, designated as L-H3. The ligand features a rigid adamantane group and a chiral indanol moiety linked through a Schiff base bond, creating a sterically demanding environment that enforces specific stereochemical outcomes during coordination. The ONO tridentate binding site, comprising phenolic oxygen, imine nitrogen, and alcoholic oxygen, forms a stable five-membered chelate ring with metal centers, ensuring high thermal and chemical stability. This specific arrangement prevents ligand dissociation under reaction conditions, which is crucial for maintaining catalytic activity and structural integrity in demanding industrial applications. The asymmetry introduced by the chiral source is effectively transferred to the metal center, creating a chiral pocket capable of distinguishing between enantiomers in substrate molecules.
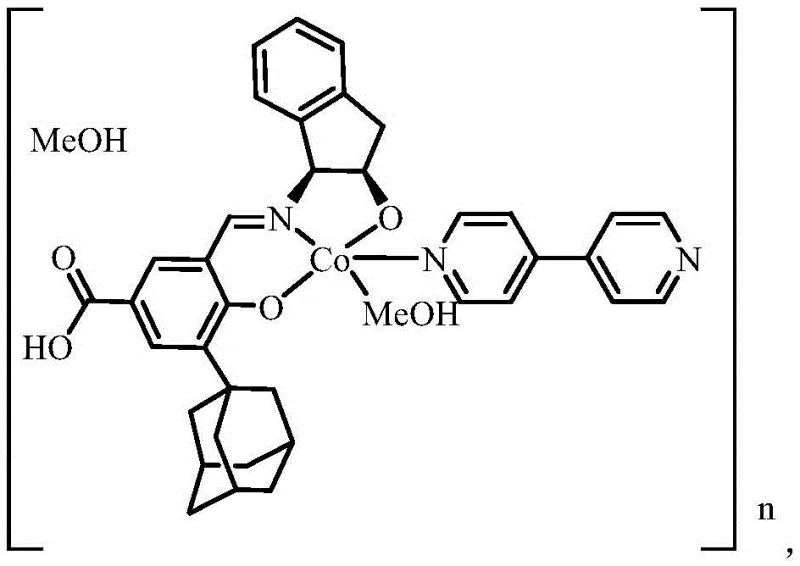
Upon coordination with cobalt ions, the ligand self-assembles into a sophisticated cobalt-based chiral complex with a unique two-dimensional network structure. The central cobalt ion adopts a distorted octahedral coordination geometry, where the equatorial plane is occupied by the ONO donor atoms of the ligand and a coordinated methanol molecule. The axial positions are filled by nitrogen atoms from 4,4'-bipyridine auxiliary ligands, extending the structure into one-dimensional chains. Crucially, the terminal carboxyl groups that do not participate in direct metal coordination serve as hydrogen bonding sites, linking adjacent chains into a robust 2D supramolecular network. This hierarchical assembly enhances the material's stability and creates defined channels that can be exploited for guest molecule inclusion or selective transport phenomena.
How to Synthesize Chiral ONO Ligand Efficiently
The synthesis of this high-value ligand is designed for operational simplicity and scalability, making it an ideal candidate for commercial production. The process involves a sequence of four distinct chemical transformations that convert readily available starting materials into the final chiral product with high purity. Each step is optimized to minimize waste and maximize yield, utilizing standard laboratory equipment and reagents that are easily sourced from global chemical suppliers. The detailed standardized synthesis steps provided below outline the precise conditions, stoichiometry, and workup procedures required to achieve consistent results. This transparency in process design allows manufacturing teams to quickly evaluate feasibility and integrate the technology into existing production lines with minimal retooling.
- Perform Friedel-Crafts alkylation of methyl 4-hydroxybenzoate with 1-adamantanol using sulfuric acid catalyst.
- Conduct formylation with paraformaldehyde and triethylamine to introduce the aldehyde group.
- Hydrolyze the ester group using lithium hydroxide to obtain the carboxylic acid precursor.
- Condense the aldehyde precursor with (1S,2R)-1-amino-2,3-dihydro-1H-indene-2-ol to form the final Schiff base ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this ligand technology translates into tangible strategic advantages regarding cost stability and supply reliability. The reliance on cheap and abundant raw materials significantly mitigates the risk of price volatility often associated with specialized chiral reagents. By eliminating the need for expensive proprietary chiral sources, manufacturers can achieve substantial cost savings in the overall production of chiral intermediates. Furthermore, the simplified synthetic route reduces the number of unit operations required, leading to lower energy consumption and reduced waste disposal costs. This efficiency gain directly impacts the bottom line, allowing companies to offer more competitive pricing for their downstream products while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The use of inexpensive starting materials like adamantanol and methyl 4-hydroxybenzoate drastically lowers the raw material cost base compared to traditional chiral ligand synthesis. The elimination of complex purification steps and the ability to use crude intermediates in subsequent reactions further reduce processing expenses. This economic efficiency allows for significant cost reduction in fine chemical manufacturing, making high-performance chiral materials accessible for broader industrial applications without compromising quality.
- Enhanced Supply Chain Reliability: Since the raw materials are commodity chemicals with established global supply chains, the risk of supply disruption is minimized. The robust nature of the synthesis process ensures consistent output quality, reducing the likelihood of batch failures that can delay downstream production schedules. This reliability is critical for maintaining continuous manufacturing operations and meeting strict delivery commitments to international clients, thereby strengthening the overall resilience of the supply network.
- Scalability and Environmental Compliance: The reaction conditions are mild, typically operating at atmospheric pressure and moderate temperatures, which simplifies the engineering requirements for scale-up. The use of common solvents and the generation of manageable waste streams facilitate compliance with environmental regulations. This ease of scale-up ensures that production can be rapidly increased from pilot to commercial scale to meet market demand, supporting the commercial scale-up of complex specialty chemicals with minimal environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the operational benefits and structural characteristics of the material. Understanding these details is essential for technical teams evaluating the feasibility of integrating this ligand into their specific catalytic or material science applications. The information provided here serves as a foundational guide for further discussions with our technical sales team.
Q: What are the cost advantages of this chiral ligand synthesis?
A: The process utilizes cheap and readily available raw materials like adamantanol and methyl 4-hydroxybenzoate, significantly reducing precursor costs compared to traditional expensive chiral sources.
Q: How does the ONO coordination mode benefit complex stability?
A: The unique ONO tridentate chelating mode provides a rigid coordination environment, enhancing the thermal and chemical stability of the resulting cobalt-based supramolecular structures.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the synthesis involves standard organic reactions at moderate temperatures and pressures, avoiding extreme conditions, which facilitates straightforward commercial scale-up and operational safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral ONO Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral ligands in driving innovation within the pharmaceutical and fine chemical sectors. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing the consistency required for sensitive asymmetric synthesis applications. Our expertise in coordination chemistry allows us to support clients not just with materials, but with the technical insights needed to optimize their processes.
We invite you to collaborate with us to unlock the full potential of this advanced ligand technology for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volume. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our supply solutions can enhance your operational efficiency. Let us be your partner in achieving excellence in chiral chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
