Revolutionizing Chiral Sensing: Scalable Synthesis of C2 Symmetric Fluorescent Ligand L-H2 for High-Performance MOFs
The landscape of chiral chemistry is undergoing a significant transformation with the emergence of multifunctional materials capable of simultaneous separation and sensing. Patent CN113831268B introduces a groundbreaking C2 symmetric fluorescent chiral carboxylic acid ligand, designated as L-H2, which addresses critical limitations in current chiral porous material design. This innovation leverages a robust molecular architecture containing luminescent conjugated groups, flexible sulfonyl linkers, and coordinative carboxylic acid moieties. By reacting this ligand with zinc ions, manufacturers can produce zinc-based chiral porous coordination polymers that exhibit open chiral nanochannels measuring approximately 1.06 nm × 0.44 nm. These materials are not merely structural curiosities but functional tools designed for the selective recognition and adsorption of styrene oxide isomers, alongside rapid fluorescence-based identification of R-configured enantiomers. For R&D directors and procurement specialists, this represents a pivotal shift towards high-value, dual-functionality intermediates that streamline analytical and separation workflows in pharmaceutical and fine chemical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of chiral porous coordination polymers (PCPs) or metal-organic frameworks (MOFs) has been hindered by a trade-off between structural complexity and functional versatility. Traditional approaches often rely on ligands that provide either chiral recognition sites or structural stability, but rarely both in a synergistic manner that allows for real-time monitoring. Many existing chiral separation materials lack intrinsic fluorescence, necessitating complex downstream analytical procedures to verify enantiomeric purity after separation. Furthermore, the synthesis of sophisticated chiral ligands frequently involves multi-step pathways with harsh conditions, expensive catalysts, or difficult-to-source starting materials, leading to prohibitive costs for large-scale application. The inability to construct materials with both defined pore structures and multiple recognition sites has limited the efficiency of chiral resolution processes, particularly for small molecule epoxy compounds like styrene oxide where rapid identification is crucial for quality control.
The Novel Approach
The methodology disclosed in the patent offers a streamlined, cost-effective solution by utilizing L-phenylalanine as a chiral source and naphthalene-1,5-disulfonyl chloride as a luminescent building block. This strategy simplifies the synthetic route into three manageable organic reactions: esterification, sulfonamide coupling, and hydrolysis. The resulting ligand L-H2 possesses a C2 symmetric structure that inherently promotes the formation of ordered porous networks when coordinated with metal ions. Unlike previous single-function materials, this novel approach integrates a flexible sulfonyl group that aids in the formation of open nanochannels while maintaining the rigidity required for stable crystal lattices. 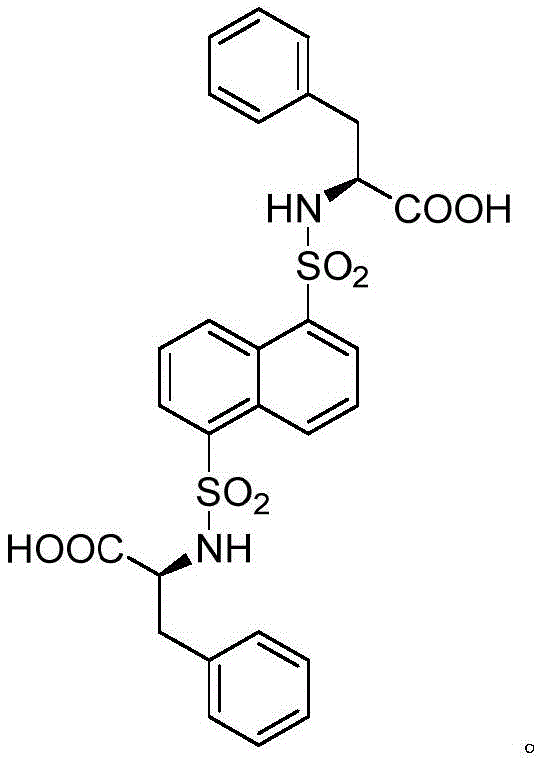 This synthetic accessibility ensures that the production of high-purity ligands can be scaled without the exponential cost increases typically associated with complex chiral auxiliaries, making it an attractive option for reliable chiral ligand suppliers aiming to reduce lead times for high-purity pharmaceutical intermediates.
This synthetic accessibility ensures that the production of high-purity ligands can be scaled without the exponential cost increases typically associated with complex chiral auxiliaries, making it an attractive option for reliable chiral ligand suppliers aiming to reduce lead times for high-purity pharmaceutical intermediates.
Mechanistic Insights into Zinc-Based Chiral Porous Coordination Polymer Formation
The formation of the functional material relies on the precise coordination chemistry between the L-H2 ligand and zinc ions, facilitated by 4,4'-bipyridine as an auxiliary ligand. The central Zn(II) ion adopts a distorted tetrahedral geometry, coordinating with two carboxyl oxygen atoms from a single L-H2 ligand and two pyridine nitrogen atoms from distinct bipyridine molecules. This specific coordination mode drives the self-assembly of the components into a unique two-dimensional network structure that further stacks into a three-dimensional supramolecular framework via hydrogen bonding and pi-pi interactions. The presence of the flexible sulfonyl bonds in the ligand backbone is critical, as it allows the carboxyl groups to orient in a 'head-to-head' fashion, creating ring-like ZnL structures that serve as the nodes for the extended lattice. This architectural precision results in the generation of narrow, open chiral nanochannels that are perfectly sized to accommodate guest molecules like styrene oxide, providing a physical basis for size-exclusion and shape-selective adsorption mechanisms.
Beyond structural integrity, the mechanism of chiral recognition is deeply embedded in the chemical environment of the pore walls. The internal surfaces of these nanochannels are lined with abundant functional groups, including amide moieties, carbonyls, benzyl groups, and naphthalene rings, all oriented towards the void space. 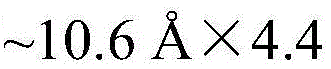 These groups act as diverse interaction sites, engaging guest molecules through hydrogen bonding, dipole-dipole interactions, and pi-pi stacking forces. Crucially, the chiral environment created by the L-phenylalanine derivatives ensures that these interactions are enantioselective; the R-configured styrene oxide fits more snugly and interacts more strongly with the pore walls than its S-counterpart. This differential binding affinity not only drives the preferential adsorption of the R-isomer but also induces a significant quenching of the material's intrinsic fluorescence, thereby enabling a dual-mode detection system that combines physical separation with optical sensing capabilities for enhanced process analytical technology.
These groups act as diverse interaction sites, engaging guest molecules through hydrogen bonding, dipole-dipole interactions, and pi-pi stacking forces. Crucially, the chiral environment created by the L-phenylalanine derivatives ensures that these interactions are enantioselective; the R-configured styrene oxide fits more snugly and interacts more strongly with the pore walls than its S-counterpart. This differential binding affinity not only drives the preferential adsorption of the R-isomer but also induces a significant quenching of the material's intrinsic fluorescence, thereby enabling a dual-mode detection system that combines physical separation with optical sensing capabilities for enhanced process analytical technology.
How to Synthesize L-H2 Efficiently
The preparation of the C2 symmetric fluorescent chiral carboxylic acid ligand L-H2 is designed for operational simplicity and high reproducibility, making it suitable for transfer from laboratory discovery to commercial manufacturing. The process begins with the activation of L-phenylalanine, followed by a condensation reaction that installs the fluorescent naphthalene core, and concludes with a hydrolysis step to reveal the active carboxylic acid groups necessary for metal coordination. Each step utilizes common reagents and standard workup procedures such as solvent extraction and column chromatography, minimizing the need for specialized equipment. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and reaction times optimized for maximum yield and purity, are outlined in the guide below for technical teams evaluating process feasibility.
- Esterify L-phenylalanine using thionyl chloride in anhydrous methanol, followed by pH adjustment and extraction to obtain the methyl ester crude product.
- React the L-phenylalanine methyl ester with naphthalene-1,5-disulfonyl chloride in ultra-dry dichloromethane with triethylamine to form the sulfonamide bridge.
- Hydrolyze the methylated intermediate using sodium hydroxide in a THF/methanol/water mixture, followed by acidification and purification to yield the final ligand L-H2.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers substantial strategic benefits rooted in raw material availability and process robustness. The reliance on L-phenylalanine, a commodity chiral amino acid produced on a massive global scale, eliminates the supply chain risks associated with exotic or proprietary chiral pool starting materials. This foundational stability ensures consistent pricing and availability, shielding manufacturing operations from the volatility often seen in the specialty chemical market. Furthermore, the synthetic pathway avoids the use of precious metal catalysts or hazardous reagents that require complex disposal protocols, thereby reducing the environmental compliance burden and associated waste management costs. The mild reaction conditions, predominantly at room temperature, also translate to lower energy consumption during production, contributing to a more sustainable and cost-efficient manufacturing profile that aligns with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The economic viability of this ligand is significantly enhanced by the elimination of expensive transition metal catalysts and the use of straightforward organic transformations. By utilizing a three-step sequence that proceeds with high efficiency, the overall process mass intensity is optimized, leading to reduced solvent usage and lower raw material costs per kilogram of finished product. The avoidance of cryogenic conditions or high-pressure reactors further decreases capital expenditure requirements for production facilities, allowing for cost reduction in pharmaceutical intermediate manufacturing without compromising on the quality or performance of the final chiral material.
- Enhanced Supply Chain Reliability: The synthesis relies on chemically stable intermediates and widely available reagents such as thionyl chloride, naphthalene disulfonyl chloride, and common organic solvents like dichloromethane and methanol. This reduces the dependency on single-source suppliers for critical inputs, thereby mitigating the risk of production stoppages due to raw material shortages. The robustness of the chemical steps ensures high batch-to-b consistency, which is essential for maintaining long-term supply contracts with downstream users in the agrochemical and pharmaceutical sectors who require rigorous quality assurance and uninterrupted material flow.
- Scalability and Environmental Compliance: The process is inherently scalable due to its linear nature and the absence of sensitive catalytic cycles that often fail upon scale-up. The byproducts generated during the synthesis are manageable and can be treated using standard effluent processing methods, ensuring compliance with stringent environmental regulations. Additionally, the final zinc-based polymer material itself offers environmental benefits by enabling more efficient chiral separations, potentially reducing the solvent load in downstream purification processes. This combination of scalable synthesis and eco-friendly application positions the technology as a sustainable choice for the commercial scale-up of complex polymer additives and chiral sensors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on performance metrics and operational parameters. Understanding these details is crucial for technical teams assessing the fit of this material within existing chiral separation workflows or sensor development pipelines.
Q: What are the primary advantages of ligand L-H2 over traditional chiral ligands?
A: Unlike traditional ligands that often possess only a single function, L-H2 combines a luminescent conjugated naphthalene group with flexible sulfonyl groups and coordinative carboxylic acids. This unique architecture allows for the construction of zinc-based porous materials that simultaneously perform chiral separation and fluorescence sensing.
Q: Is the synthesis of L-H2 scalable for industrial production?
A: Yes, the synthesis utilizes readily available L-phenylalanine and involves standard organic transformations like esterification, sulfonamide formation, and hydrolysis. The reactions proceed under mild conditions (mostly room temperature), avoiding extreme pressures or temperatures, which facilitates safe and cost-effective scale-up.
Q: What specific applications does the resulting zinc-based polymer support?
A: The resulting zinc-based chiral porous coordination polymer features open nanochannels (approx. 1.06 nm × 0.44 nm) and abundant recognition sites. It is specifically validated for the selective adsorption and rapid fluorescence sensing of styrene oxide isomers, making it valuable for chiral analysis in fine chemical manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-H2 Ligand Supplier
The technical potential of C2 symmetric fluorescent chiral ligands like L-H2 is immense, offering a pathway to next-generation chiral sensing and separation materials that outperform traditional solutions. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries can be seamlessly translated into industrial reality. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of ligand or polymer material meets the exacting standards required for high-performance applications in the life sciences and advanced materials sectors.
We invite you to collaborate with us to explore the full commercial potential of this technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your production volume and purity requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in chiral chemistry can drive efficiency and innovation in your supply chain.
