Advanced Synthesis of Fluorinated Diamines for High-Performance Polyimide and Electronic Material Manufacturing
The global demand for high-performance polymers in the electronics and aerospace sectors has necessitated the development of advanced monomers with superior thermal and electrical properties. Patent CN100368389C, filed in early 2008, introduces a groundbreaking preparation method for 2,2-bis[3-amino-4-(4-cyanophenoxy)phenyl]hexafluoropropane, a specialized fluorinated diamine critical for next-generation polyimide resins. This compound serves as a pivotal building block for manufacturing liquid crystal alignment agents, carbon fiber reinforced composites, and passivation coatings for very large scale integration (VLSI) circuits. The patented methodology addresses significant historical challenges in synthesizing fluorinated aromatic diamines, offering a pathway that combines high atomic efficiency with exceptional product purity. By leveraging a specific nucleophilic aromatic substitution strategy enhanced by azeotropic dehydration, this technology enables the production of materials that exhibit markedly lower dielectric constants and improved optical clarity compared to their non-fluorinated counterparts. For R&D directors and procurement specialists alike, understanding the nuances of this synthesis is key to securing a reliable supply chain for high-value electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aromatic diamines containing cyano groups relied heavily on reactions such as the one depicted in prior art literature, specifically the reaction between 2,6-dichlorobenzonitrile and 4-aminophenol. 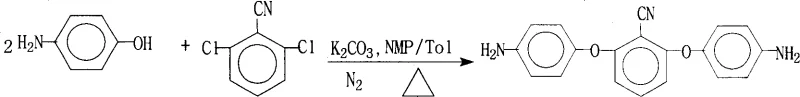 While effective for generating the basic aromatic ether linkage, polymers derived from these non-fluorinated precursors often suffer from inherent drawbacks, including deep coloration and excessively high dielectric constants and loss factors. These physical limitations severely restrict their utility in high-frequency electrical insulation materials and precision liquid crystal display applications where signal integrity and optical transparency are paramount. Furthermore, conventional processes frequently struggle with incomplete conversion rates due to the equilibrium nature of the condensation reactions, often requiring harsh conditions or complex post-synthesis purification steps to remove unreacted starting materials and oligomeric byproducts. The inability to effectively introduce bulky, electronegative fluorine groups using these older methodologies results in final polymer matrices that lack the necessary free volume and hydrophobicity required for advanced moisture resistance and low-k dielectric performance in modern microelectronics.
While effective for generating the basic aromatic ether linkage, polymers derived from these non-fluorinated precursors often suffer from inherent drawbacks, including deep coloration and excessively high dielectric constants and loss factors. These physical limitations severely restrict their utility in high-frequency electrical insulation materials and precision liquid crystal display applications where signal integrity and optical transparency are paramount. Furthermore, conventional processes frequently struggle with incomplete conversion rates due to the equilibrium nature of the condensation reactions, often requiring harsh conditions or complex post-synthesis purification steps to remove unreacted starting materials and oligomeric byproducts. The inability to effectively introduce bulky, electronegative fluorine groups using these older methodologies results in final polymer matrices that lack the necessary free volume and hydrophobicity required for advanced moisture resistance and low-k dielectric performance in modern microelectronics.
The Novel Approach
The innovative strategy outlined in patent CN100368389C fundamentally shifts the synthetic paradigm by utilizing 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane (BAHPFP) as the core nucleophile. 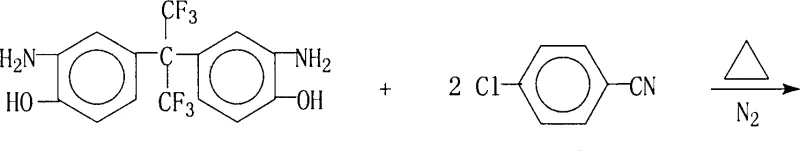 This approach reacts the fluorinated bisphenol with 4-chlorobenzonitrile in a carefully controlled molar ratio ranging from 1:2.0 to 1:2.4, ensuring a slight excess of the electrophile to drive the reaction to completion. Unlike traditional methods that may rely on single-solvent systems, this novel process employs a dual-solvent system comprising a polar aprotic solvent like N,N-dimethylformamide (DMF) and an entrainer such as toluene. This specific combination facilitates an azeotropic distillation mechanism that continuously removes water generated during the etherification, thereby pushing the chemical equilibrium decisively towards the desired product. The reaction is conducted under a nitrogen atmosphere at temperatures between 120°C and 150°C for a duration of 10 to 30 hours, conditions that are optimized to maximize yield while minimizing thermal degradation of the sensitive amino and cyano functional groups. This meticulous control over reaction parameters results in a crude product that is already of high quality, significantly reducing the burden on downstream purification units.
This approach reacts the fluorinated bisphenol with 4-chlorobenzonitrile in a carefully controlled molar ratio ranging from 1:2.0 to 1:2.4, ensuring a slight excess of the electrophile to drive the reaction to completion. Unlike traditional methods that may rely on single-solvent systems, this novel process employs a dual-solvent system comprising a polar aprotic solvent like N,N-dimethylformamide (DMF) and an entrainer such as toluene. This specific combination facilitates an azeotropic distillation mechanism that continuously removes water generated during the etherification, thereby pushing the chemical equilibrium decisively towards the desired product. The reaction is conducted under a nitrogen atmosphere at temperatures between 120°C and 150°C for a duration of 10 to 30 hours, conditions that are optimized to maximize yield while minimizing thermal degradation of the sensitive amino and cyano functional groups. This meticulous control over reaction parameters results in a crude product that is already of high quality, significantly reducing the burden on downstream purification units.
Mechanistic Insights into Base-Catalyzed Nucleophilic Aromatic Substitution
The core chemical transformation driving this synthesis is a base-catalyzed nucleophilic aromatic substitution (SnAr), where the phenoxide anion attacks the electron-deficient aromatic ring of the chlorobenzonitrile. The presence of the strong electron-withdrawing cyano group at the para-position of the chlorobenzene ring activates the carbon-chlorine bond, making it susceptible to nucleophilic attack by the oxygen atom of the fluorinated bisphenol. A critical component of this mechanism is the selection of the salt-forming agent, with potassium carbonate identified as the preferred base due to its optimal balance of basicity and solubility profile in the chosen solvent system. The molar ratio of the base to the bisphenol is maintained between 1.2:1 and 3.2:1, ensuring sufficient deprotonation of the phenolic hydroxyl groups without inducing excessive side reactions or degradation of the amine functionalities. The hexafluoroisopropylidene bridge in the BAHPFP molecule not only imparts the desired low dielectric properties to the final polymer but also influences the electron density of the phenolic rings, subtly tuning their nucleophilicity to match the reactivity of the chlorobenzonitrile electrophile.
Impurity control is intrinsically built into this mechanistic design through the management of water and inorganic salts. As the reaction proceeds, the generation of water is inevitable, which can hydrolyze the nitrile groups or reverse the etherification if not removed. The azeotropic interaction between the polar solvent and the toluene entrainer allows for the physical removal of water as a vapor, preventing hydrolysis and maintaining the activity of the phenoxide nucleophile. Furthermore, the choice of potassium carbonate leads to the formation of potassium chloride and potassium bicarbonate/carbonate byproducts which are largely insoluble in the hot reaction mixture. This insolubility allows for a simple hot filtration step to remove the bulk of inorganic impurities before the product even begins to crystallize. This "reaction-driven purification" minimizes the entrapment of salts within the crystal lattice of the final product, directly contributing to the reported purity levels of 99% and ensuring that the resulting polyimide precursors do not introduce ionic contaminants that could compromise the reliability of electronic devices.
How to Synthesize 2,2-Bis[3-Amino-4-(4-Cyanophenoxy)Phenyl]Hexafluoropropane Efficiently
The operational simplicity of this patented process makes it highly attractive for industrial adoption, as it consolidates multiple unit operations into a streamlined workflow. The synthesis begins with the precise charging of reactants into a reactor equipped with a Dean-Stark trap or similar water separator, emphasizing the importance of water management from the outset. Following the reaction period, the mixture is cooled slightly to approximately 80°C and filtered while hot to remove the precipitated inorganic salts, a step that is crucial for preventing the co-crystallization of impurities. The filtrate, containing the dissolved product, is then subjected to reduced pressure concentration to remove the bulk of the solvents, followed by controlled cooling to induce crystallization. This sequence ensures that the final solid product is obtained with minimal solvent retention and high structural integrity, ready for immediate use in polymerization or further functionalization without extensive recrystallization cycles.
- Charge the reactor with 2,2-bis(3-amino-4-hydroxyphenyl)hexafluoropropane and 4-chlorobenzonitrile in a molar ratio of 1: 2.0-2.4, along with potassium carbonate and a mixed solvent system of DMF and toluene.
- Heat the mixture under nitrogen protection, gradually raising the temperature from 120°C to 150°C over several hours to facilitate azeotropic water removal and drive the etherification reaction to completion.
- Filter the hot reaction mixture to remove inorganic salts, concentrate the mother liquor under reduced pressure, and induce crystallization upon cooling to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented synthesis route offers tangible strategic benefits beyond mere technical performance. The process is designed with industrial scalability in mind, utilizing commodity chemicals such as potassium carbonate, toluene, and DMF which are readily available in the global market, thereby mitigating the risk of raw material shortages. The ability to recover and recycle the solvent system is a major cost driver; since the solvents are removed via distillation during the concentration step, they can be purified and reused in subsequent batches, leading to substantial cost savings in raw material expenditure over the lifecycle of the production campaign. Additionally, the elimination of complex transition metal catalysts, which are often required in cross-coupling alternatives, removes the need for expensive and technically demanding heavy metal scavenging steps, further simplifying the waste treatment protocol and reducing the environmental footprint of the manufacturing facility.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the high atom economy and the simplified workup procedure. By achieving yields of up to 95% in a single pot, the process minimizes the loss of valuable fluorinated starting materials, which are typically more expensive than their non-fluorinated analogs. The avoidance of chromatographic purification or multi-step protection/deprotection sequences drastically reduces the consumption of silica gel, eluents, and labor hours. Furthermore, the energy efficiency of operating at moderate temperatures (120°C-150°C) compared to high-pressure or cryogenic alternatives contributes to lower utility costs, making the overall cost of goods sold (COGS) highly competitive for a specialty electronic chemical.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions, which tolerate minor variations in feedstock quality without catastrophic failure of the batch. The use of standard glass-lined or stainless steel reactors means that production does not require exotic metallurgy, allowing for flexible manufacturing across multiple sites or contract manufacturing organizations (CMOs). The short reaction timeline relative to the complexity of the molecule ensures a faster turnaround time from order to delivery, enabling buyers to maintain leaner inventory levels while still meeting the rigorous demands of Just-In-Time manufacturing schedules prevalent in the electronics industry.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the process generates minimal hazardous waste. The primary byproduct is inorganic salt, which can be disposed of or treated using standard wastewater protocols, and the organic solvents are contained within a closed loop system. This alignment with green chemistry principles facilitates easier regulatory approval and permitting for new production lines. The scalability is proven by the linear translation of lab-scale parameters to pilot and commercial scales, as the heat transfer and mass transfer requirements are well-understood for this type of heterogeneous slurry reaction, ensuring that quality remains consistent regardless of batch size.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology for their specific applications, we have compiled answers to common inquiries regarding the synthesis and application of this fluorinated diamine. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for process engineers. Understanding these technical nuances is essential for integrating this monomer into existing polymer formulations or developing new material systems that require the unique balance of thermal stability and low dielectric constant offered by the hexafluoroisopropylidene moiety.
Q: What are the critical advantages of using this fluorinated diamine over non-fluorinated analogs?
A: The incorporation of the hexafluoroisopropylidene group significantly lowers the dielectric constant and dielectric loss of the resulting polymers, while also improving solubility and optical transparency compared to traditional aromatic diamines.
Q: How does the azeotropic solvent system improve reaction yield?
A: The use of toluene as an entrainer in combination with a polar aprotic solvent like DMF allows for the continuous removal of water generated during the phenol-halide substitution, shifting the equilibrium towards product formation and achieving yields up to 95%.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process is designed for industrial scalability, utilizing a one-pot reaction strategy with easily recoverable solvents and simple hot filtration steps to remove inorganic byproducts, minimizing waste and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2-Bis[3-Amino-4-(4-Cyanophenoxy)Phenyl]Hexafluoropropane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced electronic materials depends on a partner who can bridge the gap between laboratory innovation and industrial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN100368389C can be reliably replicated at the tonnage scale required by global supply chains. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against critical parameters such as melting point, HPLC purity, and residual solvent content. Our commitment to quality assurance guarantees that the fluorinated diamines supplied meet the exacting standards necessary for high-performance polyimide and liquid crystal applications.
We invite R&D directors and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and formulation needs. By partnering with our technical procurement team, you can access specific COA data and comprehensive route feasibility assessments that will help you optimize your material sourcing strategy. Whether you are looking to qualify a second source for an existing program or develop a new proprietary resin system, our expertise in fluorine chemistry and process optimization positions us as the ideal strategic partner for your long-term growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
