Advanced Synthesis of E-Beta-Sulfone Methylene Thiirane for Commercial Scale-Up
The landscape of fine chemical manufacturing is continually evolving, driven by the demand for more efficient and selective synthetic routes for complex heterocyclic scaffolds. Patent CN100586943C introduces a groundbreaking methodology for the synthesis of E-beta-sulfone methylene thiirane, a critical structural motif found in numerous bioactive compounds and enzyme inhibitors. This innovation addresses long-standing challenges in organic synthesis by providing a pathway that combines operational simplicity with exceptional stereochemical control. For R&D directors and procurement specialists alike, understanding the nuances of this technology is essential for optimizing supply chains and reducing the cost of goods sold in pharmaceutical intermediate manufacturing. The ability to access these high-value intermediates through a robust, scalable process represents a significant strategic advantage in the competitive global market for specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of thiirane rings with precise stereochemistry has been a formidable challenge for synthetic chemists, often requiring multi-step sequences that suffer from poor atom economy and low overall yields. Traditional approaches frequently rely on harsh reaction conditions, such as extreme temperatures or the use of hazardous reagents, which complicate safety protocols and increase waste disposal costs significantly. Furthermore, many existing methods struggle to control regioselectivity, leading to complex mixtures of isomers that are difficult and expensive to separate on a commercial scale. These inefficiencies not only extend lead times for high-purity pharmaceutical intermediates but also introduce variability that can compromise the quality of the final active pharmaceutical ingredient. The reliance on transition metal catalysts in some conventional routes further exacerbates cost issues due to the need for rigorous metal removal steps to meet regulatory standards.
The Novel Approach
In stark contrast to these legacy techniques, the method disclosed in CN100586943C utilizes a direct electrophilic reaction between allenyl sulfones and bromine to construct the thiirane core with remarkable efficiency. This novel approach operates under mild conditions, typically at room temperature, which drastically reduces energy consumption and simplifies reactor requirements for commercial scale-up of complex polymer additives or pharmaceutical precursors. The reaction demonstrates high regioselectivity and stereoselectivity, ensuring that the desired E-isomer is produced predominantly, thereby minimizing the need for costly downstream purification processes. By enabling the simultaneous introduction of multiple substituents with precise control, this methodology offers unparalleled flexibility for generating diverse libraries of analogs, accelerating the drug discovery pipeline while maintaining a lean manufacturing footprint.
Mechanistic Insights into Electrophilic Bromination and Cyclization
The core of this technological breakthrough lies in the unique mechanistic pathway where the electron-rich pi-system of the allenyl sulfone undergoes electrophilic attack by molecular bromine. This initial interaction generates a reactive bromonium ion intermediate, which is strategically positioned to facilitate an intramolecular nucleophilic attack by the adjacent sulfur atom. The geometric constraints imposed by the sulfone group play a pivotal role in directing the stereochemical outcome of the reaction, effectively locking the conformation to favor the formation of the E-beta-sulfone methylene configuration. Understanding this mechanistic nuance is crucial for process chemists aiming to replicate these results, as it highlights the importance of substrate electronics and steric factors in achieving optimal conversion rates and selectivity profiles.
Following the cyclization event, the reaction mixture is subjected to a straightforward workup involving water and saturated sodium thiosulfate solution, which serves to quench excess bromine and stabilize the product. This step is critical for impurity control, as it ensures the complete removal of oxidative byproducts that could otherwise degrade the stability of the final thiirane derivative. The subsequent purification via flash column chromatography allows for the isolation of the target compound with high purity, meeting the stringent specifications required for reliable agrochemical intermediate supplier standards. The robustness of this mechanism against varying substituent patterns on the aryl and alkyl groups further underscores its versatility, making it a powerful tool for the synthesis of a wide array of functionalized thiiranes.
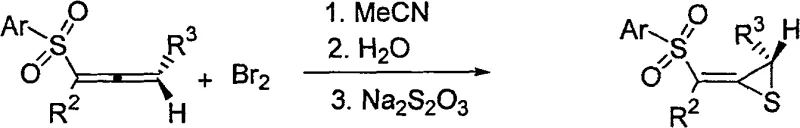
How to Synthesize E-Beta-Sulfone Methylene Thiirane Efficiently
To implement this synthesis effectively, one must adhere to the specific protocol outlined in the patent data, which emphasizes the precise stoichiometric ratios and solvent choices necessary for success. The process begins with the dissolution of the alpha-allenyl sulfone starting material in acetonitrile, followed by the controlled addition of a bromine solution to initiate the cyclization. Detailed standardized synthesis steps are provided below to guide process engineers in replicating this high-yielding transformation safely and consistently.
- Dissolve alpha-allenyl sulfone in acetonitrile at room temperature and add bromine solution dropwise.
- Stir the reaction mixture for 15-30 minutes to ensure complete electrophilic addition and cyclization.
- Quench with water and sodium thiosulfate, then extract and purify via flash column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits that directly impact the bottom line and operational resilience of chemical manufacturing enterprises. The elimination of complex catalytic systems and the use of commodity reagents like bromine and acetonitrile significantly lower the raw material costs associated with production. Moreover, the mild reaction conditions reduce the energy load on manufacturing facilities, contributing to a more sustainable and cost-effective operation that aligns with modern environmental compliance standards. These factors collectively enhance the economic viability of producing high-purity OLED material precursors or pharmaceutical building blocks using this technology.
- Cost Reduction in Manufacturing: The streamlined nature of this process eliminates the need for expensive transition metal catalysts and the associated purification steps required to remove trace metal residues. This reduction in processing complexity translates directly into lower operational expenditures, as fewer unit operations are required to achieve the final product specification. Additionally, the high yield range of 55-84% reported in the patent examples indicates efficient material utilization, minimizing waste generation and maximizing the return on investment for every kilogram of starting material processed.
- Enhanced Supply Chain Reliability: By relying on readily available and stable reagents such as bromine and common organic solvents, this method mitigates the risk of supply chain disruptions often associated with specialized or scarce chemicals. The robustness of the reaction across various substrate scopes ensures consistent output quality, which is vital for maintaining uninterrupted production schedules for downstream customers. This reliability fosters stronger partnerships between suppliers and manufacturers, ensuring that critical intermediates are delivered on time to support continuous pharmaceutical or agrochemical production lines.
- Scalability and Environmental Compliance: The simplicity of the workup procedure, involving basic extraction and washing steps, facilitates easy scale-up from laboratory benchtop to industrial reactor volumes without significant process redesign. Furthermore, the absence of heavy metals and the use of manageable solvents simplify waste treatment protocols, helping companies meet increasingly stringent environmental regulations. This alignment with green chemistry principles not only reduces compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and practical application scenarios. These insights are designed to clarify the operational parameters and potential advantages for stakeholders considering the adoption of this technology for their specific manufacturing needs.
Q: What is the stereoselectivity of this synthesis method?
A: The method described in CN100586943C achieves high regioselectivity and stereoselectivity, specifically yielding the E-isomer of beta-sulfone methylene thiirane exclusively.
Q: What are the typical reaction conditions required?
A: The process operates under mild conditions, specifically at room temperature using acetonitrile as the solvent, eliminating the need for cryogenic cooling or high-pressure equipment.
Q: Is this method suitable for large-scale production?
A: Yes, the simplicity of the workup procedure involving standard extraction and chromatography, combined with readily available reagents, makes it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable E-Beta-Sulfone Methylene Thiirane Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate complex patent methodologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of E-beta-sulfone methylene thiirane meets the highest industry standards for pharmaceutical and agrochemical applications.
We invite you to collaborate with us to optimize your supply chain and leverage the cost-saving potential of this advanced synthetic route. Please contact our technical procurement team to request a Customized Cost-Saving Analysis, where we can provide specific COA data and route feasibility assessments tailored to your project requirements. Let us help you engineer a more efficient and profitable manufacturing process today.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
