Scalable Electrochemical Production of High-Purity Carotenoid Precursors for Global Supply Chains
Scalable Electrochemical Production of High-Purity Carotenoid Precursors for Global Supply Chains
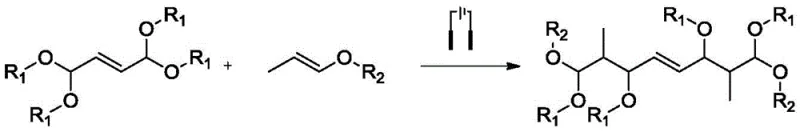
The landscape of fine chemical manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies. A pivotal advancement in this domain is documented in Chinese patent CN113604824B, which discloses a novel electrochemical process for the preparation of 1,8-dialkoxy-1,3,6,8-tetraalkoxy-2,7-dimethyl-4-octene. This compound serves as a critical precursor for C10 dialdehydes, which are indispensable building blocks in the total synthesis of carotenoids and Vitamin A derivatives. For R&D directors and supply chain leaders in the pharmaceutical and nutraceutical sectors, this technology represents a paradigm shift away from hazardous traditional chemistry towards a sustainable, metal-free electrochemical platform that promises enhanced purity profiles and streamlined operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key carotenoid intermediates has relied heavily on methodologies that pose substantial challenges for modern industrial compliance and cost efficiency. Prior art, such as the methods disclosed in CN100460378C, typically employs Lewis acid catalysts to facilitate the condensation of enol ethers. While chemically feasible, these traditional routes are plagued by severe drawbacks including significant equipment corrosion due to acidic conditions and complex post-treatment procedures required to remove metal residues. Furthermore, alternative pathways involving strong oxidants and acids, as seen in CN103172504B, impose rigorous safety requirements on production facilities and often result in lower overall yields due to uncontrolled side reactions and over-oxidation issues. These legacy processes generate substantial chemical waste, complicating environmental compliance and inflating the cost of goods sold through expensive waste disposal and purification protocols.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology outlined in CN113604824B introduces a sophisticated yet operationally simple electrochemical strategy. This innovative route utilizes indirect electrolysis mediated by iodine salts to drive the coupling of 1,1,4,4-tetraalkoxy-2-butene with propenyl alkyl ethers. By leveraging electricity as the primary reagent, the process completely circumvents the need for stoichiometric amounts of hazardous oxidants or corrosive Lewis acids. The reaction proceeds under remarkably mild conditions, typically between 20°C and 50°C, utilizing cost-effective graphite electrodes and a benign solvent system comprising ethyl acetate and water. This fundamental shift in reaction engineering not only mitigates safety risks but also drastically simplifies the downstream processing, allowing for the isolation of high-purity products through straightforward phase separation and solvent removal.
Mechanistic Insights into Iodine-Mediated Electrochemical Coupling
The core of this technological breakthrough lies in the elegant mechanism of indirect electrochemical oxidation. Unlike direct electrolysis which can lead to electrode passivation or non-selective substrate oxidation, this process employs iodide ions as a redox mediator. At the anode surface, iodide ions are selectively oxidized to generate reactive iodine radicals. These radicals act as hydrogen atom abstractors, specifically targeting the acetal carbon of the 1,1,4,4-tetraalkoxy-2-butene substrate to generate a stabilized carbon-centered radical intermediate. This selective activation is crucial for maintaining high chemoselectivity and preventing the degradation of sensitive functional groups often present in complex fragrance or pharma intermediates.
Following the initial radical generation, the mechanism proceeds through a controlled coupling sequence. The generated acetal radical undergoes fragmentation to form a hemiacetal radical and an alkoxide species. Subsequently, this reactive intermediate couples with the propenyl alkyl ether, followed by combination with methoxy radicals generated in the electrolytic medium. This cascade results in the formation of the desired 1,8-dialkoxy-1,3,6,8-tetraalkoxy-2,7-dimethyl-4-octene skeleton with exceptional regioselectivity. The use of supporting electrolytes such as tetraethylammonium hexafluorophosphate ensures efficient conductivity without interfering with the radical pathway, thereby minimizing the formation of polymeric byproducts and ensuring a clean impurity profile that is highly desirable for GMP manufacturing environments.
How to Synthesize 1,8-dialkoxy-1,3,6,8-tetraalkoxy-2,7-dimethyl-4-octene Efficiently
Implementing this electrochemical protocol requires precise control over reaction parameters to maximize the benefits of the iodine-mediated pathway. The process begins with the preparation of a mixed solvent system, preferably ethyl acetate and water, which balances the solubility of organic substrates with the ionic conductivity required for electrolysis. The molar ratio of the propenyl alkyl ether to the tetraalkoxy butene is carefully maintained, typically between 2.05:1 and 2.25:1, to drive the reaction to completion while minimizing excess reagent waste. Detailed standard operating procedures regarding electrode spacing, current density optimization, and workup protocols are essential for reproducibility.
- Prepare the electrolytic cell with graphite electrodes and add the solvent mixture of ethyl acetate and water.
- Introduce the iodine salt mediator, supporting electrolyte, 1,1,4,4-tetraalkoxy-2-butene, and propenyl alkyl ether into the reaction vessel.
- Apply constant current electrolysis at mild temperatures (20-50°C) until conversion is complete, then separate the organic phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of transition metal catalysts removes a significant bottleneck in the supply chain, as it negates the need for sourcing expensive palladium or platinum-based reagents and the subsequent specialized scavenging resins required to meet strict residual metal specifications. This simplification of the bill of materials directly translates to a more resilient supply chain that is less vulnerable to fluctuations in the precious metals market. Furthermore, the operational simplicity of the process reduces the dependency on highly specialized reactor infrastructure, allowing for more flexible manufacturing scheduling.
- Cost Reduction in Manufacturing: The economic impact of switching to this metal-free electrochemical process is profound. By replacing costly Lewis acids and oxidants with inexpensive iodine salts and electricity, the direct material costs are significantly lowered. Additionally, the use of durable graphite electrodes instead of consumable metal catalysts reduces the recurring cost of reagents. The simplified workup procedure, which avoids complex neutralization and filtration steps associated with acid catalysis, leads to substantial reductions in labor hours and utility consumption, thereby driving down the overall cost of production per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply security by utilizing commodity chemicals that are readily available in the global market. Solvents like ethyl acetate and electrolytes like potassium iodide are produced at massive scales for various industries, ensuring consistent availability and price stability. The mild reaction conditions also reduce the risk of unplanned shutdowns due to thermal runaways or equipment failures, ensuring a steady and predictable output of critical carotenoid intermediates to meet downstream demand for vitamins and flavors.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this process aligns perfectly with modern green chemistry principles. The absence of heavy metal waste streams simplifies effluent treatment and reduces the environmental footprint of the manufacturing site. The use of water as a co-solvent further demonstrates a commitment to sustainability. These factors facilitate easier regulatory approvals and reduce the long-term liability associated with hazardous waste disposal, making the commercial scale-up of complex pharmaceutical intermediates both environmentally responsible and economically viable.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on how this method resolves historical pain points in intermediate synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: Does this electrochemical process require expensive transition metal catalysts?
A: No, the process utilizes inexpensive iodine salts as mediators and graphite electrodes, completely eliminating the need for costly transition metal catalysts and the associated purification steps.
Q: How does this method improve upon traditional Lewis acid catalyzed routes?
A: Unlike Lewis acid methods which suffer from equipment corrosion and difficult post-treatment, this electrochemical approach operates under mild conditions with simple aqueous workup and higher selectivity.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the use of robust graphite electrodes, mild reaction temperatures, and common solvents like ethyl acetate makes the process highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,8-dialkoxy-1,3,6,8-tetraalkoxy-2,7-dimethyl-4-octene Supplier
As the global demand for high-quality carotenoids and Vitamin A precursors continues to rise, securing a reliable supply of key intermediates is paramount for maintaining competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry evolution, leveraging advanced electrochemical technologies to deliver superior chemical solutions. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of 1,8-dialkoxy-1,3,6,8-tetraalkoxy-2,7-dimethyl-4-octene meets the highest international standards for pharmaceutical and nutraceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our innovative electrochemical capabilities can drive value for your organization.
