Advanced Synthetic Route for Diamide Compounds: Enhancing Rare Earth Extraction Efficiency and Safety
The global demand for high-purity rare earth elements has intensified the search for more efficient and environmentally benign separation technologies. Patent CN102020579A introduces a groundbreaking synthetic methodology for diamide compounds, specifically designed as superior extractants for rare earth separation. This innovation addresses the critical bottlenecks in hydrometallurgy by replacing hazardous acid chloride intermediates with a stable ester-based pathway. By leveraging hydrogen chloride gas catalysis for esterification followed by direct amidation, this process significantly mitigates operational risks associated with traditional methods. For R&D directors and procurement specialists, this patent represents a pivotal shift towards sustainable chemical manufacturing, offering a robust framework for producing high-performance ligands that enhance selectivity and loading capacity in solvent extraction processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-oxoglutaramide extractants has relied heavily on the activation of diglycolic acid into its corresponding acid chloride using thionyl chloride. This conventional route, while chemically valid, imposes severe constraints on industrial scalability and safety protocols. The preparation of diglycolyl chloride requires rigorously anhydrous conditions, necessitating extensive drying of all reagents and apparatus to prevent hydrolysis. Furthermore, the reaction generates substantial quantities of corrosive sulfur dioxide and hydrogen chloride gases, mandating sophisticated scrubbing systems and increasing capital expenditure. The resulting acid chloride is inherently unstable, prone to rapid degradation upon exposure to atmospheric moisture, which forces a 'just-in-time' synthesis strategy that complicates inventory management and disrupts continuous production flows.
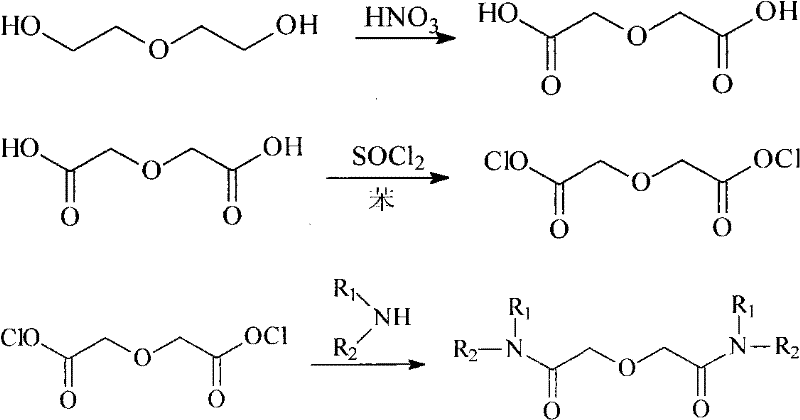
The Novel Approach
In stark contrast, the novel approach detailed in the patent circumvents these pitfalls by utilizing dimethyl diglycolate as a stable, isolable intermediate. This method employs hydrogen chloride gas to catalyze the esterification of diglycolic acid with methanol under mild conditions, effectively bypassing the need for thionyl chloride entirely. The resulting diester is chemically robust, allowing for long-term storage and decoupling the esterification step from the subsequent amidation reaction. This modularity enhances process flexibility, enabling manufacturers to stockpile the intermediate and respond rapidly to market demands without the logistical burden of handling hazardous acid chlorides. The elimination of sulfur-based byproducts also drastically reduces the environmental footprint, aligning with modern green chemistry principles and regulatory compliance standards.
Mechanistic Insights into HCl-Catalyzed Esterification and Amidation
The core of this technological advancement lies in the efficient protonation of the carbonyl oxygen by hydrogen chloride gas, which activates the carboxylic acid group of diglycolic acid towards nucleophilic attack by methanol. Unlike sulfuric acid catalysis, which can lead to complex side reactions and difficult workups, HCl gas offers a cleaner catalytic cycle that proceeds effectively at room temperature. This mild condition preserves the integrity of the ether linkage in the diglycolic backbone, preventing potential cleavage or degradation that might occur under more vigorous acidic conditions. The mechanism ensures high atom economy, as the only byproduct is water, which is easily removed during the workup phase, thereby simplifying the purification process and maximizing the yield of the desired dimethyl diglycolate intermediate.
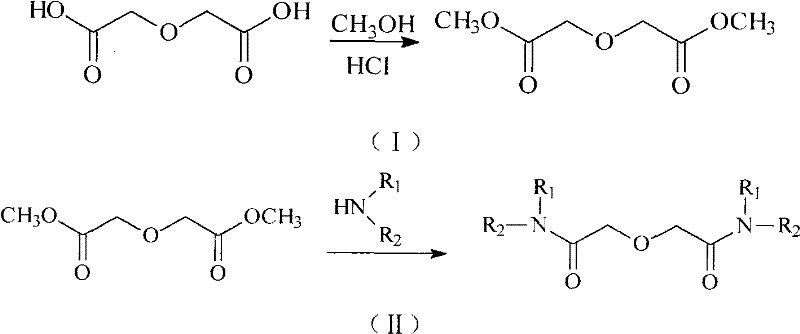
Following esterification, the transformation into the final diamide involves a nucleophilic acyl substitution where the amine attacks the ester carbonyl. This step is driven thermodynamically by the removal of methanol, often achieved through distillation at elevated temperatures around 160°C. The patent highlights that the stability of the ester intermediate allows for precise stoichiometric control, minimizing the formation of mono-substituted impurities which are detrimental to extraction performance. Furthermore, the purification protocol described, involving specific acid-base washes and potentially a nitric acid complexation step for ultra-high purity, ensures that the final product meets the stringent specifications required for separating chemically similar rare earth ions. This level of impurity control is critical for achieving high separation factors in industrial cascade operations.
How to Synthesize Diamide Compound Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable roadmap for producing high-purity diamide extractants suitable for commercial application. The process begins with the oxidation of diethylene glycol to diglycolic acid, followed by the key HCl-catalyzed esterification to form the stable dimethyl ester. The final step involves heating the ester with the desired amine to effect amidation. This sequence is designed to minimize hazard exposure while maximizing throughput. For detailed operational parameters, including specific molar ratios, temperature profiles, and purification techniques, please refer to the standardized synthesis guide below which encapsulates the critical process controls identified in the patent documentation.
- Preparation of Diglycolic Acid via nitric acid oxidation of diethylene glycol, ensuring strict temperature control between 65-70°C.
- HCl-catalyzed esterification of diglycolic acid with methanol at room temperature to form stable dimethyl diglycolate.
- Direct amidation of dimethyl diglycolate with amines (e.g., di-n-butylamine) at elevated temperatures (160°C) to yield the final diamide extractant.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the transition to this ester-based synthesis route offers profound advantages in terms of risk mitigation and cost structure optimization. By eliminating the requirement for thionyl chloride, a regulated and hazardous reagent, the process reduces the complexity of raw material sourcing and storage. The stability of the dimethyl diglycolate intermediate acts as a buffer in the production schedule, allowing for batch decoupling which smooths out production peaks and valleys. This flexibility is invaluable for maintaining consistent supply to downstream rare earth processing facilities, ensuring that extraction campaigns are not delayed by upstream synthesis bottlenecks or reagent shortages.
- Cost Reduction in Manufacturing: The elimination of the acid chloride step removes the need for expensive drying agents, specialized anhydrous solvents, and corrosion-resistant equipment required to handle thionyl chloride and its byproducts. The ability to perform esterification at room temperature significantly lowers energy consumption compared to refluxing conditions often needed for acid activation. Furthermore, the simplified workup procedure reduces solvent usage and waste disposal costs, leading to a leaner overall cost of goods sold without compromising on the quality of the final extractant.
- Enhanced Supply Chain Reliability: The robust nature of the dimethyl diglycolate intermediate means it can be produced in large batches and stored for extended periods without degradation. This capability transforms the supply chain from a fragile, just-in-time model to a resilient, inventory-backed system. Procurement managers can secure raw materials during favorable market conditions and manufacture the intermediate in advance, ensuring uninterrupted availability of the final diamide product even during fluctuations in amine supply or seasonal production constraints.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste, specifically avoiding the discharge of sulfur dioxide gas, which simplifies environmental permitting and reduces the burden on waste treatment facilities. The absence of highly reactive acid chlorides makes the process inherently safer to scale up from pilot plant to commercial tonnage, reducing the risk of runaway reactions. This alignment with green chemistry principles not only future-proofs the manufacturing site against tightening regulations but also enhances the corporate sustainability profile for clients seeking responsible sourcing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this synthetic route. They are derived from the specific challenges and solutions presented in the patent literature, focusing on the practical aspects of scaling this chemistry for industrial rare earth separation applications. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this methodology for their specific extraction workflows.
Q: Why is the new ester-based synthesis method superior to the traditional acid chloride route?
A: The new method eliminates the need for thionyl chloride, thereby avoiding the generation of hazardous sulfur dioxide and hydrogen chloride gases. Additionally, the intermediate dimethyl diglycolate is stable and can be stored long-term, unlike the highly reactive and moisture-sensitive acid chloride.
Q: What are the critical reaction conditions for the esterification step?
A: The esterification utilizes hydrogen chloride gas as a catalyst at room temperature. The reaction typically proceeds for about 10 hours, monitored by TLC, ensuring high conversion without the need for harsh heating or complex drying agents required in acid chloride formation.
Q: How is product purity ensured in this synthetic pathway?
A: Purity is maintained through rigorous washing steps, including acid and base washes to remove unreacted amines and byproducts. For ultra-high purity applications, a specialized purification involving nitric acid complexation and phase separation can be employed to remove monosubstituted impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diamide Compound Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing complex organic syntheses, such as the HCl-catalyzed esterification and amidation routes described in patent CN102020579A, to meet stringent purity specifications. We operate rigorous QC labs equipped with advanced analytical instrumentation to ensure every batch of diamide extractant delivers consistent performance in rare earth separation processes, guaranteeing the high selectivity and loading capacity your operations require.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive efficiency and reliability in your rare earth extraction projects.
