Advanced Manufacturing of High-Purity Cefoxitin Sodium for Global Pharmaceutical Supply Chains
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the persistent challenge of bacterial resistance and the critical need for high-purity active pharmaceutical ingredients (APIs). Patent CN101941983B introduces a significant technological advancement in the preparation of Cefoxitin Sodium, a second-generation cephalosporin renowned for its stability against beta-lactamases. This innovation addresses the longstanding industry pain point of impurity control during the salt formation stage. By shifting from traditional direct crystallization to a sophisticated resin adsorption protocol prior to salification, the method ensures the removal of trace organic contaminants and polymeric byproducts that often compromise clinical safety. For R&D directors and procurement specialists seeking a reliable cefoxitin sodium supplier, understanding this mechanistic shift is vital for securing a supply chain capable of delivering consistent, high-quality material.
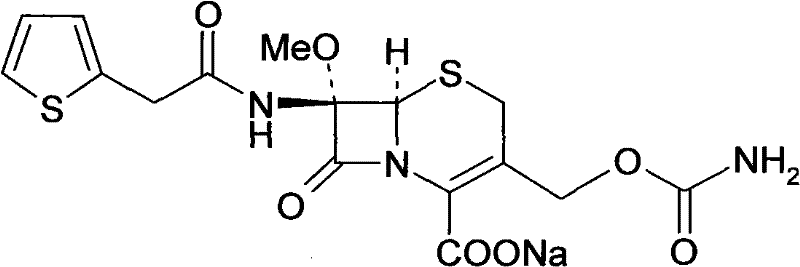
The structural integrity of Cefoxitin Sodium, as depicted above, relies on the precise stereochemistry of the cephem nucleus and the stability of the carbamate side chain. The patented process safeguards these sensitive functional groups while rigorously eliminating impurities. This approach not only elevates the chromatographic purity to exceptional levels, consistently exceeding 99.5% in experimental trials, but also streamlines the downstream processing requirements. As global regulatory bodies tighten specifications for antibiotic impurities, adopting such refined purification technologies becomes a strategic imperative for maintaining market access and ensuring patient safety in the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for cephalosporin sodium salts often rely heavily on direct salification of the free acid followed by immediate crystallization or precipitation. While seemingly straightforward, these conventional methodologies frequently suffer from inadequate impurity clearance, particularly regarding high-molecular-weight polymers and residual organic solvents that co-crystallize with the product. In the context of Cefoxitin, which possesses a complex side chain susceptible to degradation, standard solvent-based purification may fail to distinguish between the target molecule and structurally similar byproducts generated during the upstream fermentation or semi-synthetic steps. Consequently, manufacturers face significant challenges in meeting stringent pharmacopoeial standards without resorting to multiple, yield-depleting recrystallization cycles. Furthermore, the reliance on specific solvent systems for purification can introduce variability in particle size and morphology, complicating downstream formulation processes and potentially affecting the bioavailability of the final dosage form.
The Novel Approach
The methodology outlined in Patent CN101941983B represents a paradigm shift by integrating a dedicated solid-phase purification step using gamma-alumina resin before the critical salt formation reaction. This novel approach decouples the purification of the free acid from the salification process, allowing for a more targeted removal of impurities that would otherwise be trapped in the crystal lattice of the sodium salt. By passing the dissolved cefoxitin acid through a resin column, the process leverages differential adsorption affinities to retain unwanted organic species while allowing the pure acid to elute. This pre-purification ensures that the subsequent reaction with sodium salts occurs in an environment of high chemical homogeneity. The result is a drastic reduction in the burden on the final crystallization step, leading to a product with superior color, clarity, and purity profiles. This strategy effectively mitigates the risk of batch-to-batch variability, a crucial factor for cost reduction in pharmaceutical intermediates manufacturing by minimizing rework and rejection rates.
Mechanistic Insights into Resin Adsorption and Salification
The core of this technological breakthrough lies in the physicochemical interaction between the cefoxitin acid solution and the gamma-alumina resin matrix. Gamma-alumina possesses a unique surface chemistry characterized by Lewis acid-base sites that can interact selectively with polar impurities and polymeric aggregates present in the crude acid solution. When the cefoxitin acid, dissolved in a polar aprotic solvent like ethyl acetate or methanol, traverses the resin bed, these impurities are adsorbed onto the alumina surface through mechanisms such as hydrogen bonding, dipole-dipole interactions, or van der Waals forces. Crucially, the cefoxitin acid molecule itself, due to its specific steric and electronic configuration, exhibits a lower affinity for the resin under the optimized flow conditions, allowing it to pass through with minimal retention. This selective filtration acts as a molecular sieve, effectively polishing the feed stream before it enters the reactor. The elimination of these precursors prevents them from participating in or interfering with the subsequent neutralization reaction, thereby preserving the integrity of the beta-lactam ring and the carbamate moiety.
Following purification, the salification mechanism proceeds via a classic acid-base neutralization but under highly controlled conditions that favor the formation of a stable crystalline lattice. The addition of sodium salts, such as sodium isooctanoate, introduces sodium ions that displace the acidic proton on the carboxylic acid group at the C-2 position of the cephem nucleus. The patent specifies a temperature range of -20°C to 20°C, with room temperature being sufficient for high yields. This mild thermal condition is essential for preventing the hydrolysis of the beta-lactam ring, a common degradation pathway for cephalosporins in solution. The slow addition of the sodium salt solution ensures a controlled supersaturation level, promoting the growth of well-defined crystals rather than amorphous precipitates which might occlude impurities. The resulting sodium salt precipitates out of the organic solvent system, driven by its lower solubility compared to the free acid and the sodium reagent. This precise control over nucleation and growth kinetics is what enables the process to achieve yields upwards of 94% while maintaining chromatographic purity levels near 99.6%, demonstrating a robust balance between efficiency and quality.
How to Synthesize Cefoxitin Sodium Efficiently
The implementation of this synthesis route requires careful attention to solvent quality and resin conditioning to maximize the adsorption capacity of the column. The process begins with the dissolution of the crude cefoxitin acid in a suitable organic solvent, typically ethyl acetate, followed by a decolorization step using activated carbon to remove bulk colored impurities. Once filtered, this clarified solution is loaded onto the gamma-alumina column, where the critical purification occurs. The eluate, now containing highly purified cefoxitin acid, is collected in a reaction vessel maintained under sterile conditions if intended for parenteral use. The detailed standardized synthesis steps, including specific flow rates, resin bed volumes, and stoichiometric ratios for the salification, are critical for reproducibility and are outlined in the technical guide below.
- Dissolve cefoxitin acid in ethyl acetate or methanol, decolorize with activated carbon, and purify the filtrate through a gamma-alumina resin column to remove organic impurities.
- Prepare a solution of sodium salt (such as sodium isooctanoate) in ethyl acetate and filter aseptically.
- Slowly add the sodium salt solution to the purified acid solution at room temperature, stir for 2 hours, then filter and vacuum dry to obtain the final crystalline powder.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this resin-based purification technology offers substantial strategic benefits beyond mere technical superiority. The simplification of the purification train reduces the overall processing time and equipment footprint required for production. By achieving high purity in a single pass through the resin column, manufacturers can eliminate multiple recrystallization steps that are both time-consuming and costly in terms of solvent consumption and energy usage. This streamlining directly translates to improved throughput and a more agile response to market demand fluctuations. Furthermore, the use of common, commercially available reagents such as ethyl acetate and sodium isooctanoate ensures that the supply chain remains resilient against raw material shortages. The robustness of the process, capable of operating effectively at ambient temperatures, also lowers the energy burden associated with cryogenic cooling or extensive heating, contributing to a more sustainable and cost-effective manufacturing profile.
- Cost Reduction in Manufacturing: The elimination of complex, multi-stage recrystallization protocols significantly lowers the operational expenditure associated with solvent recovery and waste disposal. By utilizing a resin column to capture impurities early in the process, the yield loss typically associated with aggressive purification attempts is minimized, leading to a higher overall mass balance. The ability to use cost-effective sodium salts like sodium carbonate or bicarbonate, in addition to sodium isooctanoate, provides flexibility in raw material sourcing, allowing procurement teams to optimize costs based on market availability without compromising the quality of the final API. This economic efficiency is further enhanced by the reduced need for specialized low-temperature equipment, as the reaction proceeds efficiently at room temperature.
- Enhanced Supply Chain Reliability: The reliance on standard chemical engineering unit operations, such as column chromatography and filtration, ensures that the process can be easily scaled and transferred between different manufacturing sites. The raw materials required, including the gamma-alumina resin and organic solvents, are commodity chemicals with stable global supply chains, reducing the risk of production stoppages due to ingredient scarcity. Additionally, the high purity achieved through this method reduces the likelihood of batch failures during quality control testing, ensuring a consistent and reliable flow of finished goods to customers. This reliability is paramount for maintaining long-term contracts with major pharmaceutical companies that require uninterrupted supply of critical antibiotics.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory bench scale to multi-ton commercial production without fundamental changes to the chemistry. The use of resin adsorption generates a solid waste stream (spent resin) that is easier to manage and dispose of compared to large volumes of mother liquor from repeated crystallizations. Moreover, the high efficiency of the reaction minimizes the generation of hazardous byproducts, aligning with increasingly stringent environmental regulations regarding pharmaceutical manufacturing emissions. The simplified workflow also reduces the potential for operator error, enhancing overall plant safety and compliance with Good Manufacturing Practices (GMP).
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cefoxitin Sodium using this advanced resin adsorption method. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing production lines or for procurement officers assessing the quality credentials of potential suppliers.
Q: What is the primary advantage of using gamma-alumina resin in this synthesis?
A: The use of gamma-alumina resin effectively adsorbs large amounts of organic and polymeric impurities that are difficult to remove via standard crystallization, significantly enhancing the chromatographic purity of the final cefoxitin sodium product to over 99.5%.
Q: What temperature range is required for the salification reaction?
A: The patent specifies a broad operational window between -20°C and 20°C, with exemplary embodiments successfully conducted at room temperature, simplifying thermal control requirements for large-scale production.
Q: Which sodium salts are compatible with this preparation method?
A: The process is versatile and supports the use of sodium isooctanoate, sodium carbonate, or sodium bicarbonate, allowing manufacturers to select reagents based on availability and cost efficiency without compromising product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefoxitin Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent theory to commercial reality requires deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of resin purification and salification is maintained at every scale. We are committed to delivering Cefoxitin Sodium that meets the most stringent purity specifications, leveraging our rigorous QC labs to verify every batch against international pharmacopoeial standards. Our facility is equipped to handle the specific solvent systems and resin handling requirements of this process, guaranteeing a supply of high-quality intermediates that support your drug development and manufacturing goals.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific application. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data from recent batches and comprehensive route feasibility assessments to demonstrate our capability as your trusted partner in the global pharmaceutical supply chain.
