Advanced Metal-Free Synthesis of Trans-4-Carboxycotinine for Commercial Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for synthesizing complex heterocyclic compounds that serve as critical building blocks for bioactive molecules. Patent CN109796436B introduces a groundbreaking method for preparing high-purity (+/-) -trans-4' -carboxycotinine, a valuable pyrrolidone derivative with significant potential in the development of quinolone antibacterial drugs, antihypertensive agents, and antitumor therapies. This technical disclosure represents a major shift away from traditional metal-catalyzed routes, offering a streamlined, two-step synthesis that leverages readily available industrial raw materials such as 3-pyridylaldehyde and succinic anhydride. By eliminating the reliance on expensive and potentially toxic transition metal catalysts, this innovation not only simplifies the purification workflow but also aligns perfectly with modern green chemistry principles and stringent regulatory requirements for pharmaceutical intermediates. The ability to achieve a total yield of over 60% with an HPLC purity exceeding 99% underscores the commercial viability of this approach for high-volume manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of five-membered nitrogen-containing heterocycles like cotinine derivatives has relied heavily on metal-catalyzed processes that introduce significant complexity and cost into the supply chain. Conventional literature reports often describe methods that require precise control of stereoselectivity using rare earth or precious metal catalysts, which not only drive up the raw material costs but also create substantial downstream purification challenges. The removal of trace metal residues to meet pharmaceutical grade specifications often necessitates additional processing steps such as chelation, specialized filtration, or extensive chromatography, all of which erode profit margins and extend production lead times. Furthermore, many traditional routes suffer from moderate yields and inconsistent purity profiles, making them unreliable for consistent commercial scale-up of complex pharmaceutical intermediates. The difficulty in separating the target product from reaction by-products and catalyst residues has long been a bottleneck, preventing many promising heterocyclic compounds from reaching full industrial exploitation in a cost-effective manner.
The Novel Approach
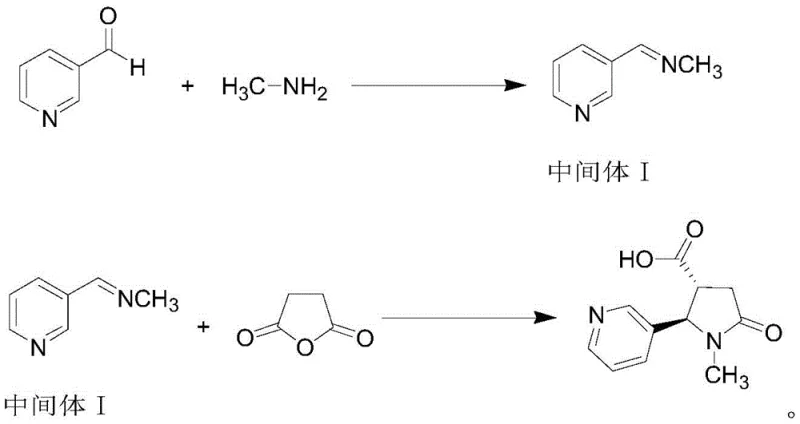
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a metal-free thermal cyclization strategy that fundamentally reengineers the synthesis pathway for efficiency and purity. By employing an eneimine intermediate generated in situ from 3-pyridylaldehyde and methylamine, the process bypasses the need for external catalytic activation, relying instead on controlled thermal energy to drive the reaction forward. This shift allows for the use of common, low-toxicity organic solvents like xylene and ethanol, which are easily recovered and recycled, further enhancing the environmental profile of the manufacturing process. The method demonstrates exceptional control over reaction conditions, maintaining mild temperatures in the initial step and utilizing standard reflux conditions for the cyclization, which reduces the energy burden on production facilities. This streamlined workflow not only accelerates the reaction kinetics but also ensures that the final crude product is of sufficient quality to undergo simple recrystallization, thereby drastically reducing the overall processing time and operational expenditure associated with producing high-purity trans-4-carboxycotinine.
Mechanistic Insights into Enamine-Mediated Cyclization
The core of this synthetic innovation lies in the formation and utilization of the eneimine intermediate, which serves as a highly reactive nucleophile in the subsequent cyclization step. In the first stage, the condensation of 3-pyridylaldehyde with methylamine in an ethanol solution at 25-30°C generates the eneimine intermediate I with high efficiency, avoiding the formation of stable by-products that often plague imine chemistry. This intermediate is not isolated but is directly concentrated and transferred into the second reaction vessel, minimizing material loss and exposure to atmospheric moisture which could degrade the reactive species. The subsequent reaction with succinic anhydride in xylene at 130-135°C facilitates a [3+2] cycloaddition-like process where the eneimine attacks the anhydride, leading to the closure of the pyrrolidone ring. This thermal activation method ensures that the reaction proceeds to completion without the need for sensitive catalysts, providing a robust mechanism that is less susceptible to variations in raw material quality or minor fluctuations in process parameters.
Impurity control is inherently built into this mechanistic pathway through the strategic use of pH-dependent extraction and selective crystallization. Following the cyclization, the reaction mixture is treated with sodium bicarbonate to adjust the pH to 8-9, which effectively solubilizes acidic by-products and unreacted anhydride derivatives into the aqueous phase while keeping the target neutral or basic organic species in the organic layer or as a precipitate. The subsequent acidification to pH 3-4 using phosphoric acid precipitates the target carboxycotinine as a white solid, leaving most organic impurities in the mother liquor or the preceding organic washes. This acid-base workup is far more scalable and cost-effective than column chromatography, allowing for the processing of hundreds of kilograms of material with minimal equipment footprint. Finally, recrystallization from ethanol at low temperatures (-5 to -10°C) further refines the crystal lattice, excluding remaining trace impurities and ensuring that the final product meets the stringent >99% HPLC purity specification required for pharmaceutical applications.
How to Synthesize Trans-4-Carboxycotinine Efficiently
Implementing this synthesis route in a commercial setting requires precise adherence to the thermal profiles and stoichiometric ratios defined in the patent to maximize yield and safety. The process begins with the careful preparation of the eneimine intermediate, where temperature control between 25-30°C is critical to prevent polymerization or degradation of the aldehyde starting material. Once the intermediate is formed and concentrated, it is immediately reacted with succinic anhydride in xylene, where the reflux temperature must be maintained steadily at 130-135°C for approximately 10 hours to ensure complete cyclization. The detailed standardized synthesis steps, including specific solvent volumes, stirring rates, and filtration protocols, are outlined in the technical guide below to ensure reproducibility across different manufacturing sites.
- React 3-pyridylaldehyde with methylamine ethanol solution at 25-30°C to form eneimine intermediate I.
- Reflux intermediate I with succinic anhydride in xylene at 130-135°C for cyclization.
- Purify via pH adjustment, solvent extraction, and ethanol recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this patent offers a compelling value proposition by addressing several critical pain points associated with the sourcing of complex heterocyclic intermediates. The elimination of transition metal catalysts removes a significant cost driver from the bill of materials, as precious metals are not only expensive to purchase but also costly to dispose of in compliance with environmental regulations. By relying on commodity chemicals like 3-pyridylaldehyde and succinic anhydride, the supply chain becomes more resilient to market fluctuations, as these raw materials are produced in high volumes by multiple global suppliers, ensuring continuous availability and competitive pricing. The simplified purification process, which avoids complex chromatography, translates directly into reduced manufacturing cycle times and lower utility consumption, allowing for faster turnaround on orders and improved cash flow for production projects.
- Cost Reduction in Manufacturing: The absence of expensive metal catalysts and the use of recyclable solvents like xylene and ethanol significantly lower the variable costs per kilogram of the final product. The ability to perform the reaction without specialized catalytic equipment reduces capital expenditure requirements, while the high yield of over 60% minimizes raw material waste, leading to substantial overall cost savings in pharmaceutical intermediate manufacturing. This economic efficiency makes the compound viable for a broader range of therapeutic applications where cost sensitivity is a key factor in drug development.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly simplified as the key starting materials are widely available industrial chemicals with stable supply lines, reducing the risk of production delays due to raw material shortages. The robustness of the metal-free process means that manufacturing is less susceptible to disruptions caused by the scarcity of specialized reagents, ensuring consistent delivery schedules for downstream pharmaceutical clients. This reliability is crucial for maintaining the continuity of supply for critical drug substances that depend on this intermediate for their synthesis.
- Scalability and Environmental Compliance: The process is designed for industrial scale-up, utilizing standard glass-lined or stainless steel reactors that are common in fine chemical plants, facilitating a smooth transition from pilot scale to commercial production. The use of low-toxicity solvents and the generation of manageable waste streams simplify environmental compliance and waste treatment processes, aligning with increasingly strict global sustainability standards. This scalability ensures that the supply can easily be ramped up to meet growing market demand without requiring significant process re-engineering or new facility construction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this high-purity intermediate, based on the specific data and claims within the patent documentation. Understanding these details is essential for R&D teams evaluating the feasibility of this route for their specific drug candidates and for procurement teams assessing the long-term viability of the supply source. The answers provided reflect the proven capabilities of the described method to deliver consistent quality and performance in a commercial setting.
Q: What are the purity levels achievable with this synthesis method?
A: The patented process consistently achieves HPLC purity levels exceeding 99% through a specialized recrystallization protocol using ethanol, eliminating the need for complex chromatographic separation.
Q: Does this method require expensive transition metal catalysts?
A: No, the process is entirely metal-free, utilizing 3-pyridylaldehyde and succinic anhydride under thermal conditions, which significantly reduces raw material costs and heavy metal contamination risks.
Q: Is the process suitable for large-scale industrial production?
A: Yes, the method uses common industrial solvents like xylene and ethanol and operates under manageable temperature ranges (25-30°C and 130-135°C), making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-Carboxycotinine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner for the supply of high-value pharmaceutical intermediates like trans-4-carboxycotinine. Our team of expert chemists has extensively analyzed this patent route and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to full industrial maturity. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, ensuring that every batch conforms to the highest standards of quality and consistency required by the global pharmaceutical industry. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this synthesis, guaranteeing a secure and compliant supply chain for your critical projects.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs through the adoption of this efficient synthetic route. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this metal-free process can improve your bottom line. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this high-purity intermediate into your drug development pipeline with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
