Optimizing Artemether Production via Advanced Mother Liquor Recycling Technology
The global demand for effective antimalarial therapies continues to drive innovation in the synthesis of key pharmaceutical intermediates, specifically artemether derivatives. Patent CN102351875B introduces a groundbreaking methodology for the recycling of mother liquor generated during the industrial production of artemether, addressing critical inefficiencies in current manufacturing workflows. Traditionally, the production of artemether from dihydroartemisinin yields a mixture of alpha and beta epimers, with the mother liquor containing significant amounts of unreacted or less active alpha-artemether and dihydroartemisinin that are often discarded or underutilized. This patented technology offers a robust solution by chemically converting these waste components back into the pharmacologically active beta-artemether, thereby enhancing the overall utilization rate of the precious artemisinin raw material. For R&D directors and process engineers, this represents a pivotal shift towards greener chemistry and improved atom economy in the synthesis of complex sesquiterpene lactones. By integrating this recycling loop, manufacturers can drastically reduce raw material costs while simultaneously minimizing environmental waste, aligning perfectly with modern sustainable manufacturing mandates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the recovery of valuable intermediates from artemether production mother liquors has been plagued by low efficiency and technical complexity. Prior art, such as the methods described in patent literature EP0330520, attempted to epimerize alpha-arteether to the beta form but achieved crystallization yields of merely 30% to 40%, which is economically unviable for large-scale operations. Other approaches, like those in CN1106011A, attempted to convert alpha-artemether back to dihydroartemisinin but often resulted in complex mixtures with high impurity profiles and questionable practical utility upon experimental repetition. The fundamental flaw in these conventional strategies lies in their inability to effectively manage the equilibrium between the alpha and beta anomers without degrading the sensitive peroxide bridge structure essential for antimalarial activity. Furthermore, direct extraction of mother liquor without prior concentration is inefficient due to the high solubility of artemether derivatives in the methanol-water matrix, leading to substantial product loss in the aqueous phase. These limitations create a bottleneck in the supply chain, forcing manufacturers to rely on fresh batches of expensive artemisinin to meet production targets, thereby inflating the cost of goods sold.
The Novel Approach
The innovative process outlined in CN102351875B overcomes these historical barriers through a sophisticated three-step protocol involving concentration, acid-catalyzed conversion, and selective crystallization. By first subjecting the mother liquor to vacuum concentration at controlled temperatures (30-50°C), the method effectively removes the bulk of the methanol, significantly reducing the solubility of the target compounds in the remaining aqueous phase and facilitating efficient extraction into low-polarity organic solvents. The core breakthrough lies in the subsequent conversion step, where an acid catalyst is employed to drive the epimerization of the inactive alpha-artemether and residual dihydroartemisinin into the desired beta-artemether. 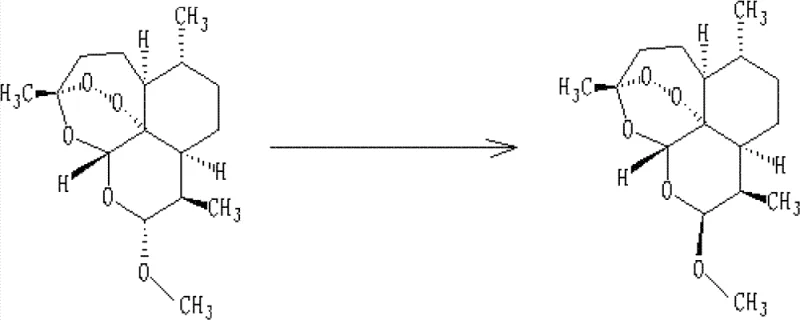 This chemical transformation allows for the recovery of value from what was previously considered waste, pushing the total yield of beta-artemether to commercially attractive levels. The process concludes with a precise neutralization and recrystallization sequence in a methanol-water system, which leverages the differential solubility of the isomers to isolate high-purity beta-artemether crystals. This approach not only simplifies the workflow compared to multi-step degradation and resynthesis routes but also ensures that the critical peroxide functionality remains intact throughout the recycling process.
This chemical transformation allows for the recovery of value from what was previously considered waste, pushing the total yield of beta-artemether to commercially attractive levels. The process concludes with a precise neutralization and recrystallization sequence in a methanol-water system, which leverages the differential solubility of the isomers to isolate high-purity beta-artemether crystals. This approach not only simplifies the workflow compared to multi-step degradation and resynthesis routes but also ensures that the critical peroxide functionality remains intact throughout the recycling process.
Mechanistic Insights into Acid-Catalyzed Epimerization
The chemical heart of this recycling technology is the acid-catalyzed equilibration of the hemiacetal center at the C-10 position of the artemether scaffold. Under acidic conditions, the methoxy group at the anomeric center becomes protonated, facilitating the formation of an oxocarbenium ion intermediate that is planar and achiral at the reaction center. This intermediate allows for the nucleophilic attack of methanol from either face of the ring, but thermodynamic control favors the formation of the beta-anomer due to the anomeric effect and steric factors inherent to the rigid sesquiterpene skeleton. The patent specifies a reaction temperature range of 10-40°C, with an optimal window of 22-32°C, which is critical for balancing the reaction kinetics against the thermal instability of the endoperoxide bridge. Operating outside this range, particularly at elevated temperatures above 40°C, risks the decomposition of the peroxide bond, leading to the formation of inactive degradation products and reducing the overall efficacy of the recycling loop. The choice of solvent system, typically involving low-polarity organics like n-heptane or toluene mixed with a small fraction of methanol, creates a microenvironment that stabilizes the transition state while preventing excessive solvation that might hinder crystallization later.
Impurity control is meticulously managed through the selection of catalyst and the precise monitoring of reaction endpoints via HPLC. The patent lists a variety of compatible acids, including hydrochloric acid, sulfuric acid, and Lewis acids like boron trifluoride etherate, providing flexibility to avoid specific counter-ion contaminants that might interfere with downstream processing. Crucially, the reaction is monitored until the ratio of alpha to beta isomers stabilizes, indicating that thermodynamic equilibrium has been reached, typically within 5 to 16 hours depending on the catalyst strength. Following the reaction, the immediate cooling of the mixture to below 30°C prior to neutralization is a vital safety and quality measure; it prevents exothermic spikes during the addition of base (such as sodium bicarbonate) which could otherwise trigger rapid peroxide decomposition. 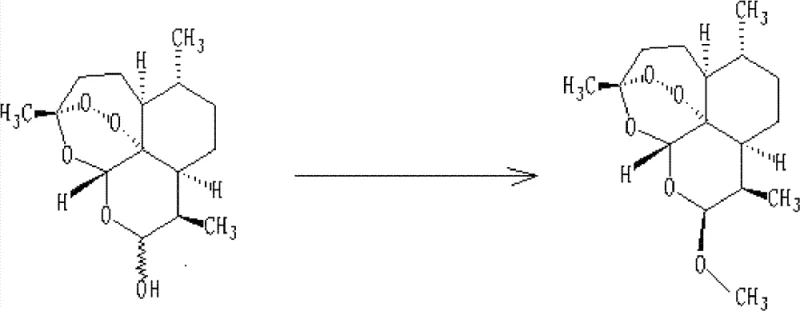 This careful thermal management ensures that the recycled material matches the quality specifications of virgin material, maintaining the integrity of the final API. The subsequent washing and drying steps are designed to remove residual acids and solvents, ensuring the crude product is ready for final refining without introducing new impurity vectors.
This careful thermal management ensures that the recycled material matches the quality specifications of virgin material, maintaining the integrity of the final API. The subsequent washing and drying steps are designed to remove residual acids and solvents, ensuring the crude product is ready for final refining without introducing new impurity vectors.
How to Synthesize Beta-Artemether Efficiently
The implementation of this recycling protocol requires strict adherence to the operational parameters defined in the patent to ensure safety and reproducibility. The process begins with the concentration of the mother liquor to remove methanol, followed by extraction into an organic phase where the actual chemical transformation occurs. Operators must carefully control the addition rate of the acid catalyst to prevent localized overheating, which is a known risk factor for peroxide stability. The final isolation step relies on the precise addition of water to the methanolic solution to induce crystallization, a technique that requires patience and precise temperature control to maximize crystal growth and purity. By following these standardized steps, production teams can reliably convert waste streams into high-value intermediates, transforming a cost center into a profit-generating asset within the facility.
- Concentrate the artemether mother liquor under vacuum at 30-50°C to remove methanol, then extract recyclable substances using a low-polarity organic solvent.
- Perform the conversion reaction by adding an acid catalyst and methanol to the organic layer at 10-40°C for 2-24 hours to epimerize alpha-artemether to beta-artemether.
- Neutralize the reaction mixture, concentrate the organic layer, and recrystallize the product in a methanol-water system to obtain high-purity beta-artemether.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this recycling technology offers profound strategic advantages beyond simple yield improvement. The primary economic driver is the drastic reduction in the consumption of fresh artemisinin, a natural product whose supply can be volatile and subject to agricultural fluctuations. By recovering and converting the alpha-isomer and dihydroartemisinin present in the mother liquor, the effective throughput of the plant increases without the need for proportional increases in raw material purchasing. This internal sourcing of intermediates acts as a hedge against market price volatility for artemisinin, providing a more predictable cost structure for the final artemether product. Furthermore, the elimination of complex waste treatment processes associated with discarding large volumes of organic-rich mother liquor translates into significant operational expenditure savings. The solvents used in the extraction, such as toluene or n-heptane, can be recovered and reused during the concentration steps, further lowering the variable costs associated with solvent procurement and disposal.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive disposal of chemical waste and reduces the dependency on fresh raw materials, leading to substantial cost savings in pharmaceutical intermediates manufacturing. By converting low-value alpha-isomers into high-value beta-products, the effective cost per kilogram of the active ingredient is significantly lowered without compromising quality standards. The ability to recycle solvents within the process loop further diminishes the operational overhead, making the production line leaner and more financially resilient against raw material price shocks.
- Enhanced Supply Chain Reliability: Implementing this recycling loop diversifies the source of the active intermediate, reducing the risk of supply disruptions caused by external factors affecting artemisinin harvests. The robustness of the acid-catalyzed conversion ensures a steady internal supply of beta-artemether, allowing for more accurate production planning and inventory management. This self-sufficiency strengthens the supply chain continuity, ensuring that downstream API synthesis is not halted due to shortages of key precursors, thereby enhancing the reliability of the manufacturer as a partner.
- Scalability and Environmental Compliance: The simplicity of the unit operations—concentration, extraction, reaction, and crystallization—means the process is highly scalable from pilot to commercial production without requiring specialized exotic equipment. The reduction in chemical waste generation aligns with stringent environmental regulations, reducing the burden on effluent treatment plants and minimizing the carbon footprint of the manufacturing site. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for securing contracts with major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this mother liquor recycling technology. These answers are derived directly from the experimental data and claims presented in the patent documentation to ensure accuracy and relevance for technical decision-makers. Understanding these nuances is essential for process engineers evaluating the feasibility of integrating this technology into existing production lines, as it highlights the balance between reaction efficiency and safety protocols required for handling peroxide-containing compounds.
Q: What is the primary advantage of this recycling method over conventional extraction?
A: Unlike conventional methods which often discard mother liquor or achieve low recovery rates (30-40%), this patented process utilizes acid-catalyzed conversion to transform inactive alpha-artemether and dihydroartemisinin into the pharmacologically active beta-artemether, significantly boosting overall yield.
Q: Which catalysts are suitable for the epimerization reaction?
A: The process supports a wide range of acid catalysts including concentrated hydrochloric acid, sulfuric acid, perchloric acid, and boron trifluoride etherate, allowing flexibility based on availability and cost considerations without compromising reaction efficiency.
Q: How does this process impact the purity of the final API intermediate?
A: By employing a controlled crystallization step in a methanol-water system at low temperatures (-5°C to 5°C), the method effectively separates the desired beta-isomer from impurities, ensuring the production of relatively high-purity beta-artemether suitable for further refining.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Artemether Supplier
At NINGBO INNO PHARMCHEM, we recognize that the future of fine chemical manufacturing lies in the optimization of existing pathways rather than solely the discovery of new ones. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex recycling loops like the one described in CN102351875B can be implemented safely and efficiently. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of recycled artemether meets the highest international standards for pharmaceutical intermediates. Our commitment to process excellence means that we can offer high-purity artemether with a supply security that is bolstered by our ability to maximize raw material utilization through advanced recycling technologies.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible economic benefits of our optimized manufacturing processes. Let us help you transform your supply chain challenges into competitive advantages through superior chemical engineering and unwavering commitment to quality.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
