Scalable Synthesis of 2-Trifluoromethyl Furan Compounds for Advanced Agrochemical Applications
The development of efficient synthetic routes for fluorinated heterocycles remains a critical priority in modern organic chemistry, particularly for the agrochemical and pharmaceutical sectors. Patent CN110272400B introduces a robust and versatile methodology for the synthesis of 2-trifluoromethyl substituted furan compounds and their derivatives. This technology leverages a base-promoted tandem reaction between benzoylacetonitrile derivatives and 3-bromo-1,1,1-trifluoroacetone, offering a streamlined alternative to traditional multi-step protocols. The significance of introducing trifluoromethyl groups into heterocyclic scaffolds cannot be overstated, as this modification often enhances lipophilicity, metabolic stability, and bioavailability. The disclosed method achieves this through a one-pot synthesis that proceeds under relatively mild conditions, utilizing common solvents like 1,4-dioxane and inorganic bases. This approach not only simplifies the operational complexity but also improves the overall atom economy of the process. For R&D teams seeking reliable agrochemical intermediate supplier partnerships, this patent represents a significant advancement in accessing high-value fluorinated building blocks efficiently.
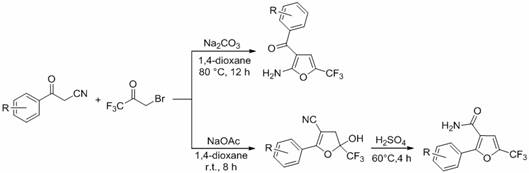
The limitations of conventional methods for constructing trifluoromethyl-substituted furan rings often stem from the requirement for pre-functionalized starting materials or the use of hazardous fluorinating reagents that demand strict safety controls. Traditional pathways may involve separate steps for ring closure and subsequent trifluoromethylation, leading to lower overall yields and increased purification burdens. In contrast, the novel approach detailed in this patent utilizes a direct cyclization strategy where the trifluoromethyl group is introduced simultaneously with the formation of the furan ring. By employing 3-bromo-1,1,1-trifluoroacetone as both the carbon source and the fluorine source, the reaction bypasses the need for external fluorinating agents. This integration of steps into a single operational sequence drastically reduces the time and resources required for production. Furthermore, the tolerance of the reaction system towards various functional groups on the benzoylacetonitrile substrate allows for the rapid generation of diverse compound libraries, which is essential for lead optimization in drug discovery and agrochemical development.
Mechanistic Insights into Base-Promoted Tandem Cyclization
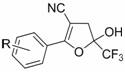
The mechanistic pathway of this transformation involves a sophisticated sequence of nucleophilic attacks and elimination reactions driven by the basic environment. Initially, the base deprotonates the active methylene group of the benzoylacetonitrile derivative, generating a nucleophilic enolate species. This enolate then attacks the electrophilic carbon of the 3-bromo-1,1,1-trifluoroacetone, displacing the bromide ion and forming a new carbon-carbon bond. Subsequent intramolecular cyclization occurs through the attack of the carbonyl oxygen or the nitrile nitrogen, depending on the specific tautomeric state, leading to the formation of a dihydrofuran intermediate. This intermediate, specifically the 5-hydroxy-2-phenyl-5-(trifluoromethyl)-4,5-dihydrofuran-3-carbonitrile derivative, is a key structural motif that can be isolated or further processed. The final aromatization to the furan ring is achieved through dehydration, often facilitated by the reaction conditions or subsequent acid treatment. Understanding this mechanism is vital for process chemists aiming to optimize reaction parameters such as temperature and base strength to maximize yield and minimize byproduct formation.
Impurity control is another critical aspect of this synthesis, particularly given the potential for side reactions involving the highly reactive trifluoroacetone moiety. The patent data indicates that the choice of base and solvent plays a pivotal role in suppressing unwanted polymerization or decomposition pathways. For instance, using sodium carbonate in 1,4-dioxane at 80°C provides a balanced environment that promotes the desired cyclization while maintaining the integrity of the sensitive nitrile and trifluoromethyl groups. The subsequent hydrolysis of the nitrile group to an amide, if desired, is performed under controlled acidic conditions using concentrated sulfuric acid at moderate temperatures (40-60°C). This step-wise control ensures that the final 2-trifluoromethyl substituted furan derivatives, such as the amides shown in the patent examples, are obtained with high purity. For procurement managers, this level of control translates to consistent quality and reduced risk of batch failure, which is essential for maintaining supply chain reliability.
How to Synthesize 2-Trifluoromethyl Furan Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates with high efficiency. The process begins with the precise weighing and mixing of the benzoylacetonitrile derivative, 3-bromo-1,1,1-trifluoroacetone, and the selected base promoter in a reaction vessel equipped with magnetic stirring. The reaction is typically conducted under a nitrogen atmosphere to prevent moisture interference, although the specific sensitivity may vary depending on the substrate. Following the reaction period, which can range from 1 to 12 hours depending on the temperature and reagents used, the mixture undergoes a standard aqueous workup involving extraction with ethyl acetate. The organic layers are combined, washed with saturated brine, and dried over anhydrous magnesium sulfate before solvent removal. The resulting crude product is then purified using silica gel column chromatography with a gradient of n-pentane and ethyl acetate. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Mix benzoylacetonitrile derivative, 3-bromo-1,1,1-trifluoroacetone, and a base promoter (e.g., Na2CO3 or NaOAc) in a solvent like 1,4-dioxane under nitrogen.
- Stir the reaction mixture at elevated temperatures (60-80°C) or room temperature for 1-12 hours to facilitate the tandem cyclization.
- Perform aqueous workup with ethyl acetate extraction, dry over magnesium sulfate, and purify the crude product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthesis method offers substantial benefits that directly address the pain points of cost and supply stability in the fine chemical industry. The reliance on commodity chemicals such as benzoylacetonitrile derivatives and 3-bromo-1,1,1-trifluoroacetone means that raw material sourcing is straightforward and less susceptible to market volatility compared to specialized fluorinating reagents. This accessibility contributes significantly to cost reduction in agrochemical intermediate manufacturing, as it eliminates the premium pricing associated with exotic catalysts or complex precursors. Additionally, the one-pot nature of the reaction reduces the number of unit operations required, thereby lowering labor costs and energy consumption associated with heating, cooling, and transferring materials between multiple reactors. For supply chain heads, the simplicity of the process enhances scalability, allowing for seamless transition from laboratory gram-scale experiments to multi-ton commercial production without the need for extensive process re-engineering.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of inexpensive inorganic bases like sodium carbonate or sodium acetate drastically lower the bill of materials. Furthermore, the high atom economy of the tandem reaction minimizes waste disposal costs, which is a significant factor in the total cost of ownership for chemical processes. By consolidating multiple synthetic steps into a single pot, manufacturers can achieve substantial operational savings through reduced solvent usage and shorter cycle times.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a wide range of functional groups including halogens, ethers, and esters, ensures that a diverse portfolio of derivatives can be produced using the same core infrastructure. This flexibility mitigates the risk of supply disruptions caused by the need for specialized equipment for different product lines. The use of stable and commercially available starting materials further secures the supply chain against raw material shortages, ensuring consistent delivery schedules for downstream customers.
- Scalability and Environmental Compliance: The process operates at moderate temperatures (60-80°C) and atmospheric pressure, removing the need for high-pressure autoclaves or cryogenic cooling systems. This simplifies the engineering requirements for scale-up and reduces the capital expenditure needed for plant expansion. Moreover, the reduced solvent volume and the absence of heavy metal contaminants simplify waste treatment protocols, aiding in compliance with increasingly stringent environmental regulations regarding hazardous waste discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, substrate scope, and biological activity. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production pipelines or R&D programs.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method utilizes a one-pot tandem reaction strategy that eliminates the need for multiple isolation steps and harsh fluorinating agents, significantly simplifying the operational workflow and reducing waste generation.
Q: What is the biological activity profile of the resulting compounds?
A: The synthesized 2-trifluoromethyl furan derivatives exhibit potent nematicidal activity, with specific examples demonstrating mortality rates against pine wood nematodes as high as 87% at a concentration of 100 mg/L.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process employs readily available starting materials and mild reaction conditions (60-80°C), making it highly adaptable for commercial scale-up from kilogram to multi-ton quantities without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Trifluoromethyl Furan Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality fluorinated intermediates in the development of next-generation agrochemicals and pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop discovery to industrial manufacturing is smooth and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-trifluoromethyl furan compounds meets the highest international standards. Our commitment to quality assurance means that our clients can rely on us for consistent material performance, which is critical for regulatory filings and final product efficacy.
We invite potential partners to engage with our technical procurement team to discuss how this patented technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of adopting this synthesis route for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive innovation and profitability in your organization.
