Industrial Scale-Up of High-Purity 5-Bromo-2-Chlorobenzoic Acid via Solid-Supported Bromination
Industrial Scale-Up of High-Purity 5-Bromo-2-Chlorobenzoic Acid via Solid-Supported Bromination
The pharmaceutical industry's relentless pursuit of safer, more effective diabetes treatments has placed SGLT-2 inhibitors at the forefront of modern medicinal chemistry. Central to the supply chain of these life-saving medications, such as dapagliflozin and empagliflozin, is the critical intermediate 5-bromo-2-chlorobenzoic acid. Patent CN112979448B introduces a groundbreaking methodology that fundamentally alters the production landscape for this key building block. By leveraging a unique solid-supported bromination strategy utilizing dibromo-amino silica gel and ferric trifluoromethanesulfonate catalysis, this technology addresses the longstanding challenges of regioselectivity and environmental impact. For R&D directors and procurement strategists alike, this innovation represents a pivotal shift towards greener, more efficient pharmaceutical intermediates manufacturing, offering a robust solution for the commercial scale-up of complex organic syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-bromo-2-chlorobenzoic acid has been plagued by significant technical and environmental hurdles that compromise both yield and purity. Traditional routes often rely on harsh liquid-phase bromination systems, such as the use of N-bromosuccinimide (NBS) in concentrated sulfuric acid. As illustrated in prior art, these conditions frequently result in poor regioselectivity, generating substantial quantities of the unwanted 3-bromo-2-chlorobenzoic acid isomer, which complicates downstream purification and reduces overall process efficiency. Furthermore, alternative pathways involving the hydrolysis of expensive precursors like 5-bromo-2-chlorotrifluoromethylbenzene suffer from low yields and high raw material costs, rendering them economically unviable for large-scale production. The reliance on strong corrosive acids like hydrobromic acid and the generation of massive volumes of waste acid not only pose severe safety risks but also create a heavy burden on environmental compliance and waste treatment infrastructure.
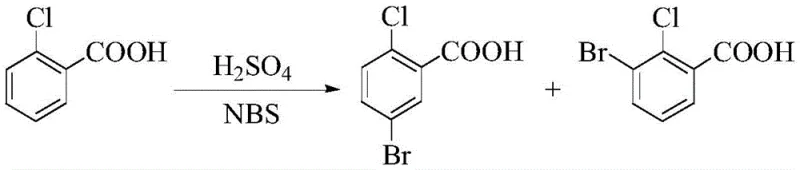
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in the patent utilizes a heterogeneous catalytic system that dramatically enhances reaction control. By employing dibromo-amino silica gel as a solid-supported brominating agent, the process achieves exceptional regioselectivity, effectively suppressing the formation of the 3-bromo isomer. This solid-phase reagent acts not merely as a source of bromine but as a structural director, interacting specifically with the substrate to guide the electrophilic substitution to the desired C5 position. The use of ferric trifluoromethanesulfonate as a Lewis acid catalyst further accelerates the reaction under mild conditions, eliminating the need for aggressive mineral acids. This transition from a homogeneous, corrosive liquid system to a heterogeneous, recyclable solid system simplifies the work-up procedure to a mere filtration step, thereby streamlining the entire manufacturing workflow and significantly improving the safety profile for industrial operators.
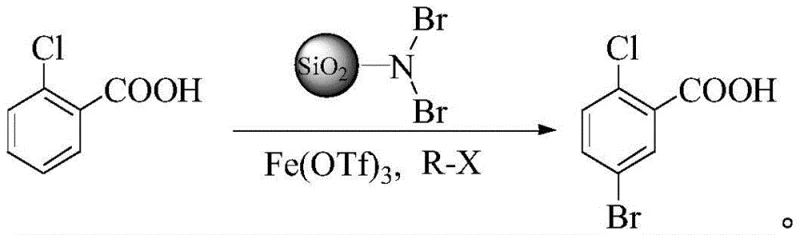
Mechanistic Insights into Fe(OTf)3-Catalyzed Solid-Supported Bromination
For the discerning R&D Director, understanding the mechanistic underpinnings of this high-selectivity transformation is crucial for process validation and optimization. The core of this innovation lies in the synergistic interaction between the ferric triflate catalyst and the functionalized silica gel support. The ferric ion (Fe3+) acts as a potent Lewis acid, activating the bromine species on the silica surface and facilitating the generation of the electrophilic bromonium ion. However, the true brilliance of the system is the supramolecular recognition event that occurs prior to bond formation. The amino groups on the silica gel surface are capable of forming transient hydrogen bonds with the carboxylic acid moiety of the 2-chlorobenzoic acid substrate. This specific interaction anchors the substrate in a precise orientation relative to the bromine source.
This anchoring effect creates a steric environment that favors attack at the C5 position while effectively blocking access to the C3 position due to the proximity of the ortho-chlorine substituent. As depicted in the mechanistic proposal, this hydrogen-bonding network ensures that the bromine transfer occurs with nearly perfect fidelity, resulting in a selectivity of 99% for the target 5-bromo isomer. This level of control is rarely achieved in traditional electrophilic aromatic substitutions, where electronic effects often compete with steric factors to produce mixtures. By locking the substrate into a specific conformation, the dibromo-amino silica gel essentially functions as a molecular template, guaranteeing the production of high-purity pharmaceutical intermediates with minimal isomeric contamination, thus reducing the need for energy-intensive recrystallization steps.
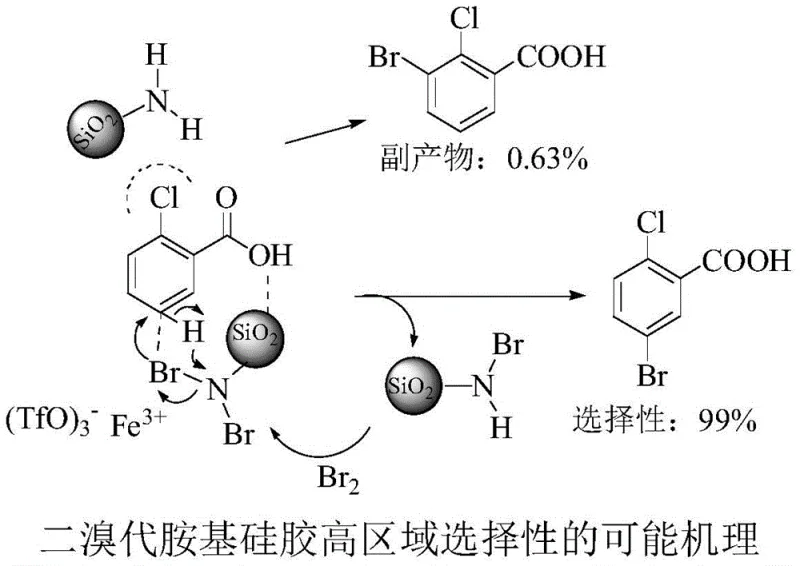
How to Synthesize 5-Bromo-2-Chlorobenzoic Acid Efficiently
The implementation of this advanced synthesis route is designed for seamless integration into existing chemical manufacturing facilities. The process begins with the in-situ or ex-situ preparation of the dibromo-amino silica gel reagent, followed by the catalytic bromination of 2-chlorobenzoic acid in a halogenated hydrocarbon solvent. The reaction conditions are mild, typically requiring temperatures between 60°C and 70°C, which minimizes thermal degradation and energy consumption. Following the reaction completion, the solid silica support is removed via simple hot filtration, allowing for immediate recovery and regeneration of the reagent. The detailed standardized operating procedures, including specific molar ratios, solvent choices, and purification protocols necessary to achieve the reported >99.8% purity, are outlined in the comprehensive guide below.
- Prepare the dibromo-amino silica gel reagent by reacting amino silica gel with bromine and potassium carbonate in dichloromethane under reflux.
- Suspend 2-chlorobenzoic acid, the prepared dibromo-amino silica gel, and ferric trifluoromethanesulfonate catalyst in a halogenated hydrocarbon solvent such as 1-chlorobutane.
- Heat the mixture to 60-70°C for 6-12 hours, then filter hot to remove silica gel, concentrate solvent, and recrystallize from toluene to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates directly into tangible operational improvements and risk mitigation. The shift away from hazardous liquid reagents like concentrated sulfuric acid and elemental bromine significantly lowers the EHS (Environment, Health, and Safety) risk profile of the facility. This reduction in hazard potential simplifies regulatory compliance and reduces the insurance and safety infrastructure costs associated with handling highly corrosive materials. Moreover, the ability to recycle the solid support multiple times with high recovery rates means a drastic reduction in the volume of solid waste generated per kilogram of product. This aligns perfectly with modern green chemistry initiatives and helps manufacturers meet increasingly stringent environmental discharge standards without compromising output.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the elimination of expensive and difficult-to-dispose waste streams. By replacing stoichiometric amounts of corrosive acids with a recyclable solid reagent, the cost of raw materials and waste treatment is substantially lowered. The simplified work-up procedure, which avoids complex aqueous quenches and extractions, reduces labor hours and solvent consumption. Furthermore, the high selectivity minimizes the loss of valuable starting material to byproducts, effectively increasing the overall mass balance efficiency and reducing the cost per unit of the final API intermediate.
- Enhanced Supply Chain Reliability: Reliance on readily available and stable reagents enhances the resilience of the supply chain. The solid-supported brominating agent is stable and easy to handle compared to volatile liquid bromine, reducing logistics risks and storage hazards. The robustness of the catalytic system ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream API manufacturers. This stability allows for better production planning and reduces the likelihood of delays caused by purification bottlenecks or failed batches due to selectivity issues.
- Scalability and Environmental Compliance: The heterogeneous nature of the reaction makes it inherently scalable from pilot plant to multi-ton commercial production. The use of standard filtration equipment for catalyst separation simplifies the engineering requirements for scale-up. Additionally, the significant reduction in acidic wastewater and solid waste generation facilitates easier permitting and operation in regions with strict environmental regulations. This sustainability advantage future-proofs the manufacturing process against tightening global environmental policies, ensuring long-term viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-selectivity bromination technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is essential for technical teams evaluating the transition from legacy processes to this advanced solid-supported methodology.
Q: How does this method improve regioselectivity compared to traditional NBS bromination?
A: Traditional methods using NBS and sulfuric acid often yield significant amounts of the 3-bromo isomer impurity. This novel method utilizes a hydrogen-bonding mechanism between the dibromo-amino silica gel and the carboxyl group, sterically directing bromination exclusively to the C5 position with 99% selectivity.
Q: Can the solid-supported brominating reagent be recycled?
A: Yes, a key advantage of this process is the recyclability of the silica gel support. After filtration, the spent reagent can be re-activated with bromine, achieving a recovery rate of up to 98% after multiple cycles, drastically reducing solid waste.
Q: What represents the primary cost-saving driver in this synthesis route?
A: The elimination of concentrated sulfuric acid and strong corrosive hydrobromic acid reduces equipment corrosion and waste treatment costs. Additionally, the simplified work-up involving simple filtration rather than complex aqueous quenching lowers operational expenses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Bromo-2-Chlorobenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped to verify the <99.8% purity and high regioselectivity required for SGLT-2 inhibitor synthesis. Our commitment to quality assurance guarantees that every batch of 5-bromo-2-chlorobenzoic acid meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for diabetes therapeutics. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this green chemistry technology can drive value and efficiency for your organization.
