Advanced Phase Transfer Catalysis for Scalable Nitroxoline Prodrug Intermediate Production
Introduction to Advanced Aromatic Ether Synthesis
The pharmaceutical industry continuously seeks robust synthetic routes to enhance the bioavailability of established therapeutic agents. Patent CN111646936A introduces a groundbreaking preparation method for aromatic ether compounds, specifically targeting the synthesis of key intermediates for Nitroxoline prodrugs. Nitroxoline, a well-known antibacterial agent, suffers from a short biological half-life and poor patient compliance due to frequent dosing requirements. To address these pharmacokinetic limitations, prodrug strategies such as ((5-nitroquinolin-8-yl) oxy) methylisobutyryl-L-prolinate have been developed. However, the efficient manufacturing of these complex molecules relies heavily on the availability of high-purity aromatic ether precursors. This patent discloses a novel etherification process that significantly outperforms prior art by utilizing a specialized phase transfer catalysis system, offering a reliable pharmaceutical intermediate supplier pathway for next-generation anti-tumor and antibacterial therapies.
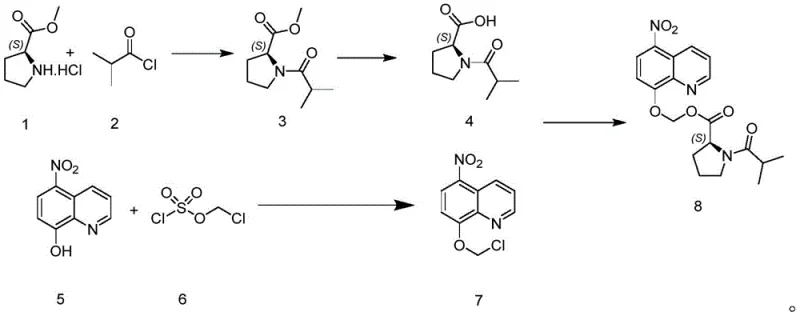
The core innovation lies in the transformation of a phenolic compound (Compound V) into a chloromethyl ether derivative (Compound VII) using chloromethyl chlorosulfonate (Compound 6). Unlike traditional methods that struggle with low conversion and difficult purification, this new approach leverages a binary or ternary solvent system involving water and organic co-solvents like tetrahydrofuran (THF) or 2-methyltetrahydrofuran (2-Me-THF). By optimizing the catalyst loading and base selection, the process achieves exceptional conversion rates, setting a new standard for cost reduction in pharmaceutical intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing 5-nitro-8-chloromethoxyquinoline, a critical building block for Nitroxoline prodrugs, have historically been plagued by inefficiency and operational complexity. As detailed in the comparative examples of the patent, conventional routes often rely on dichloromethane (DCM) as the primary solvent and tetrabutylammonium hydrogen sulfate as the catalyst. These conditions frequently result in dismal conversion rates, often hovering around 30%, which necessitates extensive downstream processing to recover unreacted starting materials. Furthermore, the isolation of the product typically requires silica gel column chromatography, a technique that is notoriously difficult to implement on an industrial scale due to high solvent consumption, safety hazards associated with large volumes of organic eluents, and significant generation of solid waste. The sensitivity of the chloromethyl ether fragment to acid, base, and heat further complicates purification, leading to substantial yield losses and increased production costs.
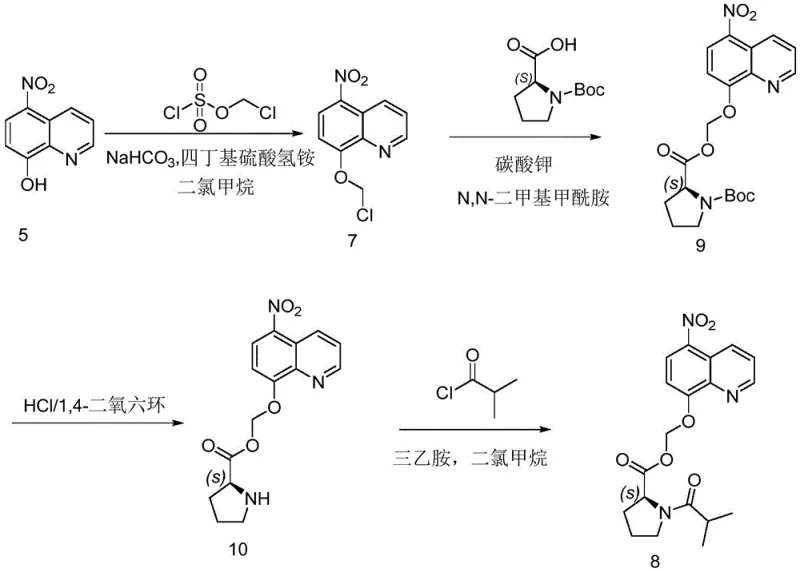
The Novel Approach
The methodology disclosed in CN111646936A represents a paradigm shift in ether synthesis technology. By replacing the traditional DCM system with a water-containing biphasic or ternary solvent mixture, the reaction environment is fundamentally altered to favor product formation and isolation. The use of tetrabutylammonium hydroxide as a phase transfer catalyst in conjunction with inorganic bases like sodium bicarbonate creates a highly reactive interface where the phenolic nucleophile efficiently attacks the electrophilic chloromethyl chlorosulfonate. Crucially, the reaction conditions are tuned such that the product precipitates directly from the reaction mixture upon completion. This phenomenon allows for the isolation of high-purity material simply through filtration and washing, completely bypassing the need for column chromatography. This streamlined workflow not only boosts the overall yield but also drastically simplifies the post-treatment operation, making it highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Phase Transfer Catalyzed Etherification
The success of this synthetic route is rooted in the precise mechanics of phase transfer catalysis (PTC) within a multiphase system. In this reaction, the phenolic starting material (Compound V) is deprotonated by the base in the aqueous phase to form a phenoxide anion. However, the electrophile, chloromethyl chlorosulfonate (Compound 6), resides primarily in the organic phase. The quaternary ammonium catalyst acts as a molecular shuttle, transporting the phenoxide anion into the organic phase where the nucleophilic substitution occurs. The specific selection of tetrabutylammonium hydroxide ensures rapid ion exchange and high solubility in the organic layer, facilitating a swift reaction kinetics profile. Moreover, the choice of solvent mixtures, such as water/THF or water/2-Me-THF/THF, is critical for maintaining the stability of the transition state while ensuring the product's solubility limit is exceeded, driving the equilibrium towards precipitation.
Impurity control is another vital aspect of this mechanism. In traditional homogeneous organic systems, side reactions such as over-alkylation or hydrolysis of the sensitive chloromethyl group can occur readily. The biphasic nature of the new process helps mitigate these risks by compartmentalizing reactive species. The rapid precipitation of the product removes it from the reactive environment, preventing further degradation or side reactions. This kinetic trapping of the product ensures that the impurity profile remains clean, with patent data indicating purities exceeding 96% directly after filtration. This level of intrinsic purity is essential for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for recrystallization or additional purification steps that often erode overall process efficiency.
How to Synthesize 5-Nitro-8-Chloromethoxyquinoline Efficiently
The synthesis of this key intermediate is designed for operational simplicity and robustness, making it accessible for both laboratory optimization and plant-scale production. The process begins with the preparation of the reaction medium, where water and an organic co-solvent are mixed with an inorganic base. The addition of the phase transfer catalyst initiates the activation of the phenolic substrate. Subsequent controlled addition of the alkylating agent triggers the etherification, leading to the spontaneous crystallization of the product. This straightforward protocol eliminates the need for specialized equipment or hazardous anhydrous conditions typically associated with ether synthesis. For detailed procedural parameters regarding stoichiometry, temperature control, and workup procedures, please refer to the standardized synthesis guide below.
- Prepare a biphasic or ternary solvent system comprising water and organic solvents like THF or 2-Me-THF, adding sodium bicarbonate as the base.
- Introduce the phenolic starting material (Compound V) and a quaternary ammonium phase transfer catalyst such as tetrabutylammonium hydroxide.
- Add chloromethyl chlorosulfonate (Compound 6) dropwise at controlled temperatures (25-30°C) to induce precipitation of the high-purity ether product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend beyond mere chemical yield. The elimination of column chromatography represents a massive reduction in operational expenditure, as it removes the cost of silica gel, vast quantities of chromatographic solvents, and the labor associated with running and monitoring columns. Additionally, the shift from dichloromethane to greener solvent systems like THF and water aligns with increasingly stringent environmental regulations, reducing the burden of hazardous waste disposal and solvent recovery. These factors combine to create a manufacturing process that is not only more cost-effective but also more sustainable and resilient against regulatory shifts.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the dramatic simplification of the isolation procedure. By enabling product recovery via simple filtration, the method removes the most expensive and time-consuming unit operation in small molecule synthesis. This leads to substantial cost savings in terms of solvent usage, energy consumption for solvent evaporation, and waste treatment. Furthermore, the high conversion rates mean that raw material utilization is maximized, reducing the effective cost per kilogram of the active intermediate. The use of commercially available and relatively inexpensive reagents like sodium bicarbonate and tetrabutylammonium hydroxide further contributes to a leaner cost structure compared to legacy methods requiring exotic catalysts or anhydrous conditions.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that are prone to failure or batch-to-batch variability. This new method demonstrates remarkable robustness, with consistent high yields achieved across different scales and solvent ratios. The reliance on commodity chemicals ensures that raw material sourcing is stable and not subject to the volatility of specialty reagent markets. Moreover, the simplified workflow reduces the total cycle time for production, allowing manufacturers to respond more agilely to demand fluctuations. This reliability is crucial for maintaining the steady supply of critical antibiotic and anti-tumor intermediates required by global pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen challenges, particularly regarding heat transfer and mixing in heterogeneous systems. However, the precipitation-driven nature of this reaction facilitates easy scale-up, as the product separation mechanism remains effective regardless of batch size. The reduction in halogenated solvent usage (replacing DCM) significantly lowers the environmental footprint of the manufacturing process, aiding companies in meeting their sustainability goals. The ability to produce high-purity material without generating tons of silica waste makes this route highly compliant with modern green chemistry principles, future-proofing the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aromatic ether synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method resolves historical bottlenecks in Nitroxoline prodrug production. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new phase transfer catalysis method improve yield compared to traditional DCM-based processes?
A: The new method utilizes a water/organic solvent system with tetrabutylammonium hydroxide, achieving conversion rates exceeding 90% compared to roughly 30% in conventional methods, while eliminating the need for column chromatography.
Q: What are the purification advantages of this aromatic ether synthesis route?
A: The product precipitates directly from the reaction mixture upon completion, allowing for isolation via simple filtration and washing, which drastically reduces solvent consumption and processing time compared to extraction and chromatography.
Q: Is this process suitable for large-scale commercial manufacturing of Nitroxoline prodrugs?
A: Yes, the process has been demonstrated to scale effectively with high purity (>96%) and uses commercially available reagents and safer solvent systems, making it ideal for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitroxoline Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the development of life-saving medications. Our team of expert chemists has thoroughly analyzed the technological breakthroughs presented in CN111646936A and is fully prepared to leverage this advanced phase transfer catalysis methodology for our clients. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. Our commitment to quality is unwavering, with stringent purity specifications and rigorous QC labs dedicated to verifying that every batch of Nitroxoline derivative meets the highest international standards for pharmaceutical applications.
We invite potential partners to engage with us to explore how this optimized synthetic route can enhance your supply chain efficiency and reduce overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex organic synthesis can support your long-term strategic goals in the pharmaceutical sector.
