Advanced Synthesis of Imipenem Intermediate 4-BMA via Recoverable Chiral Auxiliaries
Advanced Synthesis of Imipenem Intermediate 4-BMA via Recoverable Chiral Auxiliaries
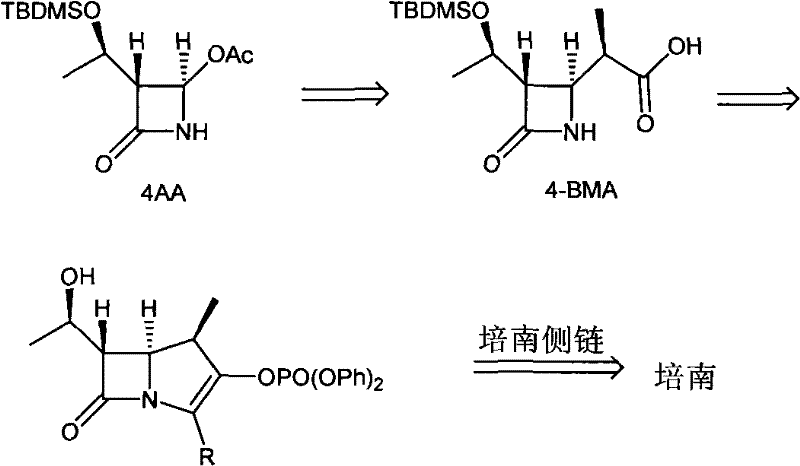
The pharmaceutical landscape for beta-lactam antibiotics continues to evolve, driven by the relentless demand for more efficient and cost-effective manufacturing routes for critical active pharmaceutical ingredients (APIs). A pivotal development in this sector is detailed in patent CN101973915A, which discloses a novel intermediate and preparation method for synthetic imipenem medicines. This technology addresses the longstanding challenges in producing Beta-methylazacyclo-2-ketone, commonly abbreviated as 4-BMA, which serves as the essential key intermediate for the main ring of carbapenem antibiotics. The introduction of a beta-methyl group at the C-1 position of 4-BMA is not merely a structural modification; it fundamentally enhances the stability of the final penem-like pharmaceutical against renal dehydropeptidase (DHP-I), thereby eliminating the need for co-administration with DHP-I inhibitors while maintaining potent antimicrobial activity. This patent presents a robust chemical strategy that leverages a unique chiral sulfonamide auxiliary to achieve high stereoselectivity through a Reformatsky reaction, offering a compelling alternative to existing industrial methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4-BMA has relied heavily on two primary technological pathways, both of which present significant bottlenecks for large-scale manufacturing. The first conventional method involves the generation of a terminal alkene compound from 4-acetoxyazetidinone (4AA), followed by asymmetric hydrogenation to introduce the beta-methyl group. While the hydrogenation step itself is chemically straightforward, the preparation of the requisite terminal alkene precursor is notoriously difficult and low-yielding, creating a supply chain vulnerability. Furthermore, this route often necessitates the use of expensive transition metal catalysts and high-pressure equipment, adding layers of complexity and capital expenditure to the production facility.
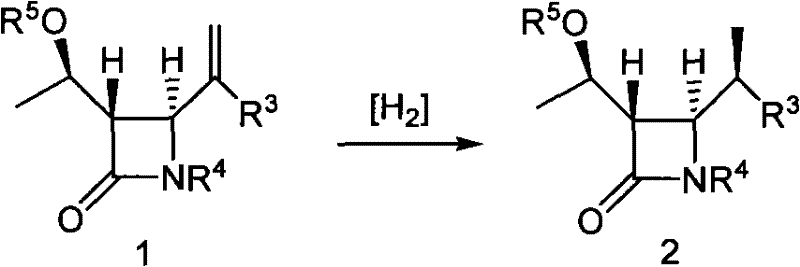
The second traditional approach utilizes aldol condensation or Reformatsky reactions dependent on specialized chiral auxiliaries with complex ring structures, such as 2-oxazolidinones or thiazolidinethiones. Although these auxiliaries can induce chirality, they suffer from severe economic and logistical drawbacks. The synthesis of these cyclic auxiliaries is multi-step and costly, and their properties often lead to poor handling characteristics during processing. Additionally, the stereoselectivity in these traditional Reformatsky examples is highly sensitive to the substituents on the auxiliary, leading to inconsistent product quality. The reliance on such expensive, single-use or difficult-to-recover auxiliaries inflates the cost of goods sold (COGS) and generates substantial chemical waste, conflicting with modern green chemistry principles and sustainability goals demanded by global regulatory bodies.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN101973915A introduces a streamlined synthesis utilizing a novel intermediate of Formula (I). This approach bypasses the need for complex cyclic auxiliaries or difficult alkene precursors. Instead, it employs a chiral sulfonamide derived from readily available amines and sulfonyl chlorides. The synthesis of this auxiliary is remarkably simple, involving a two-step sequence: first, the reaction of a chiral amine with a sulfonyl chloride in the presence of an organic base to form a sulfonamide, and second, the acylation of this sulfonamide with 2-bromopropionyl bromide. This modular design allows for easy tuning of the steric and electronic properties of the auxiliary by varying the R1 and R2 groups, optimizing the process for specific manufacturing needs without reinventing the entire workflow.
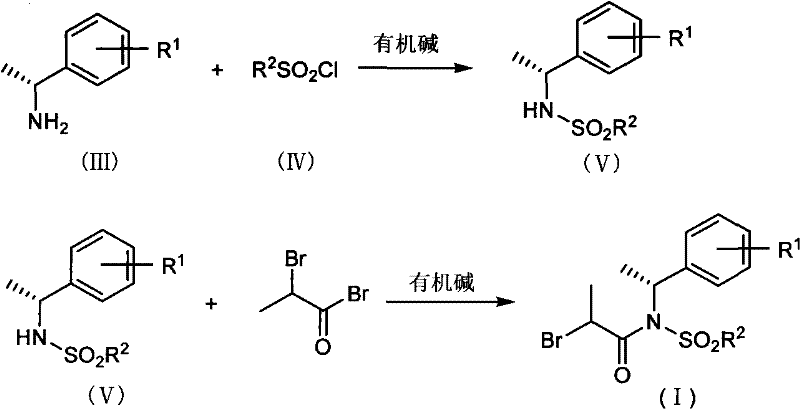
Mechanistic Insights into Zinc-Mediated Reformatsky Coupling
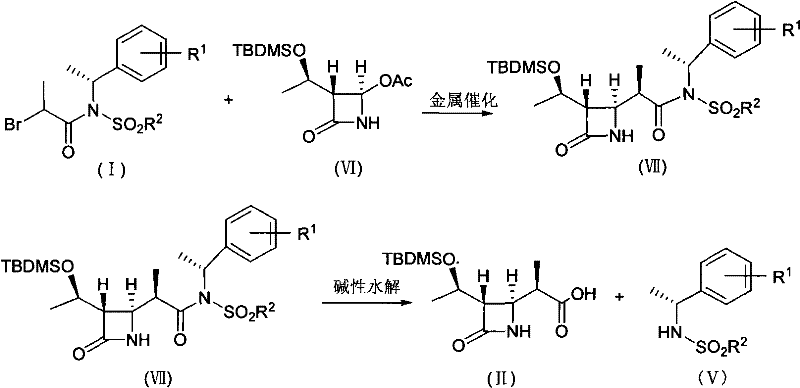
The core of this innovative process lies in the mechanistic elegance of the zinc-mediated Reformatsky reaction between the novel chiral auxiliary (Formula I) and the azetidinone substrate (4AA). Unlike traditional methods that struggle with stereocontrol, this system leverages the inherent chirality of the N-(1-arylalkyl) sulfonamide moiety to direct the facial selectivity of the nucleophilic attack. When the organozinc species generated from the bromo-propionamide attacks the carbonyl of the 4AA, the bulky sulfonamide group creates a defined chiral environment. This steric hindrance effectively shields one face of the approaching electrophile, ensuring that the methyl group is installed predominantly in the desired beta-configuration. The patent data highlights that this specific arrangement leads to a beta/alpha ratio of approximately 91:9, a level of diastereoselectivity that is critical for minimizing downstream purification burdens and maximizing the yield of the biologically active isomer.
Following the coupling reaction, the process incorporates a highly efficient cleavage strategy. The resulting adduct (Formula VII) undergoes alkaline hydrolysis, typically using mild bases like lithium hydroxide or sodium hydroxide, potentially assisted by hydrogen peroxide. This step serves a dual purpose: it hydrolyzes the ester linkage to reveal the free carboxylic acid of the target 4-BMA, and simultaneously cleaves the amide bond connecting the chiral auxiliary. Crucially, this cleavage releases the chiral sulfonamine (Formula V) intact. This recovery mechanism is a game-changer for process economics, as the valuable chiral source is not consumed but rather acts as a recyclable reagent. The ability to separate and reuse the auxiliary significantly reduces the raw material intensity of the process, aligning perfectly with the principles of atom economy and waste reduction in fine chemical manufacturing.
How to Synthesize Imipenem Intermediate 4-BMA Efficiently
The implementation of this synthesis route requires careful attention to reaction conditions to maximize yield and stereoselectivity. The process begins with the preparation of the chiral bromo-amide auxiliary under controlled low temperatures to prevent racemization or side reactions. Subsequently, the activation of zinc powder is critical for the success of the Reformatsky step; the patent suggests refluxing zinc in THF to ensure a reactive surface area before the addition of the substrates. The coupling reaction is exothermic and must be managed within a temperature window of 0 to 30°C to maintain the integrity of the beta-lactam ring while promoting the formation of the organozinc intermediate. Following the coupling, the hydrolysis step is performed under mild alkaline conditions to avoid ring opening of the sensitive azetidinone core. For a detailed breakdown of the specific molar ratios, solvent choices, and workup procedures required to replicate this high-performance synthesis, please refer to the standardized protocol below.
- React a chiral amine with a sulfonyl chloride in the presence of an organic base to form a sulfonamide intermediate.
- Acylate the sulfonamide with 2-bromopropionyl bromide to generate the key chiral auxiliary reagent (Formula I).
- Perform a zinc-mediated Reformatsky reaction between the chiral auxiliary and 4-acetoxyazetidinone (4AA), followed by alkaline hydrolysis to release 4-BMA and recover the auxiliary.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers transformative benefits that extend far beyond simple chemical yield improvements. The shift away from expensive, proprietary cyclic auxiliaries to commodity chemicals like sulfonyl chlorides and chiral amines fundamentally alters the cost structure of 4-BMA production. By utilizing raw materials that are abundant in the global chemical market, manufacturers can insulate themselves from the price volatility associated with specialized reagents. Furthermore, the recoverability of the chiral auxiliary means that the effective consumption of this high-value component is drastically reduced over multiple batches. This circular usage model translates directly into substantial cost savings in API manufacturing, allowing for more competitive pricing strategies in the generic antibiotic market without compromising on quality or purity standards.
- Cost Reduction in Manufacturing: The elimination of costly noble metal catalysts and complex cyclic auxiliaries results in a significantly leaner bill of materials. Since the chiral auxiliary can be recovered and recycled after the hydrolysis step, the operational expenditure related to chiral sources is amortized over many production cycles. This efficiency removes the need for expensive metal scavenging steps often required in hydrogenation routes, further simplifying the downstream processing and reducing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as tosyl chloride, benzyl amines, and 2-bromopropionyl bromide ensures a robust and resilient supply chain. Unlike specialized reagents that may have single-source suppliers and long lead times, the inputs for this process are produced by numerous chemical vendors globally. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages, ensuring consistent delivery schedules for downstream API producers and enhancing overall supply security.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, utilizing common solvents like dichloromethane, THF, and toluene, which are standard in industrial multipurpose reactors. The moderate temperature ranges (-20°C to 30°C) do not require extreme cryogenic cooling or high-energy heating, making the process energy-efficient. Additionally, the high stereoselectivity reduces the generation of unwanted diastereomers, minimizing the solvent and silica load required for purification. This reduction in waste generation facilitates easier compliance with increasingly stringent environmental regulations regarding solvent discharge and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and strategic value of this synthesis route for potential licensees and manufacturing partners. Understanding these nuances is essential for R&D teams evaluating process transfers and for executive leadership assessing the long-term viability of adopting this new methodology for carbapenem production.
Q: What are the advantages of this new 4-BMA synthesis method over traditional asymmetric hydrogenation?
A: Unlike traditional methods that require difficult-to-prepare terminal alkene precursors for asymmetric hydrogenation, this novel route utilizes a readily available chiral sulfonamide auxiliary. This simplifies the precursor synthesis significantly and avoids the need for expensive noble metal catalysts often associated with hydrogenation processes.
Q: Is the chiral auxiliary used in this process recoverable?
A: Yes, a key feature of this invention is the recoverability of the chiral auxiliary. After the Reformatsky reaction and subsequent alkaline hydrolysis to release the target 4-BMA, the chiral sulfonamide byproduct is liberated and can be separated and reused, drastically reducing the effective cost of goods.
Q: What level of stereoselectivity can be achieved with this Reformatsky protocol?
A: The process demonstrates high stereoselectivity, specifically favoring the formation of the beta-methyl configuration at the C-1 position of the azetidinone ring. The patent data indicates a beta/alpha ratio of approximately 91:9, which is critical for the biological activity of the final carbapenem antibiotic.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imipenem Intermediate Supplier
The technological advancements presented in CN101973915A represent a significant leap forward in the manufacture of carbapenem intermediates, yet translating laboratory success into commercial reality requires a partner with deep process engineering expertise. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering comprehensive CDMO services tailored to the complex needs of beta-lactam antibiotic production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of stereoselectivity and yield observed in the lab is maintained at the plant scale. We operate state-of-the-art facilities equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 4-BMA or related intermediate meets the exacting standards required for global pharmaceutical registration.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective synthesis route for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this auxiliary-based method for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data from our pilot runs and to discuss detailed route feasibility assessments. Let us help you secure a sustainable, high-quality, and economically superior supply of critical imipenem intermediates for the global market.
