Scalable Production of High-Purity Furanylaldehydes via Green Manganese-Catalyzed Oxidation
Introduction to Advanced Furanylaldehyde Synthesis
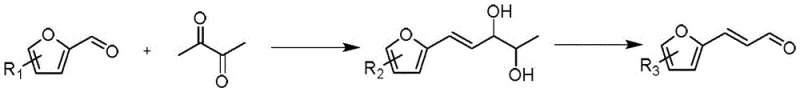
The chemical industry is constantly seeking more sustainable and efficient pathways for producing high-value intermediates, particularly those serving the pharmaceutical and agrochemical sectors. A significant breakthrough in this domain is detailed in Chinese Patent CN110862363B, which discloses a novel method for preparing furanylaldehyde compounds. These compounds, such as α-furanacrolein, are critical building blocks for synthesizing schistosomiasis treatments like furanpropylamine, as well as various fragrances and polymer additives. The patent outlines a sophisticated two-stage process that begins with an Aldol condensation between furfural derivatives and 2,3-butanedione, followed by a selective oxidative cleavage mediated by a heterogeneous manganese catalyst. This approach represents a paradigm shift from traditional methods that often rely on toxic reagents or prohibitively expensive noble metals.
By leveraging readily available starting materials like furfural and employing earth-abundant manganese catalysts, this technology addresses key pain points in cost reduction in pharmaceutical intermediate manufacturing. The process operates under relatively mild conditions, utilizing oxygen or air as a green oxidant, which drastically simplifies waste treatment protocols. For R&D directors and procurement managers alike, understanding the mechanistic nuances and commercial implications of this patent is essential for securing a reliable furanylaldehyde supplier capable of delivering high-purity materials with a reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of α-furanacrolein and related long-chain furyl aldehydes has been plagued by significant technical and economic hurdles. Early methodologies, such as those reported by Masahiro Toyota, relied on the condensation of furfural with triethylamine catalyzed by chlorotrimethylsilane. While effective, both the amine and the silane reagent possess strong toxicity, creating severe occupational health risks and complicating regulatory compliance for commercial scale-up of complex pharmaceutical intermediates. Furthermore, alternative routes utilizing gold-based catalysts (Au/Al2O3 or Au@UiO-66) on ethanol condensates face economic barriers due to the high cost of noble metals, rendering them less viable for bulk chemical production.
Another prevalent issue in legacy processes is the reliance on harsh oxidizing conditions. Traditional oxidation of secondary allylic alcohols often employs chromium-based reagents like pyridinium chlorochromate (PCC) combined with periodic acid. These stoichiometric oxidants generate substantial amounts of hazardous heavy metal waste, imposing a heavy burden on downstream purification and wastewater treatment facilities. Even more recent attempts using binary Cu-Ce oxides or magnetic Fe@C catalysts often require elevated temperatures (up to 140°C) and pressure (0.3 MPa), along with additional bases like potassium carbonate. These factors collectively increase energy consumption and operational complexity, limiting the feasibility of these methods for high-purity OLED material or fine chemical applications where impurity profiles must be tightly controlled.
The Novel Approach
The methodology presented in CN110862363B offers a compelling solution by decoupling the carbon chain extension from the oxidative functionalization through a streamlined sequence. Instead of direct oxidation of fragile intermediates, the process first constructs the carbon skeleton via a base-catalyzed Aldol condensation between furfural derivatives and 2,3-butanedione. This is followed by a selective reduction step to form a stable diol condensation product. The true innovation lies in the subsequent oxidative cleavage step, which utilizes a heterogeneous manganese catalyst supported on materials like alumina or silica. This system enables the transformation to proceed under atmospheric pressure with oxygen or air as the sole oxidant, eliminating the need for toxic chromium reagents or expensive gold catalysts.
This novel route not only enhances selectivity and minimizes by-product formation but also aligns perfectly with green chemistry principles. The use of a heterogeneous catalyst facilitates easy separation via filtration, allowing for potential catalyst recycling and reducing the overall catalyst loading cost. For supply chain heads, this translates to a more robust and continuous production capability. The mild reaction conditions, ranging from -20°C to 120°C depending on the specific step, ensure that sensitive functional groups on the furan ring remain intact, thereby preserving the integrity of the final high-purity furanylaldehydes. This technological advancement positions the process as a superior alternative for manufacturers aiming to optimize their production lines for sustainability and cost-efficiency.
Mechanistic Insights into Manganese-Catalyzed Oxidative Cleavage
To fully appreciate the technical depth of this invention, one must analyze the dual-phase reaction mechanism involving both C-C bond formation and selective oxidative scission. The process initiates with an Aldol condensation where the alpha-protons of 2,3-butanedione are abstracted by a base catalyst (such as NaOH or K2CO3), generating a nucleophilic enolate. This enolate attacks the carbonyl carbon of the furfural derivative, forming a β-hydroxy ketone intermediate which subsequently dehydrates to yield an α,β-unsaturated diketone. Crucially, the patent describes a subsequent reduction of this diketone using sodium borohydride (NaBH4) to produce a vicinal diol structure. This reduction step is vital as it stabilizes the intermediate and sets the stage for the specific oxidative cleavage required to generate the aldehyde functionality without over-oxidation to carboxylic acids.
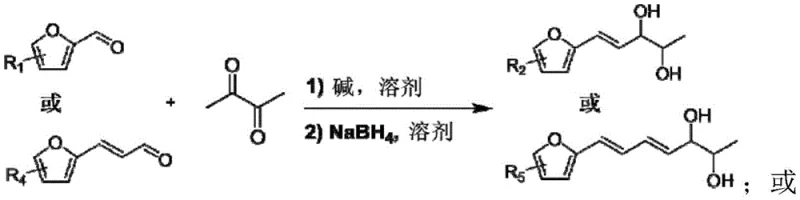
The second phase involves the heterogeneous manganese catalyst, where the active Mn species facilitate the activation of molecular oxygen. The diol condensation product adsorbs onto the catalyst surface, where the manganese centers mediate the electron transfer necessary for oxidative cleavage of the C-C bond adjacent to the hydroxyl groups. This mechanism is highly selective, preferentially cleaving the specific bond to release the target furanylaldehyde while minimizing side reactions such as ring opening of the furan or complete combustion to CO2. The heterogeneity of the catalyst ensures that the active sites are accessible yet distinct from the bulk solution, preventing homogeneous radical chains that often lead to polymeric tars. This precise control over the oxidation state is what allows the process to achieve high yields (up to 94% in optimized examples) and exceptional purity, meeting the stringent requirements for reducing lead time for high-purity pharmaceutical intermediates.
How to Synthesize Furanylaldehydes Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly the choice of solvent and catalyst support. The Aldol condensation is typically conducted in polar aprotic or protic solvents like ether, ethanol, or methanol at temperatures between -20°C and 60°C to control the exotherm and prevent polymerization. Following the reduction with NaBH4, the crude diol product is subjected to the oxidation step in a solvent such as n-butanol or toluene. The heterogeneous manganese catalyst, prepared by impregnating Mn salts onto supports like Al2O3 or SiO2 followed by calcination, is added in a mass ratio ranging from 1:1 to 100:1 relative to the substrate. The reaction is then heated to 50-200°C under an oxygen atmosphere. For a comprehensive guide on the specific molar ratios, workup procedures, and purification techniques validated by the patent data, please refer to the standardized protocol below.
- Perform Aldol condensation between furfural derivatives and 2,3-butanedione using a base catalyst in solvent at -20 to 60°C to form an enedione intermediate.
- Reduce the enedione intermediate using sodium borohydride (NaBH4) to obtain the diol condensation product.
- Oxidize the condensation product using a heterogeneous manganese catalyst (e.g., Mn/Al2O3) and an oxygen source at 50-200°C to yield the target furanylaldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this manganese-catalyzed pathway offers transformative benefits for procurement strategies and supply chain resilience. The primary driver for cost optimization is the substitution of noble metal catalysts with earth-abundant manganese. Gold and palladium catalysts are subject to volatile market pricing and supply constraints, whereas manganese is inexpensive and widely available. Furthermore, the elimination of stoichiometric toxic oxidants like chromium reagents removes the significant costs associated with hazardous waste disposal and environmental remediation. This shift not only lowers the direct cost of goods sold (COGS) but also reduces the regulatory burden, facilitating smoother audits and compliance checks for global supply chains.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the use of heterogeneous catalysis, which allows for the potential recovery and reuse of the catalyst, thereby amortizing the catalyst cost over multiple batches. Additionally, the avoidance of expensive protecting group chemistry or harsh reagents simplifies the bill of materials. By utilizing simple bases like sodium hydroxide and common solvents like ethanol, the process minimizes raw material expenditure. The high selectivity of the reaction means less material is lost to by-products, effectively increasing the overall yield and reducing the cost per kilogram of the final active ingredient. This efficiency is critical for maintaining competitive pricing in the global fine chemical intermediates market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source specialty reagents. This method relies on commodity chemicals such as furfural, 2,3-butanedione, and manganese salts, which are produced at scale by multiple vendors worldwide. This diversification of the supply base mitigates the risk of shortages. Moreover, the robustness of the heterogeneous catalyst system ensures consistent batch-to-batch quality, reducing the incidence of failed runs that can disrupt delivery schedules. For partners seeking a reliable furanylaldehyde supplier, this stability translates to predictable lead times and the ability to scale production rapidly in response to market demand without compromising quality.
- Scalability and Environmental Compliance: As regulatory pressures regarding green chemistry intensify, processes that minimize E-factors (mass of waste per mass of product) are increasingly valued. This technology's use of molecular oxygen as a clean oxidant generates water as the primary by-product, significantly lowering the environmental impact compared to chromium-based routes. The heterogeneous nature of the catalyst simplifies downstream processing; filtration removes the catalyst without the need for complex extraction or distillation steps often required for homogeneous systems. This ease of scale-up from gram to tonnage quantities ensures that the process remains viable and compliant even as production volumes increase to meet the needs of large-scale pharmaceutical intermediate manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this furanylaldehyde synthesis technology. These insights are derived directly from the experimental data and claims within patent CN110862363B, providing a factual basis for evaluating the process feasibility. Understanding these details is crucial for technical teams assessing the integration of this route into existing manufacturing frameworks.
Q: What are the advantages of using heterogeneous manganese catalysts over traditional noble metal catalysts?
A: Heterogeneous manganese catalysts significantly reduce raw material costs compared to expensive gold or palladium systems. They also offer easier separation from the reaction mixture, facilitating catalyst recycling and minimizing heavy metal contamination in the final pharmaceutical intermediate.
Q: Does this process require harsh oxidizing agents like chromium reagents?
A: No, the process described in CN110862363B utilizes molecular oxygen or air as the terminal oxidant in the presence of the manganese catalyst. This eliminates the need for toxic stoichiometric oxidants like PCC or periodic acid, greatly improving environmental compliance and safety.
Q: Can this synthesis method be scaled for industrial production of API intermediates?
A: Yes, the use of heterogeneous catalysts and mild reaction conditions (-20°C to 200°C range) makes the process highly amenable to scale-up. The robustness of the manganese support system ensures consistent performance in large-scale reactors, supporting commercial supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Furanylaldehydes Supplier
The technological potential of manganese-catalyzed oxidative cleavage represents a significant opportunity for the fine chemical industry, yet translating patent literature into commercial reality requires specialized expertise. NINGBO INNO PHARMCHEM stands at the forefront of this translation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation to ensure that every batch of furanylaldehydes meets stringent purity specifications, free from the heavy metal residues often associated with older synthetic methods. We understand that for R&D directors, consistency is key, and our process validation protocols are designed to deliver that reliability.
We invite procurement managers and supply chain leaders to engage with us to explore how this green synthesis route can optimize your sourcing strategy. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our commitment to innovation and quality can secure your supply chain for critical pharmaceutical intermediates while driving down overall manufacturing costs.
