Optimizing Nafcillin Sodium Production: A Technical Breakthrough for Commercial Scale-Up
Optimizing Nafcillin Sodium Production: A Technical Breakthrough for Commercial Scale-Up
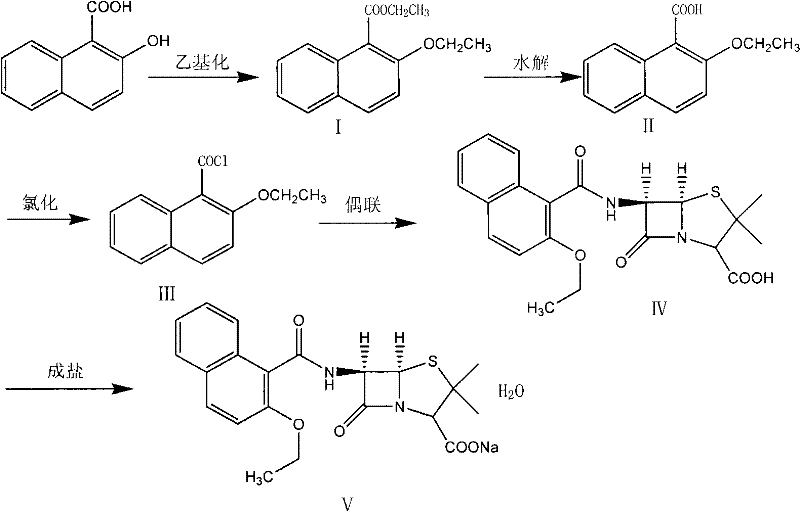
The pharmaceutical industry continuously demands more efficient and robust synthetic routes for critical antibiotics, particularly penicillinase-resistant variants like Nafcillin Sodium. Patent CN101781315A introduces a refined synthesizing method for Nafcillin Sodium Hydrate that addresses longstanding challenges in yield, purity, and process stability. By utilizing 2-hydroxy-1-naphthoic acid as the initial raw material, this protocol establishes a streamlined pathway involving ethylation, hydrolysis, chlorination, and coupling with 6-aminopenicillanic acid (6-APA). Unlike previous iterations that struggled with residual solvent control and product discoloration, this method achieves a content purity exceeding 99.5 percent while maintaining operational simplicity. For R&D directors and procurement specialists seeking a reliable antibiotic intermediate supplier, understanding the nuances of this optimized route is essential for securing high-quality supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Nafcillin Sodium has been plagued by significant technical hurdles that impact both cost and quality. Prior art, such as the method described in patent CN 101456869A, relies on reacting 2-ethoxy-1-naphthoic acid with a substantial excess of thionyl chloride at elevated temperatures, specifically around 65°C under reflux conditions. This aggressive thermal environment promotes severe side reactions, leading to increased byproduct formation and notoriously difficult post-treatment layering processes. Furthermore, literature from SIDNEYS WALKENSTEIN and E.G. BRAIN highlights alternative approaches using dimethylformamide catalysts or thionyl chloride as a solvent, yet these methods suffer from incomplete reactions, low yields, and the inherent instability of Nafcillin Sodium in aqueous solutions during evaporation. The cumulative effect of these conventional defects is a final product with poor color characteristics and inconsistent purity profiles, creating substantial bottlenecks for cost reduction in antibiotic manufacturing.
The Novel Approach
The methodology outlined in CN101781315A represents a paradigm shift by decoupling the harsh chlorination conditions from the sensitive coupling step. Instead of direct high-temperature reflux, the novel approach employs a controlled low-temperature chlorination at 0 to 5°C, followed by the complete removal of thionyl chloride and methylene dichloride under vacuum prior to coupling. This strategic separation prevents the degradation of the beta-lactam ring and minimizes the generation of colored impurities. Additionally, the introduction of a specific salification step using sodium bicarbonate or sodium carbonate in a mixed acetone-water solution allows for the direct preparation of the hydrate form without the need for complex recrystallization sequences. This innovation not only simplifies the workflow but also ensures that residual solvents are easily controlled, directly addressing the pain points of previous syntheses and offering a viable path for the commercial scale-up of complex antibiotic intermediates.
Mechanistic Insights into Optimized Acylation and Coupling
The core of this synthetic advancement lies in the precise management of the acyl chloride intermediate (Intermediate III). In the chlorination step, the reaction between 2-ethoxy-1-naphthoic acid and thionyl chloride is conducted in methylene dichloride with catalytic DMF at strictly maintained low temperatures. This condition is critical because it facilitates the formation of the acid chloride while suppressing the electrophilic attack on the naphthalene ring or the ether linkage, which are common sources of structural impurities. Following the reaction, the vacuum distillation of volatiles ensures that no acidic residues remain to catalyze the decomposition of the subsequent beta-lactam product. When this purified acid chloride is introduced to the 6-APA solution, which is pre-cooled to approximately -10°C in the presence of triethylamine, the nucleophilic attack by the amino group of 6-APA proceeds with high regioselectivity. This meticulous temperature gradient from chlorination to coupling preserves the stereochemical integrity of the penicillin nucleus, a factor paramount for biological activity.
Impurity control is further enhanced during the workup and salification phases. The process utilizes a pH-swing extraction strategy where the coupled product is first dissolved in alkaline water (pH 8) to separate organic impurities, then re-acidified to pH 2 to 3 to precipitate the free acid. This dual-phase purification effectively removes unreacted 6-APA and non-polar side products before the final salt formation. The final conversion to Nafcillin Sodium Hydrate in an acetone-water mixture leverages the differential solubility of the sodium salt versus potential inorganic salts like sodium chloride or unreacted alkali. By filtering off intact mineral alkali before the final crystallization, the process inherently limits inorganic contamination. For technical teams evaluating high-purity API intermediates, this mechanism demonstrates a robust defense against the typical impurity profiles associated with semi-synthetic penicillins.
How to Synthesize Nafcillin Sodium Efficiently
Executing this synthesis requires strict adherence to the sequential transformation of the naphthoic acid scaffold. The process begins with the ethylation of 2-hydroxy-1-naphthoic acid in DMF, followed by hydrolysis to secure the 2-ethoxy substitution pattern. The subsequent conversion to the acid chloride and coupling with 6-APA must be performed under anhydrous and低温 conditions to prevent hydrolysis of the reactive intermediates. The detailed standardized synthesis steps, including specific reagent ratios and temperature ramps derived from the patent embodiments, are provided in the guide below for technical reference.
- Ethylation of 2-hydroxy-1-naphthoic acid using ethyl sulfate or bromoethane in DMF with sodium hydroxide to form Intermediate I.
- Hydrolysis of Intermediate I using alcoholic potassium or sodium hydroxide, followed by acidification to obtain Intermediate II (2-ethoxy-1-naphthoic acid).
- Chlorination of Intermediate II with thionyl chloride at 0-5°C to form the acid chloride (Intermediate III), followed by coupling with 6-APA and final salification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage stems from the drastic simplification of the purification train. By eliminating the need for extensive chromatographic purification or multiple recrystallizations to fix color issues, the overall processing time is significantly reduced. This efficiency translates directly into lower utility consumption and reduced labor costs per kilogram of finished product. Furthermore, the use of commodity solvents like ethanol, acetone, and dichloromethane ensures that raw material sourcing remains stable and unaffected by niche supply shocks, thereby enhancing supply chain reliability for long-term production contracts.
- Cost Reduction in Manufacturing: The elimination of excessive thionyl chloride usage and the avoidance of high-temperature reflux conditions result in substantial cost savings regarding reagent consumption and energy input. Conventional methods often require large excesses of chlorinating agents to drive reactions to completion, generating significant hazardous waste that requires expensive disposal. By optimizing the stoichiometry and reaction temperature, this new method minimizes waste generation at the source. Additionally, the high yield and purity reduce the loss of valuable 6-APA, a high-cost starting material, ensuring that every gram of input contributes maximally to the final output value.
- Enhanced Supply Chain Reliability: The robustness of this process against minor fluctuations in reaction parameters makes it highly suitable for multi-ton production campaigns. Traditional methods that produce dark-colored oils or gums often lead to batch failures or extended reprocessing times, causing unpredictable delays in delivery schedules. In contrast, the consistent formation of crystalline intermediates and the final white solid product in this method ensures predictable cycle times. This predictability allows supply chain planners to maintain leaner inventory levels while confidently meeting just-in-time delivery commitments to downstream formulation partners.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the controlled addition of reagents and the closed-system vacuum distillation steps minimize operator exposure to corrosive gases and volatile organics. The process generates less acidic wastewater compared to methods requiring massive aqueous quenches of excess thionyl chloride. This reduction in effluent load simplifies compliance with increasingly stringent environmental regulations. Moreover, the straightforward crystallization behavior of the final hydrate form facilitates easy filtration and drying on industrial centrifuges and dryers, removing common bottlenecks associated with scaling up sticky or amorphous pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Nafcillin Sodium Hydrate using this specific patent methodology. These insights are derived directly from the experimental data and comparative analysis provided in the intellectual property documentation. They serve to clarify the operational advantages and quality benchmarks associated with this synthesis route.
Q: How does this synthesis method improve product color compared to conventional routes?
A: Conventional methods often utilize excessive thionyl chloride at elevated temperatures (e.g., 65°C reflux), leading to side reactions and poor product color. This patented method strictly controls the chlorination temperature at 0-5°C and removes excess thionyl chloride under vacuum before coupling, significantly reducing byproduct formation and ensuring a superior white to off-white product appearance.
Q: What are the critical control points for ensuring high purity (>99.5%)?
A: Critical control points include the precise pH adjustment during the extraction of Nafcillin acid (pH 2-3 for precipitation, pH 8 for extraction) and the strict temperature control during the coupling reaction (-10°C to 10°C). Additionally, the final salification in an acetone-water system allows for effective removal of residual solvents and unreacted alkali, guaranteeing high purity specifications.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial scalability. It utilizes common solvents like dichloromethane, ethanol, and acetone, and avoids complex purification steps. The examples provided in the patent demonstrate successful execution in reactors ranging from 50L to 200L, indicating robust feasibility for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nafcillin Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this optimized synthesis are fully realized in our manufacturing facilities. We maintain stringent purity specifications through our rigorous QC labs, employing advanced analytical techniques to verify that every batch of Nafcillin Sodium meets the >99.5 percent purity threshold required for modern pharmaceutical applications. Our commitment to quality assurance ensures that the impurity profiles remain well within regulatory limits, providing peace of mind for your regulatory filings.
We invite global partners to leverage our technical capabilities for their antibiotic supply needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our mastery of this advanced synthetic method can drive efficiency and reliability in your supply chain, positioning your organization for success in the competitive global antibiotics market.
