Advanced Manufacturing of L-2-Aminobutanamide Hydrochloride for High-Purity Levetiracetam Production
Advanced Manufacturing of L-2-Aminobutanamide Hydrochloride for High-Purity Levetiracetam Production
The global demand for next-generation antiepileptic drugs continues to surge, driving the need for robust and cost-efficient supply chains for critical intermediates. Patent CN101928229A introduces a transformative process method for producing L-2-aminobutanamide hydrochloride, a pivotal precursor in the synthesis of Levetiracetam. This technology addresses longstanding industry challenges regarding excessive impurity content and prohibitive production costs that have historically burdened manufacturers. By shifting away from hazardous halogenation routes toward a streamlined aminonitrile hydrolysis pathway, this innovation offers a compelling value proposition for pharmaceutical supply chains seeking reliability and quality. The following analysis details how this proprietary methodology enhances both technical feasibility and commercial viability for API producers worldwide.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-2-aminobutanamide has relied on pathways fraught with significant operational and safety hazards. Traditional methods often involve the bromination of butyric acid, a process that generates corrosive hydrogen bromide gas and creates a poor workshop environment due to the volatility of bromine. Furthermore, the lack of selectivity in alpha-position substitution frequently leads to di- or tri-brominated byproducts, complicating downstream purification and resulting in suboptimal product purity that fails to meet stringent international client specifications. Alternative routes utilizing methionine desulfuration, while operationally simpler, suffer from exorbitant raw material costs that render them economically unviable for large-scale commercial adoption. Additionally, older Strecker synthesis variations often require high-pressure and high-temperature hydrolysis conditions, posing potential safety risks and necessitating expensive, specialized reactor infrastructure that limits accessibility for small to medium-sized enterprises.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes propionaldehyde as a readily available starting material, reacting it with ammonia water, ammonium chloride, and sodium cyanide to form 2-aminobutyronitrile under controlled conditions. This intermediate is subsequently hydrolyzed under mild alkaline conditions using sodium hydroxide, avoiding the extreme pressures associated with legacy methods. The process is characterized by its operational simplicity and the use of common industrial reagents, which drastically lowers the barrier to entry for manufacturing. By optimizing reaction parameters such as temperature and feed ratios, this method achieves a finished product purity exceeding 99.5% after recrystallization, effectively removing sodium chloride byproducts. This shift not only mitigates safety risks associated with halogen handling but also ensures a consistent supply of high-quality intermediate suitable for sensitive pharmaceutical applications.
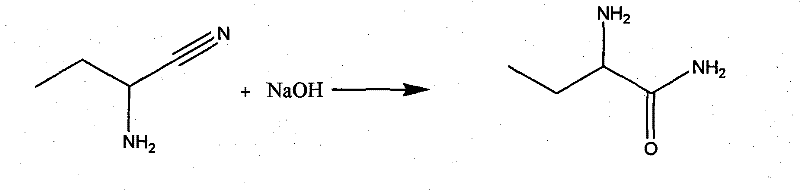
Mechanistic Insights into Alkaline Hydrolysis and Chiral Resolution
The core of this technological advancement lies in the precise control of the hydrolysis and resolution steps. The conversion of 2-aminobutyronitrile to 2-aminobutanamide is executed using a 10% sodium hydroxide solution at temperatures between 60°C and 100°C. This specific thermal window is critical; it provides sufficient energy to drive the hydrolysis of the nitrile group to the amide without promoting further hydrolysis to the carboxylic acid or causing racemization of the chiral center. The stoichiometry is carefully managed, with caustic soda consumption optimized to 1 to 1.5 grams per gram of propionaldehyde equivalent, ensuring complete conversion while minimizing excess base that could complicate workup. Following hydrolysis, the crude racemic amide undergoes a sophisticated resolution process using L-tartaric acid. This chiral resolving agent forms a diastereomeric salt with the desired L-enantiomer, leveraging differences in solubility to separate it from the unwanted D-isomer. The resulting L-2-amino amides tartrate salt is then treated with sodium ethylate and subsequently acidified with hydrochloric acid to yield the final hydrochloride salt, ensuring the optical purity reaches levels above 99.5%.
Impurity control is inherently built into this mechanistic design. By avoiding the use of bromine, the process eliminates the formation of poly-brominated impurities that are notoriously difficult to remove via standard crystallization. Furthermore, the mild alkaline hydrolysis conditions prevent the degradation of the amino group, which can occur under harsher acidic or high-pressure alkaline environments. The recrystallization step serves as a final polishing stage, effectively scrubbing out inorganic salts like sodium chloride generated during the neutralization phases. This multi-layered approach to purity assurance means that the final API intermediate requires minimal additional purification before being used in the subsequent cyclization step to form Levetiracetam, thereby streamlining the overall manufacturing timeline and reducing solvent consumption.
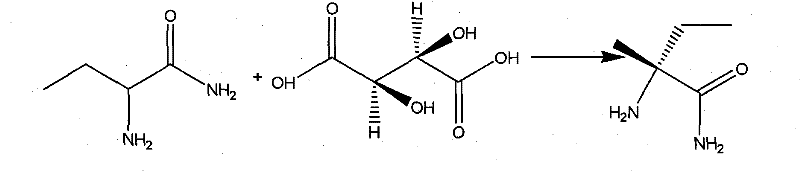
How to Synthesize L-2-Aminobutanamide Hydrochloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing reproducibility and safety. The process begins with the formation of the aminonitrile backbone, followed by a controlled hydrolysis and a final chiral separation. Operators must pay close attention to temperature gradients during the exothermic addition of reagents and maintain strict vacuum conditions during solvent removal to prevent product degradation. The detailed标准化 synthesis steps provided below offer a comprehensive guide for technical teams looking to implement this route, ensuring that critical parameters such as pH, temperature, and stirring rates are maintained within the optimal ranges defined by the intellectual property.
- React propionaldehyde with ammonia, ammonium chloride, and sodium cyanide at 50-80°C to form 2-aminobutyronitrile.
- Hydrolyze the nitrile intermediate under mild alkaline conditions (60-100°C) using sodium hydroxide to obtain 2-aminobutanamide.
- Perform chiral resolution using L-tartaric acid followed by acidification with HCl to isolate high-purity L-2-aminobutanamide hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route represents a strategic opportunity to optimize cost structures and mitigate supply risks. The transition from hazardous halogenated reagents to commodity chemicals like propionaldehyde and ammonia fundamentally alters the cost profile of the intermediate. By eliminating the need for expensive specialty reagents and complex waste treatment systems required for bromine disposal, manufacturers can achieve substantial cost savings in raw material acquisition and environmental compliance. Furthermore, the simplified equipment requirements mean that production can be scaled using standard glass-lined or stainless steel reactors found in most multipurpose chemical facilities, reducing capital expenditure barriers and accelerating time-to-market for new production lines.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of high-cost raw materials with inexpensive commodity chemicals. Unlike methionine-based routes which rely on amino acids with volatile market prices, propionaldehyde is a bulk chemical with stable availability and pricing. Additionally, the elimination of bromine removes the significant overhead costs associated with handling hazardous materials, including specialized storage, personal protective equipment, and rigorous effluent treatment for halogenated waste. The high yield and purity achieved directly translate to reduced solvent usage per kilogram of product, as fewer recrystallization cycles are needed to meet specification, further driving down the variable cost of goods sold.
- Enhanced Supply Chain Reliability: Supply continuity is significantly bolstered by the use of widely available starting materials that are not subject to the same geopolitical or logistical constraints as specialty fine chemicals. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor fluctuations in utility supply compared to high-pressure alternatives. This resilience ensures that pharmaceutical companies can maintain consistent inventory levels of this critical intermediate, preventing bottlenecks in the downstream synthesis of Levetiracetam. The ability to source raw materials from multiple global suppliers reduces dependency on single-source vendors, thereby de-risking the entire procurement strategy.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route offers a markedly greener profile. The absence of toxic bromine gas and the reduction of hazardous waste streams simplify the regulatory compliance landscape, facilitating faster permitting and inspection approvals in stringent jurisdictions. The process is inherently scalable, having been designed with industrial feasibility in mind, allowing for seamless transition from kilogram-scale development to multi-ton commercial production. This scalability ensures that as market demand for Levetiracetam grows, the supply of its key intermediate can be expanded rapidly without requiring fundamental changes to the process chemistry or significant new infrastructure investments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this technology for integration into their existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis route over traditional bromination methods?
A: This route eliminates the use of hazardous bromine and avoids the generation of corrosive HBr gas, significantly improving workshop safety and reducing environmental compliance costs associated with halogenated waste treatment.
Q: How is high optical purity achieved in this process?
A: High optical purity exceeding 99.5% is achieved through a precise chiral resolution step using L-tartaric acid, which effectively separates the desired L-enantiomer from the racemic mixture formed during the initial synthesis.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common industrial raw materials like propionaldehyde and operates under moderate temperature and pressure conditions, making it highly scalable for medium to large-sized enterprises without requiring specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-2-Aminobutanamide Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving medications like Levetiracetam. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of this patent are fully realized in practical manufacturing settings. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of L-2-aminobutanamide hydrochloride meets the exacting standards required by global regulatory bodies. Our commitment to quality assurance guarantees that our clients receive a product with consistent optical purity and minimal impurity profiles, safeguarding the integrity of their final API.
We invite pharmaceutical partners to engage with our technical procurement team to discuss how this optimized synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the potential economic benefits specific to your operational context. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can support your supply chain goals and drive efficiency in your antiepileptic drug manufacturing programs.
