Industrial Synthesis of (R)-3-Amino Piperidine Hydrochloride via D-Glutamic Acid Route
Introduction to Advanced Chiral Synthesis
The pharmaceutical industry's relentless pursuit of potent antidiabetic agents has placed significant demand on the supply chain for high-quality chiral intermediates, specifically (R)-3-amino piperidine hydrochloride. This critical building block is essential for the synthesis of Dipeptidyl Peptidase-IV (DPP-IV) inhibitors, a class of drugs that has revolutionized diabetes management. Patent CN103864674A discloses a groundbreaking five-step synthetic methodology that utilizes D-glutamic acid as a renewable, chiral starting material. This approach represents a paradigm shift from traditional resolution-based methods, offering a streamlined pathway that preserves stereochemistry while drastically simplifying the operational complexity. By leveraging abundant fermentation-derived precursors, this technology addresses the twin challenges of cost-efficiency and environmental sustainability, making it an ideal candidate for reliable pharmaceutical intermediate supplier networks seeking to optimize their API manufacturing portfolios.
The significance of this patent lies not merely in the chemical transformation but in its holistic approach to process chemistry. Traditional routes often suffer from low atom economy due to the discard of unwanted enantiomers or the use of hazardous reagents requiring cryogenic conditions. In contrast, the disclosed method operates under mild thermal conditions, typically ranging from 0°C to 30°C, utilizing common solvents like methanol, ethanol, and acetonitrile. This accessibility of reagents and conditions lowers the barrier to entry for commercial scale-up of complex chiral amines, ensuring that production facilities can maintain high throughput without necessitating exotic infrastructure. The result is a robust, scalable process capable of delivering high-purity intermediates consistent with stringent global regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of (R)-3-amino piperidine derivatives has been plagued by inefficiencies inherent to racemic synthesis and subsequent resolution. Conventional Route One, for instance, relies on the hydrogenation and fractionation of racemic 3-amino piperidine, a process that inherently caps the maximum theoretical yield at 50% unless dynamic kinetic resolution is employed, which adds further complexity and cost. Other historical pathways, such as those utilizing nicotinamide or ethyl nipecotate, involve cumbersome sequences including Hofmann degradation and multiple protection-deprotection cycles that accumulate impurities and reduce overall yield. Furthermore, many of these legacy processes require harsh reaction conditions, such as low-temperature cryogenic setups or the use of pyrophoric reducing agents, which pose significant safety risks and increase capital expenditure for specialized equipment. These factors collectively contribute to a volatile supply chain and elevated costs for cost reduction in API manufacturing.
The Novel Approach
The methodology outlined in CN103864674A circumvents these bottlenecks by employing a chiral pool strategy starting from D-glutamic acid. This naturally occurring amino acid provides the necessary stereochemical foundation, eliminating the need for resolution entirely. The synthetic sequence is elegantly concise, proceeding through esterification, reduction, activation, cyclization, and final deprotection. 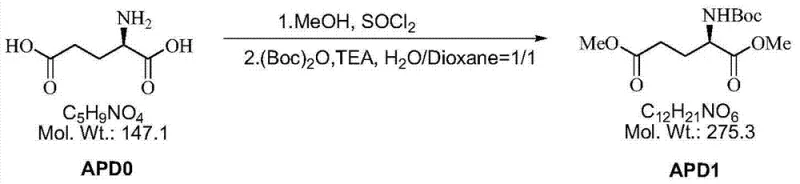 As illustrated in the initial step, the conversion of D-glutamic acid to the protected diester APD1 is achieved with exceptional efficiency, setting the stage for downstream transformations. By avoiding the generation of unwanted enantiomers, this route effectively doubles the material throughput compared to racemic splits. Moreover, the use of sodium borohydride for reduction instead of lithium aluminum hydride enhances operational safety, while the cyclization step utilizes aqueous ammonia, a benign and inexpensive reagent, thereby aligning with green chemistry principles.
As illustrated in the initial step, the conversion of D-glutamic acid to the protected diester APD1 is achieved with exceptional efficiency, setting the stage for downstream transformations. By avoiding the generation of unwanted enantiomers, this route effectively doubles the material throughput compared to racemic splits. Moreover, the use of sodium borohydride for reduction instead of lithium aluminum hydride enhances operational safety, while the cyclization step utilizes aqueous ammonia, a benign and inexpensive reagent, thereby aligning with green chemistry principles.
Mechanistic Insights into Intramolecular Cyclization
The cornerstone of this synthetic route is the formation of the piperidine ring, a transformation that dictates the structural integrity and purity of the final API intermediate. The mechanism involves the conversion of the linear diol APD2 into the dimesylate APD3, followed by an intramolecular nucleophilic substitution. 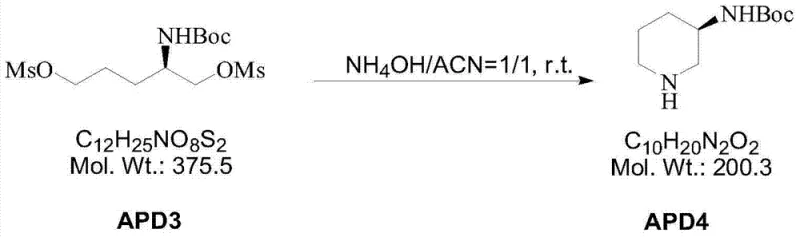 In this critical step, the primary amine source (ammonium hydroxide) attacks the electrophilic carbon centers activated by the mesylate leaving groups. The reaction is driven by the entropic favorability of forming a six-membered ring, which is thermodynamically stable. The choice of acetonitrile as the solvent facilitates this substitution by solubilizing the organic substrate while allowing the aqueous ammonia to participate effectively. This mechanistic pathway is highly selective, minimizing the formation of polymeric byproducts or intermolecular coupling species that often plague linear amine syntheses. The preservation of the chiral center throughout this sequence is paramount, and the mild conditions (30°C) ensure that no racemization occurs at the alpha-position, guaranteeing high enantiomeric excess in the final product.
In this critical step, the primary amine source (ammonium hydroxide) attacks the electrophilic carbon centers activated by the mesylate leaving groups. The reaction is driven by the entropic favorability of forming a six-membered ring, which is thermodynamically stable. The choice of acetonitrile as the solvent facilitates this substitution by solubilizing the organic substrate while allowing the aqueous ammonia to participate effectively. This mechanistic pathway is highly selective, minimizing the formation of polymeric byproducts or intermolecular coupling species that often plague linear amine syntheses. The preservation of the chiral center throughout this sequence is paramount, and the mild conditions (30°C) ensure that no racemization occurs at the alpha-position, guaranteeing high enantiomeric excess in the final product.
Impurity control is another vital aspect of this mechanism, particularly during the activation and reduction phases. The reduction of the diester APD1 to the diol APD2 using sodium borohydride must be carefully monitored to prevent over-reduction or incomplete conversion, which could lead to hydroxy-ester impurities that complicate downstream purification. The patent specifies a reflux protocol in ethanol that balances reaction kinetics with selectivity. Similarly, the mesylation step requires precise temperature control at 0°C initially to manage the exotherm and prevent side reactions such as elimination. By adhering to these specific mechanistic parameters, manufacturers can achieve a clean impurity profile, reducing the burden on downstream purification units like chromatography or recrystallization. This level of control is essential for producing high-purity DPP-IV inhibitor intermediate suitable for direct use in sensitive coupling reactions.
How to Synthesize (R)-3-Amino Piperidine Hydrochloride Efficiently
Executing this synthesis on a commercial scale requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the protection and esterification of D-glutamic acid, followed by a controlled reduction to establish the carbon backbone. Subsequent activation of the hydroxyl groups prepares the molecule for the pivotal ring-closing event. The final steps involve the removal of the protecting groups and salt formation to stabilize the product. Each stage has been refined to maximize yield and minimize waste, utilizing standard unit operations found in modern multipurpose chemical plants. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Protect D-Glutamic acid with Boc group and esterify to form N-Boc-D-Glu dimethyl ester (APD1).
- Reduce the diester APD1 using Sodium Borohydride in ethanol to obtain the diol APD2.
- Activate the hydroxyl groups of APD2 using Methanesulfonyl chloride (MsCl) to form the dimesylate APD3.
- Perform intramolecular cyclization of APD3 using ammonium hydroxide to form the piperidine ring (APD4).
- Remove the Boc protecting group and form the hydrochloride salt using Thionyl Chloride in methanol to yield the final product APD5.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic route offers compelling advantages that directly impact the bottom line and supply security. The shift from resolution-based methods to a chiral pool synthesis fundamentally alters the cost structure of the intermediate. By utilizing D-glutamic acid, a commodity chemical produced via large-scale fermentation, the reliance on expensive, specialized chiral catalysts or resolving agents is eliminated. This transition results in substantial cost savings in raw material acquisition and reduces the volatility associated with sourcing niche reagents. Furthermore, the increased overall yield—driven by the retention of chirality rather than its separation—means that less starting material is required to produce the same amount of final product, effectively lowering the cost per kilogram of the active pharmaceutical ingredient.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the use of inexpensive reagents like sodium borohydride and thionyl chloride significantly lower the variable costs of production. Unlike processes requiring precious metal catalysts or cryogenic cooling, this route operates at near-ambient temperatures, reducing energy consumption and utility costs. The simplified workflow also decreases labor hours and reactor occupancy time, allowing for higher asset utilization rates. These factors combine to create a highly competitive cost structure that supports margin expansion for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Sourcing D-glutamic acid is far more secure than relying on complex chiral building blocks that may have single-source suppliers. The robustness of the chemistry, which tolerates standard industrial solvents and equipment, ensures that production can be easily transferred between different manufacturing sites without significant re-validation. This flexibility mitigates the risk of supply disruptions caused by equipment failure or regional logistical issues. Additionally, the high purity of the intermediate reduces the likelihood of batch failures due to specification deviations, ensuring a steady flow of material to the formulation stage.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial feasibility in mind. The avoidance of heavy metals and the use of recyclable solvents like ethanol and methanol simplify waste treatment and disposal, aligning with increasingly stringent environmental regulations. The aqueous workups and straightforward extractions minimize the generation of hazardous waste streams. This environmental compatibility not only reduces compliance costs but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs. The answers are derived directly from the experimental data and process descriptions provided in the patent literature, ensuring accuracy and relevance.
Q: What are the advantages of using D-Glutamic acid over racemic resolution?
A: Using D-Glutamic acid as a chiral pool starting material eliminates the need for resolution steps, theoretically doubling the yield compared to racemic synthesis which loses 50% of the material. It also ensures high optical purity from the outset.
Q: Is Sodium Borohydride safe for large-scale reduction of diesters?
A: Yes, Sodium Borohydride is significantly safer and easier to handle on an industrial scale compared to Lithium Aluminum Hydride (LiAlH4). The patent specifies reflux conditions in ethanol, which are manageable and safe for commercial production.
Q: How is the piperidine ring formed in this specific pathway?
A: The ring is formed via an intramolecular nucleophilic substitution. The dimesylate intermediate (APD3) reacts with ammonia, where the nitrogen attacks one of the activated carbon centers, closing the six-membered ring to form the piperidine structure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-3-Amino Piperidine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has extensively analyzed the route described in CN103864674A and possesses the expertise to implement this chemistry at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of (R)-3-amino piperidine hydrochloride meets the exacting standards required for GMP API synthesis.
We invite you to collaborate with us to leverage this efficient synthetic technology for your projects. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in driving innovation and efficiency in your pharmaceutical supply chain.
