Advanced Synthesis of Brigatinib Intermediates: Technical Breakthroughs and Commercial Scalability
Advanced Synthesis of Brigatinib Intermediates: Technical Breakthroughs and Commercial Scalability
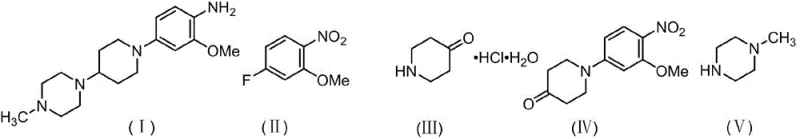
The pharmaceutical landscape for oncology treatments continues to evolve rapidly, driven by the demand for potent Tyrosine Kinase inhibitors such as Brigatinib. Central to the efficient production of this life-saving medication is the availability of high-quality key intermediates. Patent CN113024454A discloses a robust and economically viable synthesis method for 2-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)aniline, a critical building block in the Brigatinib value chain. This technical insight report analyzes the novel methodology, contrasting it with historical approaches to highlight its superiority in yield, purity, and operational simplicity. By leveraging a streamlined two-step sequence involving nucleophilic aromatic substitution and catalytic hydrogenation, this process addresses long-standing bottlenecks in intermediate manufacturing. For R&D directors and procurement specialists, understanding these mechanistic nuances is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
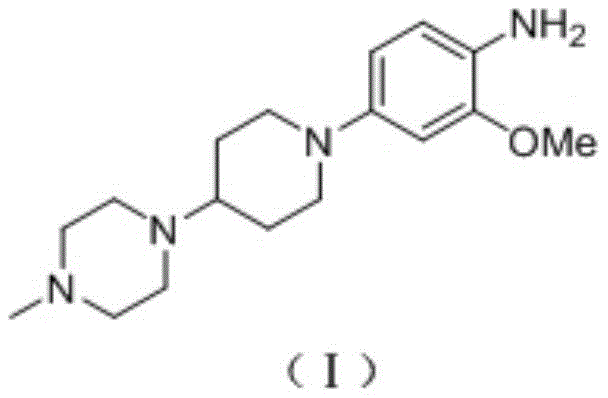
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this complex aniline derivative has been plagued by inefficient multi-step sequences and the reliance on hazardous or costly reagents. Early methodologies, such as those disclosed in WO 2004080980, utilized a reductive amination strategy employing sodium triacetoxyborohydride. While chemically feasible, this approach suffers from a low overall yield of approximately 34.5% and utilizes a reducing agent that is prohibitively expensive for large-scale industrial application and cannot be recycled. Furthermore, alternative routes reported in WO 2017076355A1 relied on zinc powder and ammonium chloride for nitro group reduction. This generates substantial amounts of heavy metal waste, creating significant environmental compliance burdens and disposal costs that negatively impact the total cost of ownership. Another variation found in WO2017088784 employed Leuckart-Wallach conditions with formic acid, which introduces safety risks associated with high-temperature formic acid handling and often results in difficult-to-remove formamide byproducts.
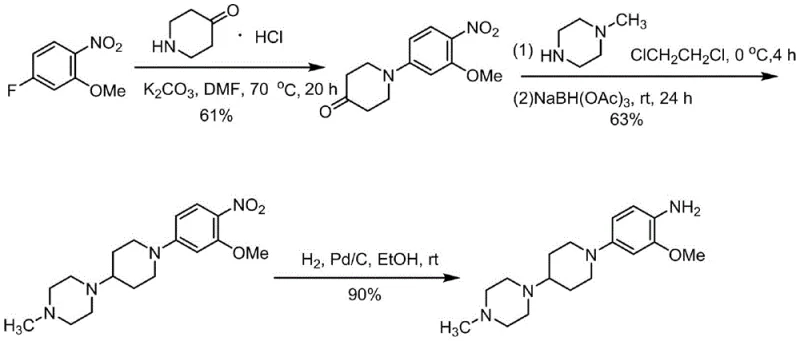
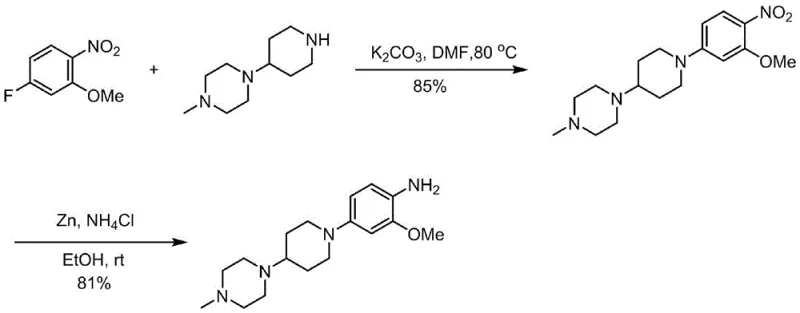
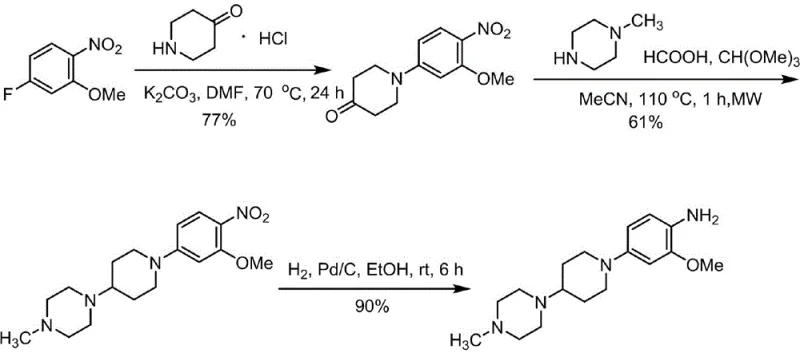
The Novel Approach
In stark contrast to these legacy methods, the technology protected under CN113024454A introduces a highly efficient pathway that merges reductive amination and nitro reduction into a single catalytic hydrogenation step. The process initiates with the formation of a ketone intermediate via nucleophilic substitution, followed by a direct reaction with N-methylpiperazine under hydrogen pressure. This telescoped approach eliminates the need for stoichiometric chemical reducing agents entirely. By utilizing molecular sieves as dehydrating agents during the imine formation and subsequently employing a reusable Pd/C catalyst for hydrogenation, the process achieves exceptional atom economy. The result is a streamlined operation that not only boosts the total yield significantly but also drastically simplifies the work-up procedure, making it ideally suited for cost reduction in API manufacturing where efficiency and waste minimization are paramount.
Mechanistic Insights into Catalytic Hydrogenation and Reductive Amination
The core innovation of this synthesis lies in the precise orchestration of the second step, where reductive amination and nitro reduction occur concurrently or sequentially under hydrogenation conditions. Initially, the ketone intermediate reacts with N-methylpiperazine to form an imine or iminium ion species. The presence of a dehydrating agent, specifically 4A molecular sieves, is critical here as it shifts the equilibrium towards imine formation by sequestering the water byproduct. Once the imine is formed, the introduction of hydrogen gas in the presence of a 10% Pd/C catalyst facilitates the reduction of both the C=N double bond of the imine and the nitro group on the aromatic ring. This dual-reduction capability is mechanistically elegant, as it prevents the isolation of unstable intermediates and ensures that the final amine product is formed directly. The choice of solvent, preferably tetrahydrofuran (THF), provides the necessary solubility for both organic substrates while maintaining stability under hydrogenation pressures of 0.6-1.0 MPa.
Impurity control is another sophisticated aspect of this mechanism. The initial nucleophilic substitution step utilizes tetrabutylammonium bromide (TBAB) as a phase transfer catalyst in a DMF/Water or DMSO/Water system. This specific solvent mixture enhances the nucleophilicity of the piperidone nitrogen while suppressing side reactions such as hydrolysis of the methoxy group. The subsequent crystallization of the intermediate ketone using n-hexane pulping effectively removes unreacted starting materials and inorganic salts before the hydrogenation step begins. This rigorous purification prior to the final catalytic step ensures that the Pd/C catalyst is not poisoned by sulfur or other heteroatom impurities, thereby maintaining high catalytic activity and allowing for the reported 99.8% purity of the final aniline product.
How to Synthesize 2-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)aniline Efficiently
The execution of this synthesis requires careful attention to reaction parameters, particularly temperature and pressure control during the hydrogenation phase. The process is designed to be scalable, moving seamlessly from laboratory glassware to industrial reactors. Operators must ensure that the dehydration step is complete before introducing hydrogen to maximize the conversion of the imine intermediate. The following guide outlines the standardized operational procedure derived from the patent examples, ensuring reproducibility and safety.
- React 5-fluoro-2-nitrobenzyl ether with piperidone hydrochloride using a phase transfer catalyst and base in a polar solvent system at 75-80°C to form the nitro-ketone intermediate.
- Perform reductive amination with N-methylpiperazine in the presence of a dehydrating agent, followed by catalytic hydrogenation using Pd/C at 50-55°C and 0.6-1.0 MPa pressure.
- Filter off the catalyst and dehydrating agent, concentrate the filtrate, and recrystallize the crude product using a petroleum ether-ethyl acetate mixture to obtain high-purity target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere chemical elegance. The elimination of expensive stoichiometric reagents like sodium triacetoxyborohydride directly translates to a lower bill of materials. Additionally, the ability to recycle the palladium catalyst multiple times reduces the dependency on precious metal markets, stabilizing input costs against volatility. The process operates under relatively mild conditions compared to high-temperature formic acid reductions, lowering energy consumption and extending equipment lifespan. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous demands of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The most significant economic driver is the removal of non-recyclable reducing agents. By replacing sodium triacetoxyborohydride with catalytic hydrogen, the process avoids the high unit cost of boron reagents. Furthermore, the recovery and reuse of the Pd/C catalyst for multiple batches mean that the effective cost of the catalyst per kilogram of product is drastically reduced. The simplified work-up, which involves filtration and crystallization rather than complex extractions or chromatography, reduces solvent usage and labor hours, leading to substantial cost savings in the overall production budget.
- Enhanced Supply Chain Reliability: The raw materials required for this process, such as 5-fluoro-2-nitrobenzyl ether and piperidone hydrochloride, are commodity chemicals with established global supply chains. Unlike specialized reagents that may have long lead times or single-source restrictions, these inputs are readily available from multiple reliable pharmaceutical intermediate suppliers. The robustness of the reaction conditions also means that production schedules are less likely to be disrupted by minor variations in raw material quality, ensuring consistent on-time delivery for downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental perspective, this method represents a significant improvement in green chemistry metrics. The avoidance of zinc powder eliminates the generation of heavy metal sludge, a major liability in waste treatment. The use of aqueous solvent mixtures in the first step reduces the load of organic volatile compounds (VOCs). These features facilitate easier regulatory approval and reduce the environmental footprint of the manufacturing site, aligning with the sustainability goals of modern chemical enterprises and simplifying the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity for technical teams evaluating this route for adoption.
Q: What are the primary advantages of this new synthesis route over prior art methods?
A: The new method eliminates the need for expensive stoichiometric reducing agents like sodium triacetoxyborohydride and avoids the environmental hazards associated with zinc powder reduction. It achieves higher overall yields (up to 90% in step 1) and allows for catalyst recycling.
Q: Can the palladium catalyst be reused in this process?
A: Yes, the patent specifies that the Pd/C catalyst can be recovered and reused for up to three batches without significant loss in yield, provided the filtration and washing steps are performed correctly.
Q: What is the expected purity of the final brigatinib intermediate?
A: The optimized process described in the patent consistently achieves a purity of 99.8% (by HPLC area normalization) after recrystallization, meeting stringent requirements for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-methoxy-4-(4-(4-methylpiperazin-1-yl)piperidin-1-yl)aniline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs like Brigatinib depends on the uninterrupted supply of high-purity intermediates. Our technical team has thoroughly analyzed the pathways described in CN113024454A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch meets the 99.8% purity benchmark required for GMP API synthesis. Our commitment to quality assurance guarantees that your supply chain remains robust and compliant with international regulatory standards.
We invite you to collaborate with us to optimize your sourcing strategy for this critical intermediate. By leveraging our expertise in catalytic hydrogenation and process optimization, we can offer a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines and quality expectations.
