Scalable Production of Methyl (methylamino)squarate via Novel One-Pot Esterification-Aminolysis
Scalable Production of Methyl (methylamino)squarate via Novel One-Pot Esterification-Aminolysis
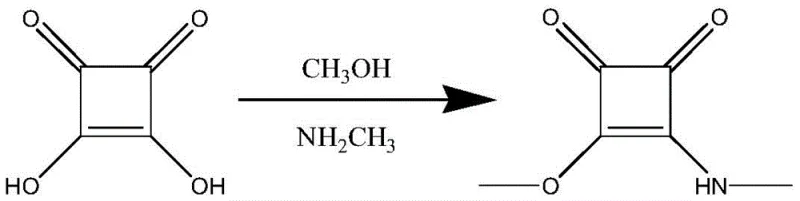
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational safety and cost efficiency. A significant breakthrough in this domain is detailed in patent CN111349014A, which discloses an innovative preparation method for Methyl (methylamino)squarate, a critical building block in the synthesis of various bioactive molecules and functional materials. Unlike conventional pathways that rely on hazardous ether solvents and unstable intermediates, this novel approach utilizes a direct, one-pot cascade reaction starting from squaric acid, methanol, and methylamine. By integrating esterification and aminolysis into a streamlined sequence, the technology effectively mitigates the risks associated with volatile organic compounds while simultaneously boosting overall process yield. For R&D directors and procurement strategists, this represents a pivotal shift towards greener, more economically viable manufacturing protocols that align with modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of methyl (methylamino)squarate has been plagued by significant technical and safety hurdles inherent to the traditional two-step methodology. The legacy process typically necessitates the initial preparation of dimethyl squarate, which is subsequently reacted with a methylamine solution in tetrahydrofuran (THF). This reliance on THF introduces severe volatility and flammability risks, demanding expensive explosion-proof infrastructure and rigorous solvent recovery systems to maintain workplace safety. Furthermore, the final purification stage traditionally employs methyl ethyl ketone (MEK) as a recrystallization solvent; however, MEK is a strictly controlled chemical precursor in many jurisdictions, creating substantial regulatory bottlenecks and supply chain friction. Additionally, the intermediate dimethyl squarate is prone to decomposition, leading to inconsistent reaction yields and a complex impurity profile that requires costly downstream purification efforts to meet pharmaceutical grade specifications.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111349014A revolutionizes the production landscape by adopting a direct, one-step synthesis strategy that bypasses the isolation of unstable intermediates entirely. This advanced route initiates with the reflux of squaric acid in methanol, utilizing trimethyl orthoformate as an effective water scavenger to drive the equilibrium towards the desired ester formation without generating excessive waste. By carefully controlling the feeding sequence and reaction temperature, the process seamlessly transitions into the aminolysis phase upon the addition of a methylamine methanol solution. This integration not only eliminates the need for hazardous THF and controlled MEK solvents but also significantly simplifies the operational workflow. The result is a robust, scalable process that delivers superior yields—demonstrated up to 80% in specific embodiments—while drastically reducing the environmental footprint and operational complexity associated with solvent handling and disposal.
Mechanistic Insights into the Cascade Esterification-Aminolysis
The core innovation of this synthesis lies in the clever manipulation of reaction equilibria through the use of trimethyl orthoformate within a methanol medium. In the initial phase, squaric acid undergoes esterification where the orthoformate acts as a dehydrating agent, reacting with the water byproduct to form methyl formate and methanol, thereby preventing the hydrolysis of the newly formed ester bonds. This drives the reaction to completion, ensuring a high concentration of the reactive dimethyl squarate intermediate in situ without the need for isolation. Following the removal of low-boiling solvents via distillation, the system is primed for nucleophilic attack. The subsequent addition of methylamine at controlled temperatures (0-50°C) facilitates a selective aminolysis, where the amine nucleophile displaces one of the methoxy groups. This selectivity is crucial, as it prevents the formation of bis-amide byproducts, ensuring the final structure retains the necessary asymmetry for downstream applications.
Controlling the impurity profile in this cascade reaction is achieved through precise thermal management and stoichiometric balance. The patent specifies a reflux period ranging from 24 to 72 hours, or a split-stage reflux, which ensures complete conversion of the starting squaric acid before the introduction of the amine. If the amine were introduced prematurely or at elevated temperatures, competitive reactions could lead to polymerization or the formation of insoluble tars. By maintaining the aminolysis phase at normal temperature for 1 to 10 hours, the kinetic energy is sufficient for the desired substitution but low enough to suppress degradation pathways. Furthermore, the choice of recrystallization solvents—ranging from simple alcohols to chloroalkanes—allows for the selective precipitation of the target molecule while leaving polar impurities and unreacted salts in the mother liquor, resulting in a final product with purity levels reaching 98% as evidenced in the patent examples.
How to Synthesize Methyl (methylamino)squarate Efficiently
Implementing this synthesis requires strict adherence to the sequential addition of reagents and thermal profiles to maximize the benefits of the one-pot design. The process begins with charging a reaction vessel with squaric acid, methanol, and trimethyl orthoformate, followed by a prolonged reflux to ensure full esterification. Once the low-boiling components are distilled off, the temperature must be carefully moderated before the dropwise addition of the methylamine solution to manage the exotherm.
- Reflux squaric acid with methanol and trimethyl orthoformate for 24-72 hours to drive esterification and remove water.
- Distill off low-boiling solvents to concentrate the mixture, then cool to 0-50°C before adding methylamine methanol solution.
- React at ambient temperature for 1-10 hours, followed by distillation, cooling crystallization, and filtration to isolate the crude product.
- Recrystallize the crude product using safe solvents like alcohols or chloroalkanes to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers profound strategic advantages that extend far beyond simple yield improvements. The most immediate impact is the drastic simplification of the raw material portfolio; by sourcing stable squaric acid instead of pre-synthesized dimethyl squarate, companies can reduce dependency on specialized intermediate suppliers and mitigate supply disruption risks. Moreover, the elimination of tetrahydrofuran and methyl ethyl ketone from the process flow removes the burden of managing highly regulated, controlled substances, thereby streamlining logistics, reducing storage compliance costs, and accelerating the speed of material throughput through the facility. This transition to safer, commodity-grade solvents like methanol creates a more resilient supply chain that is less susceptible to regulatory shocks and price volatility associated with specialty solvents.
- Cost Reduction in Manufacturing: The economic implications of this process are substantial, primarily driven by the consolidation of unit operations and the removal of expensive solvent recovery loops. By avoiding the isolation of the dimethyl squarate intermediate, the manufacturer saves on filtration, drying, and re-dissolution steps, which directly translates to lower labor and utility consumption. Additionally, the replacement of controlled solvents with inexpensive methanol and recyclable chloroalkanes significantly lowers the variable cost of goods sold (COGS). The higher reaction yield further amplifies these savings by maximizing the output per batch, ensuring that raw material spend is converted into saleable product with minimal waste generation.
- Enhanced Supply Chain Reliability: From a logistics perspective, the stability of the starting material, squaric acid, ensures a consistent and reliable feedstock supply compared to the more sensitive dimethyl squarate. The process tolerance for a wider range of recrystallization solvents, including esters and various alcohols, provides procurement teams with the flexibility to source materials from multiple vendors based on regional availability and pricing. This flexibility acts as a buffer against market fluctuations, ensuring continuous production schedules even when specific solvent supplies are constrained, thereby guaranteeing on-time delivery to downstream pharmaceutical clients.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly superior, aligning with global trends towards green chemistry and sustainable manufacturing. The absence of volatile ethers reduces the risk of atmospheric emissions and workplace exposure incidents, lowering insurance premiums and environmental compliance overheads. The process generates fewer hazardous waste streams, simplifying effluent treatment and disposal protocols. This clean operational footprint facilitates easier permitting for capacity expansion, allowing manufacturers to scale from pilot plants to multi-ton commercial production with fewer regulatory hurdles and a reduced carbon footprint.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this synthesis route, derived directly from the experimental data and comparative analysis provided in the patent documentation. These answers are designed to clarify the operational parameters and quality control measures necessary for successful technology transfer.
Q: Why is the new method superior to the traditional THF-based process?
A: The traditional method utilizes tetrahydrofuran (THF) and methyl ethyl ketone (MEK), which are volatile, flammable, and strictly controlled chemicals. The novel patent CN111349014A replaces these with methanol and safer recrystallization solvents, significantly improving operational safety and regulatory compliance while enhancing yield.
Q: How does the process control impurities during synthesis?
A: By employing trimethyl orthoformate as a dehydrating agent during the initial reflux, the reaction equilibrium is driven towards the ester intermediate, minimizing hydrolysis byproducts. Furthermore, the sequential addition of methylamine at controlled temperatures (0-50°C) prevents over-reaction and decomposition, ensuring a cleaner impurity profile.
Q: What are the scalability advantages of this route?
A: This one-pot cascade reaction eliminates the need to isolate unstable intermediates like dimethyl squarate. Starting directly from stable squaric acid simplifies the workflow, reduces unit operations, and allows for easier scale-up from kilogram to multi-ton production without complex solvent exchange steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl (methylamino)squarate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN111349014A are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific thermal and solvent requirements of this cascade reaction, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for pharmaceutical intermediates.
We invite forward-thinking R&D and procurement leaders to collaborate with us to optimize their supply chains using this superior technology. By leveraging our process engineering capabilities, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring your project moves forward with the highest level of confidence and efficiency.
