Industrial Scale-Up Of Alpha-Damascenone Via Optimized Catalytic Cyclization
Industrial Scale-Up Of Alpha-Damascenone Via Optimized Catalytic Cyclization
The global demand for high-quality rose-note fragrances continues to drive innovation in the synthesis of alpha-damascenone, a critical component in modern perfumery. Patent CN103058841A introduces a robust, three-step synthetic pathway that addresses the longstanding challenges of yield and cost associated with traditional manufacturing methods. By utilizing dehydrolinalool as a readily available starting material and employing a sequence of copper-catalyzed alkylation, molybdenum-mediated rearrangement, and solid-acid cyclization, this technology offers a compelling solution for reliable synthetic flavors & fragrances supplier networks seeking to optimize their production capabilities. The strategic integration of recyclable solvents and mild reaction conditions underscores a commitment to both economic efficiency and environmental sustainability, positioning this method as a benchmark for future industrial standards in fine chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-damascenone has been plagued by inefficient routes that struggle to meet the rigorous demands of modern industrial production. The seminal work by Karl H. Schulte-Elte in 1975, while foundational, established a pathway characterized by low overall yields and complex operational requirements that rendered it economically unviable for large-scale application. Traditional methods often suffer from harsh reaction conditions that promote the formation of undesirable by-products, necessitating expensive and time-consuming purification steps to achieve the requisite purity levels for fragrance applications. Furthermore, the reliance on non-recyclable solvents and stoichiometric reagents in older protocols contributes to excessive waste generation, creating significant environmental liabilities and inflating the cost of goods sold. These structural inefficiencies have long hindered the ability of manufacturers to provide cost reduction in synthetic flavors & fragrances manufacturing without compromising on quality or supply reliability.
The Novel Approach
In stark contrast to these legacy limitations, the methodology outlined in patent CN103058841A presents a streamlined, high-efficiency route that fundamentally redefines the production landscape for this valuable aroma chemical. The novel approach leverages a telescoped strategy where dehydrolinalool is first coupled with 3-bromopropene under mild basic conditions to form a key alkynol intermediate with exceptional conversion rates. This is followed by a sophisticated isomerization step utilizing a molybdenum catalyst system to generate pseudodamascenone, which is subsequently cyclized under controlled acidic conditions to yield the final alpha-damascenone product. The entire process is engineered for simplicity and scalability, utilizing common solvents like toluene and methanol that can be easily recovered and reused, thereby closing the loop on material usage. This holistic optimization ensures that the commercial scale-up of complex fragrance intermediates becomes not only feasible but highly profitable, offering a distinct competitive advantage in the marketplace.
Mechanistic Insights into Copper and Molybdenum Catalyzed Transformations
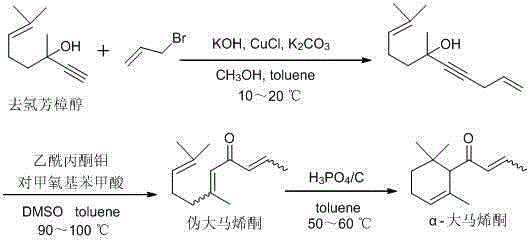
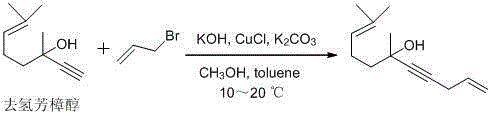
The success of this synthetic route hinges on the precise orchestration of catalytic cycles that govern each transformation step, beginning with the copper-catalyzed alkylation of dehydrolinalool. In this initial phase, the presence of CuCl alongside potassium hydroxide and potassium carbonate facilitates a nucleophilic substitution reaction where the terminal alkyne of dehydrolinalool attacks the allylic bromide. The careful control of temperature between 10-20°C is critical here, as it suppresses potential side reactions such as polymerization or elimination, ensuring that the formation of 6,10-dimethylundecane-1,9-dien-4-yn-6-ol proceeds with a remarkable yield of up to 98%. This high selectivity is paramount for R&D directors focused on purity, as it minimizes the burden on downstream purification units and establishes a clean foundation for subsequent transformations. The mechanistic efficiency of this step demonstrates how optimizing catalyst loading and base strength can dramatically improve atom economy in fine chemical synthesis.
Following the alkylation, the conversion to pseudodamascenone involves a complex rearrangement driven by a molybdenum acetylacetonate catalyst in the presence of p-methoxybenzoic acid. This step operates at elevated temperatures of 90-100°C in a toluene and DMSO solvent system, promoting the necessary skeletal reorganization to form the conjugated enone structure. The role of the molybdenum center is to coordinate with the alkyne and facilitate the migration of bonds, a process that requires precise thermal management to avoid over-isomerization or decomposition. Finally, the cyclization to alpha-damascenone is achieved using activated carbon-supported phosphoric acid, a heterogeneous catalyst that offers the dual benefits of easy separation and tunable acidity. Operating at a mild 50-60°C, this solid acid catalyst promotes the intramolecular cyclization while minimizing the formation of polymeric tars, a common issue with liquid strong acids. This mechanistic clarity provides a robust framework for troubleshooting and process optimization during technology transfer.
How to Synthesize Alpha-Damascenone Efficiently
Implementing this synthesis protocol requires strict adherence to the specified reaction parameters to maximize yield and safety across all three stages. The process begins with the preparation of the alkynol intermediate, followed by the catalytic rearrangement to the pseudo-form, and concludes with the acid-catalyzed cyclization. Each stage demands specific attention to solvent ratios, catalyst loading, and temperature gradients to ensure reproducibility. For detailed operational procedures, including exact reagent quantities and workup protocols, please refer to the standardized synthesis guide below which encapsulates the critical process parameters defined in the patent documentation.
- React dehydrolinalool with 3-bromopropene in toluene/methanol using KOH, K2CO3, and CuCl catalysts at 10-20°C to form the alkynol intermediate.
- Convert the alkynol intermediate to pseudodamascenone using molybdenum acetylacetonate and p-methoxybenzoic acid in toluene/DMSO at 90-100°C.
- Cyclize pseudodamascenone to alpha-damascenone using activated carbon-supported phosphoric acid in toluene at 50-60°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical conversion. The primary value proposition lies in the substantial cost savings achieved through the elimination of expensive, single-use reagents and the implementation of a closed-loop solvent recovery system. By utilizing toluene and methanol, which are commodity chemicals with stable pricing and widespread availability, manufacturers can insulate themselves from the volatility associated with specialized solvents. Furthermore, the high yields observed in each step—particularly the near-quantitative conversion in the first alkylation step—mean that less raw material is required to produce the same amount of finished goods, directly improving the margin profile. This efficiency translates into a more resilient supply chain capable of meeting fluctuating market demands without the risk of raw material bottlenecks or excessive inventory holding costs.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the workflow and the high efficiency of the catalyst systems employed. Unlike traditional routes that may require stoichiometric amounts of costly reagents or precious metals that are difficult to recover, this method utilizes earth-abundant metals like copper and molybdenum in catalytic quantities. The ability to recycle the bulk solvents, toluene and methanol, further reduces the operational expenditure related to waste disposal and fresh solvent procurement. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower overall carbon footprint and utility cost. These factors combine to create a lean manufacturing model that significantly lowers the cost of goods sold, allowing for more competitive pricing strategies in the global fragrance market.
- Enhanced Supply Chain Reliability: Supply chain stability is critically dependent on the availability of starting materials and the robustness of the production process. Dehydrolinalool and 3-bromopropene are commercially available in large quantities, ensuring that production schedules are not disrupted by raw material shortages. The process tolerance, evidenced by the broad temperature ranges that still yield acceptable results (e.g., 50-60°C for the final step), provides operational flexibility that mitigates the risk of batch failures due to minor equipment fluctuations. This reliability is essential for maintaining consistent delivery timelines to downstream customers, fostering long-term partnerships based on trust and dependability. Moreover, the simplified purification requirements mean that production throughput can be increased without proportional increases in processing time, effectively expanding capacity without major capital investment.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify, the environmental profile of a chemical process becomes a key determinant of its long-term viability. This synthesis route excels in scalability because it avoids the use of hazardous reagents that require special handling or disposal protocols. The use of a heterogeneous catalyst (activated carbon-supported phosphoric acid) in the final step eliminates the need for neutralization and salt waste generation associated with homogeneous liquid acids. The high atom economy and solvent recyclability align perfectly with green chemistry principles, making it easier for facilities to maintain compliance with stringent environmental regulations. This forward-looking design ensures that the manufacturing asset remains viable and compliant for years to come, protecting the company from future regulatory risks and potential fines.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alpha-damascenone synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering a transparent view of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the key advantages of this alpha-damascenone synthesis route over conventional methods?
A: The patented process offers significantly higher yields (up to 98% in the first step) compared to older methods like the Schulte-Elte route. It utilizes recyclable solvents like toluene and methanol, drastically reducing waste disposal costs and raw material consumption.
Q: How does the catalyst system ensure high purity in the final fragrance product?
A: The use of specific catalysts such as activated carbon-supported phosphoric acid in the final cyclization step allows for mild reaction conditions (50-60°C). This minimizes thermal degradation and side reactions, ensuring a cleaner impurity profile essential for high-end fragrance applications.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is explicitly designed for industrialization. It features simple operations, mild temperature controls, and the ability to recover and reuse solvents, which are critical factors for maintaining cost-efficiency and supply continuity at the multi-ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Damascenone Supplier
The technical sophistication of this synthesis route highlights the complexity involved in producing high-purity fragrance intermediates at an industrial scale. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications required by the global flavor and fragrance industry. Our rigorous QC labs are equipped to analyze complex impurity profiles, guaranteeing that our alpha-damascenone delivers the consistent olfactory performance that luxury brands demand. We understand that consistency is key in perfumery, and our commitment to process validation ensures that the rose-note character remains uniform across all production lots.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this high-yield methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless transition to a more efficient and sustainable sourcing strategy.
