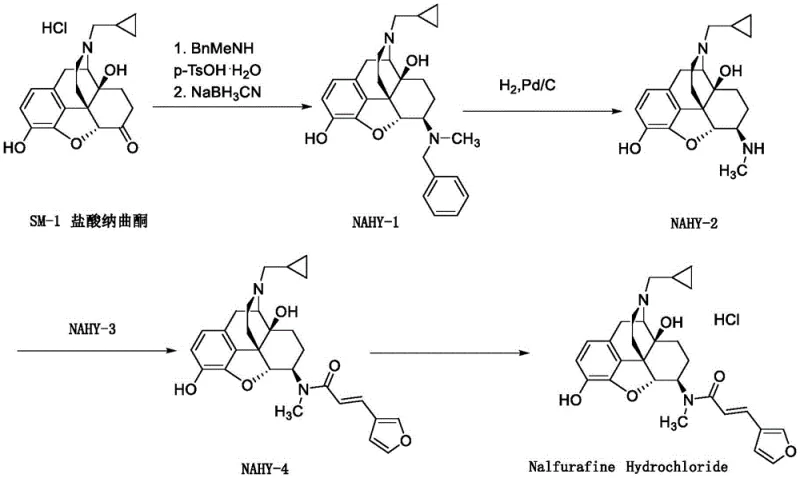
Scalable Production of Nalfurafine Intermediate NAHY-2 via Safe Transfer Hydrogenation
The pharmaceutical landscape for treating uremic pruritus has been significantly advanced by the development of Nalfurafine Hydrochloride, a potent kappa-opioid receptor agonist. However, the commercial viability of this critical medication relies heavily on the efficient and safe production of its key precursor, the intermediate known as NAHY-2 (17-cyclopropylmethyl-3,14 beta-dihydroxy-4,5 alpha-epoxy-6 beta-N-methylmorphinan). Recent intellectual property disclosures, specifically patent CN114437089A, have illuminated a transformative approach to synthesizing this complex morphinan derivative. This patent details a novel debenzylation strategy that replaces hazardous high-pressure hydrogenation with a mild transfer hydrogenation protocol using formic acid. For global procurement leaders and R&D directors, this technological shift represents a pivotal opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. By leveraging this method, manufacturers can bypass the substantial capital expenditures associated with hydrogenation reactors while simultaneously improving reaction kinetics and product purity profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
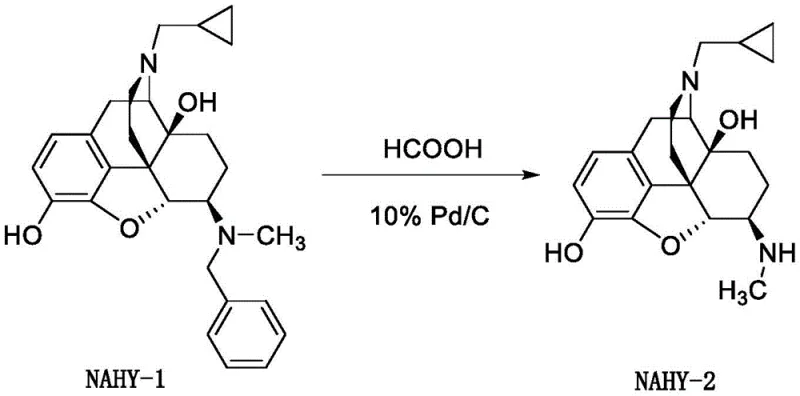
Historically, the conversion of the benzyl-protected precursor NAHY-1 into the target intermediate NAHY-2 has been plagued by significant operational inefficiencies and safety concerns inherent to traditional catalytic hydrogenation. Standard protocols, such as those described in earlier patents like US6277859, necessitate the use of gaseous hydrogen under pressure within specialized autoclave equipment. This requirement introduces severe safety hazards regarding hydrogen storage and handling, alongside the need for expensive, pressure-rated infrastructure that limits scalability in standard multipurpose plants. Furthermore, the reaction kinetics of conventional hydrogenation are often sluggish; literature indicates that even after extending reaction times to twenty-four hours, residual starting material often persists, requiring additional acidic promoters to drive completion. Perhaps most critically from a yield perspective, the traditional workup involves aqueous extraction processes. Given the polar nature of the morphinan scaffold, the product exhibits high water solubility, leading to substantial partitioning losses into the aqueous phase during extraction, which drastically reduces the overall isolated yield to levels often below seventy percent.
The Novel Approach
The methodology outlined in CN114437089A offers a robust solution to these entrenched problems by employing a transfer hydrogenation mechanism utilizing formic acid as the hydrogen source. This approach fundamentally alters the risk profile of the synthesis by eliminating the need for gaseous hydrogen and high-pressure vessels, allowing the reaction to proceed safely at atmospheric pressure and mild temperatures ranging from twenty to forty degrees Celsius. The reaction rate is markedly accelerated, typically reaching completion within just two hours, which significantly enhances throughput capacity. Moreover, the post-reaction processing is streamlined to avoid aqueous extractions entirely; instead, the protocol utilizes filtration to remove the palladium catalyst followed by direct concentration and silica gel chromatography. This modification effectively mitigates the yield losses associated with water solubility, ensuring that the valuable intermediate is retained in the organic phase throughout the purification process, thereby delivering superior mass balance and economic efficiency.
Mechanistic Insights into Formic Acid-Mediated Transfer Hydrogenation
The core chemical innovation driving this process is the in situ generation of active hydrogen species on the surface of the palladium catalyst through the decomposition of formic acid. In this transfer hydrogenation cycle, formic acid adsorbs onto the 10% Pd/C surface and undergoes dehydrogenation to release carbon dioxide and reactive hydride equivalents directly at the catalytic site. These surface-bound hydrides are then immediately available for the reductive cleavage of the benzylic carbon-nitrogen bond in NAHY-1. This mechanism is particularly advantageous for sensitive morphinan structures because it avoids the harsh reducing environments sometimes associated with high-pressure hydrogen gas, which can occasionally lead to over-reduction of other functional groups such as the furan ring or the epoxide bridge. The mild thermal conditions further preserve the stereochemical integrity of the chiral centers at positions 4, 5, and 6, ensuring that the diastereomeric purity remains high and consistent with the stringent requirements for opioid receptor ligands.
Impurity control in this system is managed through a combination of selective catalysis and optimized downstream purification. The use of formic acid generates carbon dioxide as the only byproduct of the hydrogen donation, which evolves as a gas and does not contaminate the reaction mixture with organic residues. Following the reaction, the addition of ammonia water serves a dual purpose: it neutralizes any residual formic acid and adjusts the pH to a basic range of eight to nine, which ensures the free base form of the amine is prevalent for optimal adsorption on silica gel. The subsequent chromatographic step using a dichloromethane and methanol mixture effectively separates the target NAHY-2 from any trace diastereoisomers or unreacted starting materials. This rigorous purification strategy, coupled with a final crystallization step using dichloromethane and cyclohexane, guarantees a product with purity exceeding ninety-nine percent and excellent physical characteristics, such as a white solid appearance, which is critical for downstream coupling reactions.
How to Synthesize NAHY-2 Efficiently
The synthesis of NAHY-2 via this transfer hydrogenation route is designed for operational simplicity and reproducibility across different scales of production. The process begins with the dissolution of the starting material NAHY-1 in a lower alcohol solvent, such as methanol or ethanol, under an inert nitrogen atmosphere to prevent catalyst oxidation. The standardized procedure involves the addition of a specific loading of 10% Pd/C followed by the controlled dropwise addition of formic acid, which initiates the exothermic dehydrogenation process. Detailed standard operating procedures regarding exact stoichiometric ratios, temperature ramping profiles, and crystallization parameters are essential for maintaining batch-to-batch consistency.
- Dissolve NAHY-1 in an alcohol solvent (methanol, ethanol, or isopropanol) under nitrogen protection and add 10% Pd/C catalyst.
- Dropwise add formic acid at 20-30°C, allow the temperature to rise naturally to 20-40°C, and react for approximately 2 hours.
- Adjust pH to 8-9 with ammonia water, filter off the catalyst, concentrate, purify via silica gel chromatography, and crystallize to obtain pure NAHY-2.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain strategists, the adoption of this formic acid-mediated synthesis offers profound advantages in terms of cost structure and logistical reliability. The elimination of high-pressure hydrogenation equipment represents a significant reduction in capital expenditure (CapEx), as facilities no longer require specialized autoclaves or the extensive safety infrastructure needed for bulk hydrogen storage. This democratization of the manufacturing process allows for production in a wider range of standard chemical plants, increasing the potential supplier base and reducing supply chain bottlenecks. Furthermore, the reagents involved, specifically formic acid and palladium on carbon, are commodity chemicals with stable global supply chains, unlike gaseous hydrogen which often requires on-site generation or complex logistics.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the operational workflow and the improvement in material efficiency. By removing the extraction step, the process avoids the loss of product into aqueous waste streams, directly translating to higher yields per kilogram of starting material. Additionally, the shorter reaction time of approximately two hours compared to the twenty-four hours required by conventional methods drastically reduces energy consumption for heating and stirring, as well as labor costs associated with monitoring long-duration batches. The ability to operate at atmospheric pressure also lowers maintenance costs for reactor vessels and removes the regulatory burden associated with high-pressure systems.
- Enhanced Supply Chain Reliability: From a logistics perspective, the reliance on liquid reagents rather than compressed gases enhances the stability and predictability of the supply chain. Formic acid is easily transported and stored in standard chemical drums, eliminating the risks and delays associated with hydrogen cylinder deliveries or pipeline dependencies. The robustness of the reaction conditions, which tolerate mild temperature fluctuations without compromising yield, ensures that production schedules are less susceptible to disruptions caused by minor equipment variances. This reliability is crucial for maintaining continuous supply of the intermediate to downstream API manufacturers, preventing costly production stoppages.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is notably smaller, aligning with modern green chemistry principles and increasingly strict regulatory standards. The absence of high-pressure hydrogen reduces the risk of catastrophic accidents, while the simplified workup generates less aqueous waste containing dissolved organic products. The use of recyclable solvents like methanol and the ability to recover the palladium catalyst from the filtration cake further contribute to a sustainable manufacturing model. This ease of scale-up, demonstrated by the patent's successful transition from gram to hectogram scales without loss of efficiency, assures partners that the technology is ready for multi-ton commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Nalfurafine intermediates, based on the specific innovations found in recent patent literature. Understanding these details is vital for technical teams evaluating the feasibility of integrating this new route into their existing manufacturing portfolios. The answers provided reflect the consensus on best practices for transfer hydrogenation in complex alkaloid synthesis.
Q: What are the safety advantages of using formic acid over hydrogen gas for NAHY-2 synthesis?
A: The use of formic acid as a hydrogen donor eliminates the need for high-pressure autoclaves and gaseous hydrogen, significantly reducing explosion risks and infrastructure costs associated with traditional catalytic hydrogenation.
Q: How does the new method improve yield compared to conventional extraction methods?
A: By avoiding aqueous extraction steps which often lead to product loss due to the high water solubility of the morphinan skeleton, the new method utilizes direct concentration and chromatography, achieving yields of 75-85%.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process operates at mild temperatures (20-40°C) and atmospheric pressure without specialized high-pressure equipment, making it highly scalable and operationally simple for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable NAHY-2 Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to safer, more efficient synthetic routes is critical for the long-term success of pharmaceutical projects. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the formic acid transfer hydrogenation method are fully realized in practice. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of NAHY-2 delivered meets the exacting standards required for opioid receptor agonist synthesis. We are committed to providing a seamless bridge between innovative patent chemistry and reliable commercial supply.
We invite forward-thinking procurement leaders to engage with our technical team to discuss how this advanced manufacturing route can optimize your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this non-hydrogenation method. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments tailored to your volume needs.
