Advanced Catalyst-Free Synthesis of Alpha-Difluoro Selenomethyl Ketones for Commercial Scale-Up
Introduction to Breakthrough Fluorine-Selenium Chemistry
The integration of fluorine and selenium motifs into organic scaffolds represents a frontier in modern medicinal chemistry, offering unique metabolic stability and electronic properties essential for next-generation therapeutics. Patent CN115215778A introduces a transformative methodology for synthesizing alpha-difluoro selenomethyl ketone derivatives, addressing critical bottlenecks in current synthetic strategies. This innovation leverages a catalyst-free oxidative nucleophilic addition between 2,2-difluoroolefins and diaryl diselenides, utilizing potassium persulfate as a benign oxidant. Unlike traditional approaches that demand rigorous anhydrous conditions and transition metal catalysts, this protocol operates efficiently under ambient air atmosphere, significantly lowering the barrier for entry in complex molecule synthesis. For R&D directors and process chemists, this represents a paradigm shift towards greener, more accessible chemical space exploration.
The strategic value of this technology lies in its ability to construct carbon-selenium bonds adjacent to difluoromethylene groups with high regioselectivity and yield. The difluoromethyl group serves as a potent bioisostere for hydroxyl or thiol groups, enhancing lipophilicity and membrane permeability in drug candidates. By streamlining the installation of this pharmacophore, the patented method accelerates the lead optimization phase for pharmaceutical pipelines. Furthermore, the avoidance of heavy metal catalysts simplifies downstream purification, directly impacting the purity profile of the final active pharmaceutical ingredient (API) intermediates. This alignment with green chemistry principles makes it an attractive candidate for sustainable manufacturing processes in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for constructing alpha-difluoro selenomethyl ketones have historically relied on base-mediated C-C bond cleavage strategies, typically employing cesium carbonate (Cs2CO3) to generate difluoroenol intermediates. These conventional routes suffer from inherent inefficiencies, primarily due to poor atom economy where a benzoyl anion molecule must be eliminated to form the reactive intermediate. This necessitates the addition of nucleophilic scavengers to trap the byproduct, generating unnecessary waste and complicating the reaction mixture. Moreover, existing literature indicates that these systems exhibit poor substrate compatibility specifically for selenium incorporation, often favoring sulfur analogs with significantly lower yields for seleno-derivatives. Such limitations restrict the structural diversity accessible to medicinal chemists and hinder the规模化 production of these valuable building blocks.
The Novel Approach
In stark contrast, the invention disclosed in CN115215778A utilizes a direct oxidative coupling strategy that bypasses the need for pre-functionalized enol equivalents or harsh basic conditions. By employing potassium persulfate (K2S2O8) as a radical initiator and oxidant, the reaction proceeds through a smooth nucleophilic addition-oxidation cascade. This approach not only improves atom economy by incorporating all reactant atoms into the final product structure but also dramatically expands the substrate scope to include complex polycyclic aromatic systems and heterocycles. The operational simplicity is further enhanced by the ability to conduct the reaction in air, removing the capital expenditure associated with gloveboxes or extensive nitrogen purging systems. This robustness makes the process highly amenable to technology transfer from laboratory bench to pilot plant.
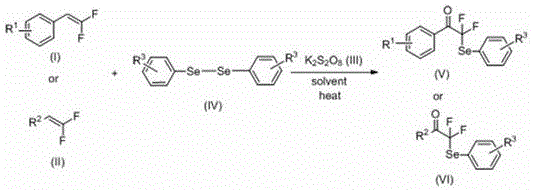
The visual representation of the general reaction scheme underscores the elegance of this transformation, where diverse R groups on the olefin and diselenide components are tolerated without significant loss in efficiency. This modularity is crucial for generating libraries of analogs during the drug discovery phase. The use of dimethyl sulfoxide (DMSO) as a preferred solvent further enhances solubility of the polar intermediates, driving the reaction to high conversion rates. For procurement managers, the shift away from exotic catalysts to commodity chemicals like potassium persulfate signals a potential reduction in raw material volatility and cost. The method effectively decouples the synthesis of these high-value intermediates from the supply chain constraints often associated with specialized organometallic reagents.
Mechanistic Insights into Oxidative Nucleophilic Addition
The mechanistic pathway of this transformation involves the generation of selenium-centered radicals initiated by the thermal decomposition of potassium persulfate. These radicals add across the electron-deficient double bond of the 2,2-difluoroolefin, forming a carbon-centered radical intermediate stabilized by the adjacent fluorine atoms. Subsequent oxidation by the persulfate species converts this radical into a cationic or oxo-species, which undergoes hydrolysis or rearrangement to yield the final ketone functionality. This radical-polar crossover mechanism is distinct from ionic pathways and allows for the activation of relatively inert C-H and C-Se bonds under mild thermal conditions. Understanding this mechanism is vital for process optimization, as it explains the tolerance for various functional groups that might otherwise be sensitive to strong bases or nucleophiles used in traditional methods.
Impurity control in this system is inherently managed by the selectivity of the radical addition step. Since the reaction does not involve strong bases, side reactions such as aldol condensations or elimination reactions are minimized. The use of air as the terminal oxidant source, facilitated by the persulfate cycle, ensures that over-oxidation to sulfones or selenones is kept in check by the stoichiometry of the oxidant. For quality control teams, this translates to a cleaner crude reaction profile, reducing the burden on chromatographic purification steps. The ability to tune the reaction by adjusting the molar ratio of oxidant to substrate provides an additional handle for maximizing yield while minimizing the formation of selenium-containing byproducts, ensuring the final material meets stringent pharmaceutical specifications.
How to Synthesize Alpha-Difluoro Selenomethyl Ketone Efficiently
The practical execution of this synthesis is designed for ease of operation, requiring standard laboratory glassware and commercially available reagents. The protocol involves charging a reaction vessel with the difluoroolefin substrate, the diaryl diselenide coupling partner, and the oxidant in a polar aprotic solvent. Heating the mixture to moderate temperatures facilitates the radical generation and subsequent bond formation. Detailed standard operating procedures for scaling this reaction, including specific workup and purification parameters, are outlined in the technical guide below to ensure reproducibility across different manufacturing sites.
- Combine 2,2-difluoroolefin derivative, diaryl diselenide, and potassium persulfate in a Schlenk reaction bottle with DMSO solvent.
- Stir the reaction mixture at 40°C under an air atmosphere for approximately 24 hours to ensure complete conversion.
- Perform post-processing via ethyl acetate extraction, washing with saturated brine, drying, and purification by column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers compelling advantages that directly address the pain points of cost and supply chain reliability in fine chemical manufacturing. The elimination of transition metal catalysts removes the need for expensive metal scavenging resins and the associated analytical testing for residual metals, which is a significant cost driver in API production. Furthermore, the use of air-stable reagents reduces the complexity of storage and handling, lowering the operational overhead for warehouse and logistics teams. The robustness of the reaction conditions means that batch-to-batch variability is minimized, leading to more predictable production schedules and reduced risk of campaign failures.
- Cost Reduction in Manufacturing: The replacement of specialized catalysts and bases with commodity oxidants like potassium persulfate results in substantial raw material cost savings. Additionally, the simplified workup procedure, which avoids complex quenching steps required for reactive organometallics, reduces solvent consumption and waste disposal costs. The high atom economy ensures that a greater proportion of purchased raw materials ends up in the final product, maximizing the return on investment for every kilogram of input. This efficiency is critical for maintaining margin integrity in competitive generic drug markets.
- Enhanced Supply Chain Reliability: The starting materials, including 2,2-difluoroolefins and diaryl diselenides, are either commercially available or can be synthesized via straightforward routes from abundant aldehydes. This diversifies the supplier base and mitigates the risk of single-source dependency. The ability to run the reaction under air atmosphere removes the dependency on bulk inert gases like nitrogen or argon, which can be subject to supply fluctuations in certain regions. This resilience ensures continuous production capability even during logistical disruptions, securing the supply of critical intermediates for downstream drug synthesis.
- Scalability and Environmental Compliance: The patent data confirms successful scale-up to gram quantities with maintained yields, demonstrating the feasibility of transitioning to kilogram and ton scales. The absence of toxic heavy metals aligns with increasingly strict environmental regulations regarding effluent discharge and worker safety. The use of DMSO, a recyclable solvent, further supports sustainability goals. For supply chain heads, this means faster regulatory approval for new manufacturing sites and reduced liability associated with hazardous chemical handling, facilitating smoother global distribution of the manufactured intermediates.
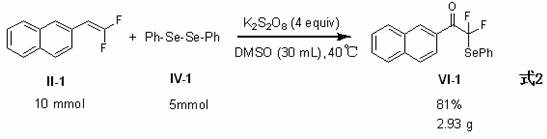
The successful demonstration of gram-scale synthesis, as depicted in the experimental data, serves as a proof-of-concept for industrial viability. It confirms that heat and mass transfer issues do not impede the reaction progress at larger volumes. This scalability is a key determinant for contract development and manufacturing organizations (CDMOs) when evaluating new technologies for client projects. The ability to seamlessly transition from milligram screening to multi-kilogram production without re-optimizing the core chemistry saves significant time and resources. For partners looking to secure long-term supply agreements, this technological maturity offers assurance of consistent quality and volume availability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders assess the fit of this chemistry within their existing manufacturing frameworks and quality systems.
Q: What are the key advantages of this catalyst-free method over traditional Cs2CO3 mediated routes?
A: The novel method eliminates the need for expensive catalysts and bases, operates under mild air conditions, and offers superior atom economy by avoiding the generation of benzoyl anion byproducts common in prior art.
Q: Is this synthesis protocol suitable for large-scale industrial production?
A: Yes, the patent explicitly demonstrates successful gram-scale amplification with high yields, indicating robust scalability and suitability for commercial manufacturing without complex inert atmosphere requirements.
Q: What is the substrate scope for the difluoroolefin starting materials?
A: The method exhibits excellent compatibility with various substituted aryl groups, including naphthyl, phenanthryl, and heterocycles like thiophene, allowing for diverse structural modifications in drug design.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Difluoro Selenomethyl Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced fluorination and selenylation technologies in accelerating drug discovery timelines. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are translated into robust industrial processes. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and chiral purity assessments, guaranteeing that every batch meets the exacting standards required by global regulatory agencies. We are committed to delivering high-purity pharmaceutical intermediates that empower your R&D teams to push the boundaries of medicinal chemistry.
We invite you to engage with our technical procurement team to discuss how this catalyst-free synthesis can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this greener methodology for your specific project needs. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to optimize your synthesis routes, reduce time-to-market, and secure a reliable supply of critical building blocks for your next generation of therapeutics.
