Scalable Production of High-Purity Maropitant Intermediates via Advanced Asymmetric Catalysis
Scalable Production of High-Purity Maropitant Intermediates via Advanced Asymmetric Catalysis
The pharmaceutical industry continuously seeks robust methodologies for synthesizing complex chiral molecules, particularly for veterinary and human therapeutics like Maropitant, a potent neurokinin-1 (NK1) receptor antagonist. Patent CN112300151A discloses a groundbreaking preparation method for Maropitant intermediates that fundamentally shifts the paradigm from wasteful resolution techniques to efficient asymmetric catalysis. This technology enables the simultaneous introduction of two critical chiral centers in a single reaction step, utilizing specialized transition metal catalysts to achieve exceptional stereocontrol. For global supply chain leaders and R&D directors, this innovation represents a significant leap forward in process chemistry, offering a pathway to high-purity intermediates with minimized environmental impact and enhanced production efficiency.
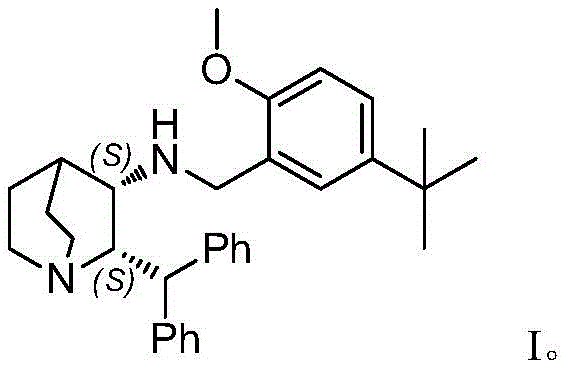
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Maropitant has relied heavily on classical chemical resolution strategies to establish the necessary stereochemistry. These legacy processes typically involve constructing a single chiral center first, followed by multiple rounds of catalytic reductive amination using platinum or palladium on carbon under hydrogen pressure. A major drawback of these conventional routes is the inherent inefficiency of resolution, which theoretically caps the yield at 50% for the desired enantiomer, necessitating the disposal or recycling of the unwanted isomer. Furthermore, the requirement for multiple distinct hydrogenation steps—often involving different catalysts like Pt/C for amination and Pd/C for deprotection—complicates the operational workflow, increases the accumulation of heavy metal impurities, and extends the overall production timeline, thereby inflating manufacturing costs and supply chain risks.
The Novel Approach
In stark contrast, the novel methodology described in the patent utilizes a sophisticated transition metal-catalyzed asymmetric reductive amination to construct the molecular architecture in a highly convergent manner. By reacting a ketone precursor (Compound 02) directly with an amine source in the presence of a chiral transition metal catalyst and a hydrogen donor, the process installs both chiral centers simultaneously with high diastereo- and enantioselectivity. This one-pot transformation eliminates the need for tedious resolution steps and reduces the total number of synthetic operations, leading to a drastic simplification of the process flow. The result is a streamlined route that not only improves the overall yield but also ensures superior product quality by minimizing the formation of optical isomer impurities that are difficult to remove in later stages.
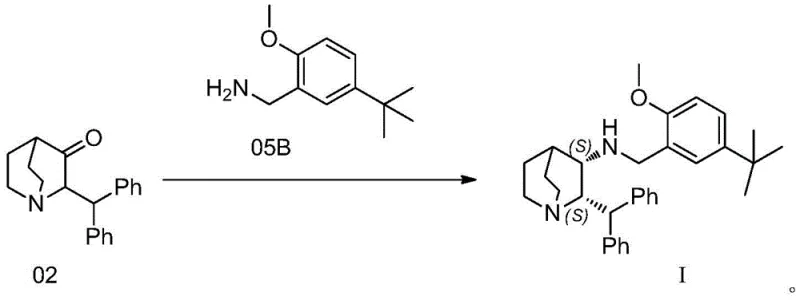
Mechanistic Insights into Transition Metal-Catalyzed Asymmetric Reductive Amination
The core of this technological breakthrough lies in the precise design of the transition metal catalyst, generally represented by Formula II, which orchestrates the stereochemical outcome of the reaction. The catalyst typically features a central metal atom such as Iridium, Rhodium, or Ruthenium, coordinated with chiral ligands that create a sterically constrained environment around the active site. During the reaction, the catalyst facilitates the formation of an imine intermediate from the ketone and amine, followed by a highly selective hydride transfer from the hydrogen donor (such as formic acid or ammonium formate) to the imine double bond. The chiral ligands, often derived from sulfonamides with bulky substituents like triisopropylphenyl groups, effectively shield one face of the prochiral imine, forcing the hydride attack to occur from a specific trajectory that yields the desired (S,S) configuration.
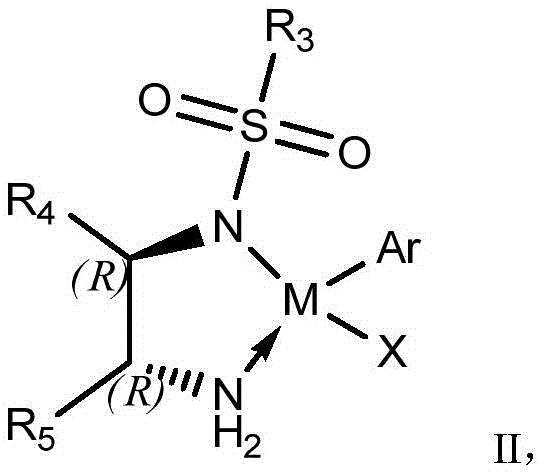
Impurity control is intrinsically built into this catalytic cycle, as the high selectivity of the metal complex suppresses the formation of the three potential optical isomer impurities that plague non-selective routes. The patent specifies that by fine-tuning the substituents on the catalyst ligand (R3, R4, R5) and optimizing reaction parameters such as temperature (0°C to 50°C) and molar ratios, the enantiomeric excess (ee) of the product can be pushed to greater than 99%. This level of precision is critical for regulatory compliance, as it ensures that the final bulk drug substance meets stringent specifications for isomeric purity without requiring extensive downstream purification. The robustness of the catalyst system allows it to function effectively in common organic solvents like tetrahydrofuran or toluene, making it compatible with existing industrial infrastructure.
How to Synthesize Maropitant Intermediate Efficiently
The synthesis protocol outlined in the patent provides a clear and reproducible framework for manufacturing the key intermediate Compound 03 or the final precursor Compound I. The process begins with the condensation of the ketone substrate and the amine component, promoted by agents like tetraisopropyl titanate to drive imine formation. Subsequently, the chiral catalyst and hydrogen source are introduced to effect the asymmetric reduction under mild thermal conditions. Detailed standard operating procedures regarding reagent addition sequences, temperature gradients, and workup protocols are essential for maximizing yield and optical purity. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized steps below.
- Mix Compound 02 with an amine source (benzylamine or Compound 05B) and a promoting agent like tetraisopropyl titanate in an organic solvent such as THF or toluene.
- Introduce a transition metal catalyst (Formula II, preferably Iridium-based), a hydrogen donor like formic acid, and an optional base reagent to the reaction mixture.
- Maintain the reaction at 0°C to 50°C (or reflux for imine formation) for 15-24 hours, followed by pH adjustment, extraction, and crystallization to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this catalytic technology offers profound strategic benefits for procurement managers and supply chain directors focused on cost optimization and reliability. By transitioning from a multi-step resolution process to a direct asymmetric synthesis, manufacturers can significantly reduce the consumption of raw materials and solvents, directly translating to lower variable costs per kilogram of product. The elimination of resolution steps removes the inherent 50% yield penalty associated with discarding unwanted enantiomers, effectively doubling the theoretical output from the same amount of starting material. This efficiency gain allows for substantial cost reduction in pharmaceutical intermediate manufacturing, enabling more competitive pricing structures for the final veterinary drug while maintaining healthy margins for suppliers.
- Cost Reduction in Manufacturing: The streamlined nature of the one-step catalytic process drastically reduces the number of unit operations required, such as filtration, drying, and solvent swaps, which are major cost drivers in chemical production. Furthermore, the avoidance of expensive stoichiometric resolving agents and the reduction in catalyst loading (down to 0.001:1 molar ratio in some embodiments) contribute to a leaner cost profile. The ability to use common hydrogen donors like formic acid instead of high-pressure hydrogen gas in certain steps also lowers safety compliance costs and equipment investment requirements.
- Enhanced Supply Chain Reliability: The simplified process flow shortens the overall manufacturing lead time, allowing suppliers to respond more rapidly to market demand fluctuations. Since the method relies on readily available starting materials and robust catalyst systems that tolerate minor variations in conditions, the risk of batch failures is minimized, ensuring a consistent and continuous supply of high-quality intermediates. This reliability is crucial for pharmaceutical companies managing tight inventory schedules and seeking to mitigate the risks associated with complex, multi-vendor supply chains.
- Scalability and Environmental Compliance: The reaction conditions described, ranging from ambient temperature to moderate reflux, are easily scalable from laboratory benchtop to multi-ton commercial production without requiring exotic high-pressure or cryogenic equipment. Additionally, the reduction in waste generation—due to higher yields and fewer purification steps—aligns with modern green chemistry principles and environmental regulations. This makes the process not only economically attractive but also environmentally sustainable, reducing the burden of waste treatment and enhancing the corporate social responsibility profile of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and performance of this advanced synthesis route. These answers are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production portfolios.
Q: What enantiomeric excess (ee) can be achieved with this catalytic method?
A: The patented process consistently achieves an ee value greater than 95%, with optimized embodiments reaching greater than 99% ee, ensuring high optical purity for the final API.
Q: Which transition metal catalysts are most effective for this synthesis?
A: Iridium, rhodium, or ruthenium complexes represented by Formula II are effective, with Iridium-based catalysts containing cyclopentadienyl ligands showing superior selectivity for introducing the dual chiral centers.
Q: How does this method improve upon traditional chemical resolution?
A: Unlike traditional methods that require multi-step hydrogenation and manual resolution with a maximum 50% theoretical yield, this one-step asymmetric reductive amination creates both chiral centers simultaneously with high yield and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Maropitant Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic methodologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the catalytic asymmetric amination technology described in CN112300151A and possesses the technical capability to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee ee values exceeding 99% as required by top-tier pharmaceutical clients.
We invite you to collaborate with us to leverage this innovative process for your Maropitant supply needs. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact us today to request specific COA data from our pilot batches and discuss detailed route feasibility assessments that demonstrate how we can deliver high-purity intermediates with superior economic and operational advantages.
