Advanced Manufacturing of Metopimazine: Overcoming Oxidation Selectivity Barriers
Advanced Manufacturing of Metopimazine: Overcoming Oxidation Selectivity Barriers
The pharmaceutical industry's relentless pursuit of effective antiemetics has placed significant focus on the scalable production of Metopimazine, a potent dopamine D2 and D3 receptor inhibitor. As detailed in the recent patent CN109053716B, a transformative synthetic methodology has been disclosed that fundamentally restructures the manufacturing landscape for this critical active pharmaceutical ingredient. Unlike historical approaches that struggle with the thermodynamic and kinetic challenges of late-stage functionalization, this novel process employs a convergent strategy that installs the crucial methylsulfonyl moiety prior to ring closure. This strategic inversion of the synthetic sequence not only mitigates the notorious selectivity issues associated with phenothiazine oxidation but also delivers a final product with exceptional purity exceeding 99.3 percent. For global supply chain stakeholders, this represents a pivotal shift towards more robust and predictable manufacturing protocols.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Metopimazine have long been plagued by the inherent chemical instability and reactivity conflicts within the phenothiazine scaffold. The seminal route reported by Jacob et al. in 1959, for instance, relies on the protection of the phenothiazine nitrogen followed by the oxidation of a thioether side chain. 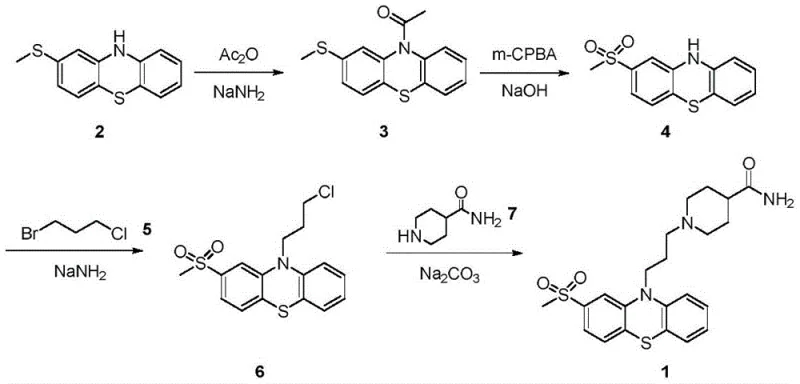 As illustrated in the reaction scheme, the critical bottleneck lies in the oxidation of intermediate 3, where the presence of two distinct thioether functionalities creates a severe selectivity challenge. Achieving mono-oxidation to the sulfone without affecting the ring sulfur or over-oxidizing the side chain requires precise control that is difficult to maintain on a multi-ton scale. Subsequent optimizations by researchers such as Sindalar and Reddy attempted to circumvent these issues through alternative ring-closing strategies or phase-transfer catalysis, yet these methods often resulted in cumulative yields as low as 24 percent due to persistent byproduct formation and complex purification requirements.
As illustrated in the reaction scheme, the critical bottleneck lies in the oxidation of intermediate 3, where the presence of two distinct thioether functionalities creates a severe selectivity challenge. Achieving mono-oxidation to the sulfone without affecting the ring sulfur or over-oxidizing the side chain requires precise control that is difficult to maintain on a multi-ton scale. Subsequent optimizations by researchers such as Sindalar and Reddy attempted to circumvent these issues through alternative ring-closing strategies or phase-transfer catalysis, yet these methods often resulted in cumulative yields as low as 24 percent due to persistent byproduct formation and complex purification requirements.
The Novel Approach
The methodology outlined in patent CN109053716B introduces a paradigm shift by constructing the phenothiazine core around a pre-functionalized aromatic system. Instead of attempting to oxidize a sensitive thioether on the finished heterocycle, this route begins with the nucleophilic aromatic substitution between o-fluorobenzothiophenol and o-nitro p-methylsulfonyl chlorobenzene. 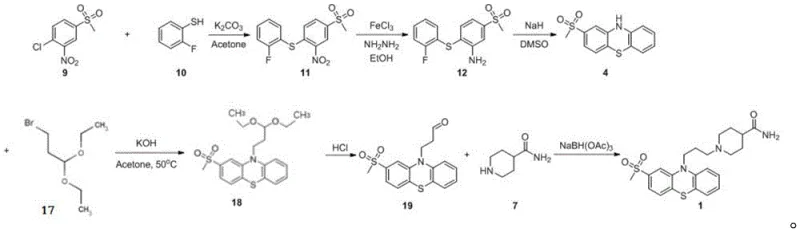 This initial coupling efficiently generates the nitro-intermediate 11, which carries the methylsulfonyl group in its final oxidation state from the very beginning. By deferring the formation of the phenothiazine ring until after the sulfone is securely in place, the process entirely eliminates the risk of non-selective oxidation. The subsequent reductive cyclization and N-alkylation steps proceed with high fidelity, leveraging the electronic properties of the pre-installed sulfone to facilitate clean ring closure, thereby drastically simplifying the downstream isolation and purification workflows.
This initial coupling efficiently generates the nitro-intermediate 11, which carries the methylsulfonyl group in its final oxidation state from the very beginning. By deferring the formation of the phenothiazine ring until after the sulfone is securely in place, the process entirely eliminates the risk of non-selective oxidation. The subsequent reductive cyclization and N-alkylation steps proceed with high fidelity, leveraging the electronic properties of the pre-installed sulfone to facilitate clean ring closure, thereby drastically simplifying the downstream isolation and purification workflows.
Mechanistic Insights into FeCl3-Catalyzed Reduction and Cyclization
The heart of this optimized synthesis lies in the efficient transformation of the nitro-intermediate 11 into the cyclized phenothiazine 4. The reduction step utilizes ferric chloride hexahydrate and hydrazine hydrate in an ethanol medium, a system chosen for its ability to selectively reduce the nitro group to an amine without compromising the sulfone functionality. This chemoselectivity is paramount, as harsher reducing conditions could potentially degrade the sulfonyl group or induce unwanted side reactions on the fluorophenyl ring. Following the reduction to intermediate 12, the system undergoes an intramolecular nucleophilic aromatic substitution mediated by sodium hydride in dimethyl sulfoxide. The strong base deprotonates the newly formed aniline nitrogen, generating a highly nucleophilic species that attacks the ortho-position of the adjacent phenyl ring, displacing the fluoride leaving group. This cyclization is driven by the formation of the stable aromatic phenothiazine system and is significantly aided by the electron-withdrawing nature of the sulfone group, which activates the ring towards nucleophilic attack while stabilizing the transition state.
Furthermore, the impurity profile of this route is inherently superior due to the avoidance of radical-mediated oxidation steps. In traditional thioether oxidations, radical intermediates often lead to dimerization or polymerization byproducts that are structurally similar to the target molecule, making them exceptionally difficult to remove via crystallization. In contrast, the ionic mechanisms governing the nucleophilic substitution and base-mediated cyclization in this new process generate byproducts that are either inorganic salts or structurally distinct organic fragments. These impurities are far more amenable to standard workup procedures such as aqueous washing and solvent pulping. The result is a crude product with significantly higher assay values, reducing the reliance on resource-intensive chromatographic purification and ensuring that the final API meets stringent regulatory specifications for genotoxic impurities and residual solvents.
How to Synthesize Metopimazine Efficiently
The execution of this synthetic pathway requires careful attention to reaction stoichiometry and temperature control, particularly during the exothermic cyclization and the final reductive amination steps. The process is designed to be telescoped where possible, minimizing the number of isolation points to maximize overall throughput. Operators should note that the use of anhydrous conditions during the sodium hydride mediated ring closure is critical to prevent hydrolysis of the reactive intermediates. For a comprehensive breakdown of the specific reaction parameters, stoichiometric ratios, and workup procedures validated in the patent examples, please refer to the standardized synthesis guide below.
- Perform nucleophilic aromatic substitution between o-fluorobenzothiophenol and o-nitro p-methylsulfonyl chlorobenzene to form the nitro-intermediate.
- Reduce the nitro group to an amine using ferric chloride and hydrazine hydrate in ethanol.
- Execute ring closure using sodium hydride in DMSO to form the phenothiazine core with the sulfone group already in place.
- Alkylate the nitrogen with a protected propyl chain, followed by deprotection and reductive amination with piperidine-4-carboxamide.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthetic route offers profound advantages for procurement managers and supply chain directors seeking to optimize the cost of goods sold for antiemetic formulations. The primary economic driver is the substantial reduction in processing time and material consumption associated with the elimination of complex purification stages. By avoiding the low-yielding and selectivity-poor oxidation steps characteristic of legacy routes, manufacturers can achieve a significantly higher throughput per batch without expanding reactor capacity. This efficiency gain translates directly into lower variable costs per kilogram of produced API, providing a competitive edge in price-sensitive markets. Moreover, the robustness of the chemistry reduces the incidence of failed batches, thereby enhancing supply continuity and reducing the safety stock requirements typically needed to buffer against production volatility.
- Cost Reduction in Manufacturing: The economic benefits of this process are largely derived from the simplification of the synthetic sequence. Traditional routes often necessitate multiple protection and deprotection steps, alongside expensive oxidizing agents and extensive chromatographic purification to separate regioisomers. This new method streamlines the workflow by installing the sulfone group early using commodity chemicals, effectively removing the need for costly selective oxidation reagents and the associated waste disposal costs. The high purity of the intermediates allows for simple crystallization or pulping operations instead of column chromatography, which dramatically lowers solvent consumption and labor hours, resulting in substantial cost savings in API manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable starting materials such as o-fluorobenzothiophenol and substituted chlorobenzenes. Unlike specialized oxidants or catalysts that may be subject to supply constraints or long lead times, the reagents utilized in this process are commodity chemicals with mature global supply networks. This accessibility ensures that production schedules are not disrupted by raw material shortages. Additionally, the high yield and reproducibility of the reaction steps mean that production forecasts are more accurate, allowing for better inventory planning and a more reliable delivery schedule to downstream formulation partners.
- Scalability and Environmental Compliance: The environmental footprint of the manufacturing process is significantly reduced, aligning with modern green chemistry principles and regulatory expectations. The avoidance of heavy metal oxidants and the reduction in solvent usage for purification minimize the generation of hazardous waste streams. This simplifies wastewater treatment and lowers the cost of environmental compliance. Furthermore, the reaction conditions are amenable to large-scale batch processing, with thermal profiles that are manageable in standard glass-lined steel reactors. This scalability ensures that the process can be seamlessly transferred from pilot plant to commercial production scales of 100 kgs to 100 MT without requiring specialized equipment or posing significant safety risks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the operational benefits and chemical rationale behind the new process. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer.
Q: Why is the conventional oxidation of phenothiazine thioethers problematic?
A: Conventional routes often require oxidizing a thioether to a sulfone on an already formed phenothiazine ring. This step suffers from poor chemoselectivity, often over-oxidizing to sulfones or failing to convert fully, leading to difficult purification and low yields.
Q: What purity levels does the new process achieve?
A: The optimized process described in patent CN109053716B achieves a final product purity of greater than 99.3%, significantly reducing the burden on downstream purification processes.
Q: How does this route improve supply chain reliability?
A: By utilizing readily available starting materials like o-fluorobenzothiophenol and avoiding complex chromatographic separations associated with isomer removal, the route ensures more consistent batch-to-batch quality and shorter production cycles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metopimazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN109053716B can be reliably replicated in our facilities. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Metopimazine meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific supply chain needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this optimized synthesis can enhance your product portfolio. By collaborating with us, you gain access to a secure, high-quality supply of Metopimazine that supports your commitment to patient care while optimizing your operational expenditures.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
