Industrial Scale-Up of Watermelon Ketone: Advanced Synthesis and Purification Strategies for Global Supply Chains
The global demand for high-quality olfactory compounds continues to drive innovation in fine chemical synthesis, particularly for marine-ozonic fragrances like Watermelon Ketone (Calone). A significant technological breakthrough in this sector is detailed in patent CN114292256A, which discloses a robust preparation and purification method specifically optimized for industrial production. This patent addresses the longstanding challenges of low conversion rates and difficult purification associated with traditional synthesis routes. By utilizing a direct condensation reaction between 4-methyl catechol and 1,3-dichloroacetone in a 2-butanone system, the inventors have established a pathway that significantly simplifies the operational workflow. Furthermore, the introduction of a novel tar-removal pretreatment prior to distillation represents a critical advancement in process engineering, ensuring that the final product meets rigorous purity standards while maximizing equipment uptime and overall yield.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Watermelon Ketone has been plagued by complex multi-step sequences that hinder scalability and increase production costs. Early methodologies, such as those described in CN108164499A, relied on ketal protection strategies followed by cyclization. As illustrated in the reaction scheme below, these routes often necessitate the use of ether solvents for extraction, which pose significant safety hazards due to their low boiling points and anesthetic properties, making them unsuitable for modern large-scale manufacturing environments.
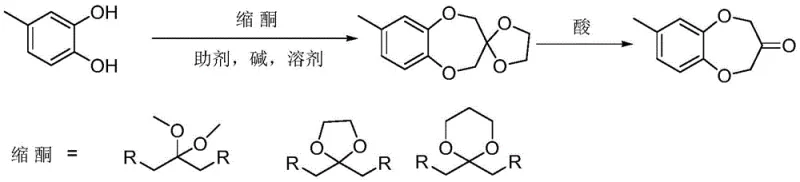
Other conventional approaches, such as the Williamson etherification followed by Dickmann condensation, require harsh reagents like sodium hydride (NaH) or potassium tert-butoxide. These strong bases demand strictly anhydrous conditions and generate substantial amounts of salt waste, complicating the post-reaction workup. Additionally, routes involving oxidation steps often result in the formation of numerous by-products, leading to low yields and necessitating expensive column chromatography for purification, a technique that is economically unviable for ton-scale production.
The Novel Approach
In stark contrast to the convoluted pathways of the past, the method disclosed in CN114292256A streamlines the synthesis into a direct, efficient condensation. The core innovation lies in the reaction of 4-methyl catechol with 1,3-dichloroacetone, catalyzed by iodide salts in a carbonate-buffered 2-butanone medium. This approach eliminates the need for protecting groups and harsh strong bases, replacing them with milder, more manageable reagents.
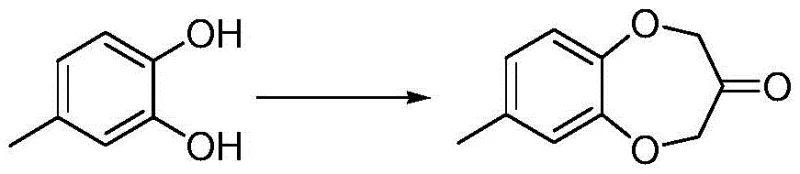
The simplicity of this novel route is visually evident when comparing it to prior art; it reduces the number of unit operations and avoids the generation of difficult-to-remove impurities at the source. By optimizing the molar ratios and reaction temperature (75-80°C), the process achieves high conversion rates with minimal starting material remaining. This directness not only accelerates the production cycle but also fundamentally alters the impurity profile, making downstream purification far more straightforward and cost-effective compared to traditional oxidative or multi-step condensation methods.
Mechanistic Insights into Iodide-Catalyzed Cyclization
The chemical efficacy of this process relies heavily on the synergistic interaction between the iodide catalyst and the carbonate base within the 2-butanone solvent system. Mechanistically, the iodide ion (from KI or NaI) acts as a nucleophilic catalyst, facilitating the displacement of the chloride leaving groups on the 1,3-dichloroacetone through a Finkelstein-type activation. This generates a more reactive iodo-intermediate in situ, which undergoes rapid nucleophilic attack by the phenolic hydroxyl groups of the 4-methyl catechol. The carbonate base serves a dual purpose: it neutralizes the hydrochloric acid by-product generated during the etherification, driving the equilibrium forward, and it maintains a pH environment that prevents the degradation of the sensitive catechol substrate.
Beyond the reaction kinetics, the patent details a sophisticated physical-chemical mechanism for impurity control during the purification phase. The 'pulping' step with n-heptane is critical; it exploits the differential solubility of the target ketone versus high-molecular-weight tars and polymeric by-products. By suspending the crude product in a non-polar solvent like n-heptane, the tars remain insoluble or form a separate phase that can be mechanically separated or adsorbed onto activated carbon. This pre-distillation cleaning step is vital because it prevents the thermal polymerization of tars inside the distillation column, which would otherwise lead to fouling, reduced heat transfer efficiency, and contamination of the final distillate, thereby ensuring a consistent high-purity output.
How to Synthesize Watermelon Ketone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory bench scale to industrial manufacturing. The process begins with the dissolution of 4-methylcatechol in 2-butanone, followed by the sequential addition of the iodide catalyst and carbonate base. The reaction is initiated by the controlled dropwise addition of 1,3-dichloroacetone at elevated temperatures, ensuring exothermic control and complete conversion. Following the reaction, the mixture is filtered to remove inorganic salts, and the solvent is recovered under reduced pressure. The resulting crude solid then undergoes the proprietary purification sequence involving solvent pulping, carbon treatment, and recrystallization. For a detailed, step-by-step breakdown of the standardized operating procedures and critical process parameters, please refer to the technical guide below.
- React 4-methyl catechol with 1,3-dichloroacetone in 2-butanone solvent using potassium iodide and sodium carbonate catalysts at 75-80°C.
- Remove reaction salts via filtration and concentrate the filtrate to obtain the crude product.
- Purify the crude product by pulping with n-heptane to remove tar, followed by activated carbon treatment, distillation, and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the supply chain for raw materials and the reduction of processing hazards. By eliminating the need for specialized, hazardous reagents like sodium hydride or complex protecting group chemistries, the process reduces the dependency on niche chemical suppliers and lowers the regulatory burden associated with handling dangerous goods. This simplification translates directly into a more resilient supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of expensive purification techniques. Traditional methods often require column chromatography or multiple recrystallizations to remove stubborn impurities, which consume vast quantities of solvent and silica gel. In contrast, this method utilizes a 'tar removal' strategy that allows for direct distillation and a single recrystallization step. Furthermore, the replacement of ether solvents with 2-butanone reduces solvent costs and recovery energy requirements. The avoidance of transition metal catalysts or precious metal scavengers further strips away layers of cost, resulting in a significantly leaner cost of goods sold (COGS) structure.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthesis route ensures consistent delivery schedules. The use of commodity chemicals such as 4-methyl catechol, 1,3-dichloroacetone, and 2-butanone means that raw material sourcing is not a bottleneck. Unlike processes relying on custom-synthesized intermediates which may have long lead times, the inputs for this method are widely available on the global market. Additionally, the simplified workup procedure reduces the total batch cycle time, allowing manufacturing facilities to increase throughput and respond more agilely to fluctuations in market demand without requiring capital-intensive expansion of reactor capacity.
- Scalability and Environmental Compliance: The design of this process inherently supports green chemistry principles, which is increasingly critical for maintaining social license to operate. The removal of tar prior to distillation minimizes the generation of hazardous still-bottoms waste, reducing disposal costs and environmental footprint. The solvent system, primarily consisting of 2-butanone and n-heptane, is amenable to efficient recycling and recovery loops, minimizing volatile organic compound (VOC) emissions. This environmental compatibility facilitates easier permitting for plant expansions and aligns with the sustainability goals of major multinational fragrance houses, making the supplier a more attractive partner for long-term contracts.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms legacy technologies in terms of purity, yield, and operational safety. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing production lines.
Q: How does the new purification method improve distillation efficiency?
A: The patented process introduces a specific 'pulping' step using n-heptane prior to distillation. This effectively removes tar and heavy impurities that typically accumulate in distillation kettles, preventing product damage and significantly improving distillation yield and equipment longevity.
Q: What are the advantages of using 2-butanone over ether solvents?
A: Unlike ether solvents which have low boiling points and safety concerns regarding anesthesia and flammability, 2-butanone (MEK) offers a safer operational profile suitable for large-scale industrial production while maintaining excellent solubility for the reactants.
Q: Can this method achieve pharmaceutical-grade purity?
A: Yes, the combination of tar removal, activated carbon adsorption, vacuum distillation, and final recrystallization from toluene/n-heptane allows the process to consistently achieve purity levels exceeding 99%, meeting stringent specifications for high-end fragrance applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Watermelon Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into tangible commercial reality through expert process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield demonstrated in the lab are faithfully reproduced at the plant level. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Watermelon Ketone meets the exacting standards required by the global flavors and fragrances industry.
We invite procurement leaders and R&D directors to engage with us to explore how this advanced synthesis route can optimize your supply chain. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your fragrance ingredient sourcing strategy.
