Advanced Catalytic Recovery of S-Praziquantel Intermediate for Sustainable L-Praziquantel Manufacturing
The pharmaceutical industry continuously seeks sustainable methodologies to optimize the production of chiral active pharmaceutical ingredients (APIs), particularly for broad-spectrum antiparasitic agents like Praziquantel. Patent CN107814797B introduces a groundbreaking approach to address a significant inefficiency in the synthesis of L-Praziquantel, the biologically active enantiomer. Traditionally, the production of L-Praziquantel via chiral resolution generates a substantial waste stream of the unwanted S-configured intermediate, which is typically discarded. This patent discloses a novel catalytic hydrogenation method that successfully recovers this wasted S-praziquantel intermediate (S-1) and converts it back into the racemic form (Compound 1). By closing this material loop, manufacturers can significantly enhance atom economy and reduce the overall cost burden associated with chiral drug synthesis. This technology represents a pivotal shift towards circular chemistry in pharmaceutical intermediate manufacturing, offering a robust solution for maximizing resource utilization without compromising product quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of L-Praziquantel has been plagued by economic and technical hurdles inherent to chiral resolution and asymmetric synthesis strategies. Conventional asymmetric synthesis often relies on expensive chiral catalysts, such as Noyori catalysts, or chiral auxiliaries, which not only drive up raw material costs but frequently suffer from modest yields and optical purities. For instance, literature precedents indicate that asymmetric catalytic reduction steps may yield only around 52% conversion with limited enantiomeric excess, necessitating complex purification protocols. Furthermore, alternative racemization strategies for tetrahydroisoquinoline derivatives have attempted oxidation-reduction sequences. However, these methods are fraught with difficulties; the oxidation step often employs harsh reagents like sodium hypochlorite or metal catalysts that promote unwanted aromatization to isoquinoline derivatives. This side reaction generates difficult-to-remove impurities and lowers the overall recovery yield, rendering such processes economically unviable for large-scale operations. Additionally, traditional acid or base-mediated racemization attempts have proven ineffective for this specific substrate, often leading to decomposition rather than stereoinversion due to the sensitivity of the amide-protected ring system.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the technology described in CN107814797B offers a streamlined, high-efficiency pathway for recycling the S-enantiomer. The core innovation lies in the direct catalytic hydrogenation of the discarded S-praziquantel intermediate (S-1) to regenerate the racemic mixture (Compound 1). 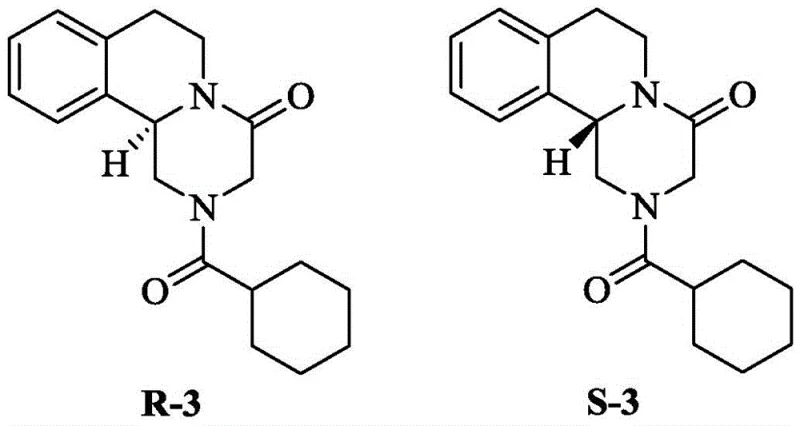 This process bypasses the need for oxidative steps that risk structural degradation. Instead, it utilizes common heterogeneous metal catalysts, such as Palladium on Carbon (Pd/C), Platinum on Carbon (Pt/C), or Raney Nickel, under elevated hydrogen pressure and temperature. The method demonstrates exceptional robustness, achieving near-quantitative yields (up to 99%) with high chemical purity. By transforming a waste byproduct into a valuable feedstock, this approach effectively eliminates the 50% material loss typically associated with resolution processes. The operational simplicity of using standard hydrogenation equipment and readily available solvents like toluene or ethanol further underscores its potential for immediate industrial adoption, providing a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in cost management.
This process bypasses the need for oxidative steps that risk structural degradation. Instead, it utilizes common heterogeneous metal catalysts, such as Palladium on Carbon (Pd/C), Platinum on Carbon (Pt/C), or Raney Nickel, under elevated hydrogen pressure and temperature. The method demonstrates exceptional robustness, achieving near-quantitative yields (up to 99%) with high chemical purity. By transforming a waste byproduct into a valuable feedstock, this approach effectively eliminates the 50% material loss typically associated with resolution processes. The operational simplicity of using standard hydrogenation equipment and readily available solvents like toluene or ethanol further underscores its potential for immediate industrial adoption, providing a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in cost management.
Mechanistic Insights into Catalytic Hydrogenation Racemization
The success of this recovery method hinges on a sophisticated understanding of the catalytic dehydrogenation-hydrogenation equilibrium on the metal surface. Unlike simple acid-base epimerization which fails due to the stability of the chiral center in this specific amide-protected tetrahydroisoquinoline framework, the catalytic route likely proceeds through a reversible dehydrogenation mechanism. Under the applied thermal conditions (130-150°C) and hydrogen pressure, the catalyst facilitates the temporary removal of hydrogen from the nitrogen-adjacent carbon, generating a planar imine or enamine-like intermediate species adsorbed on the metal surface. 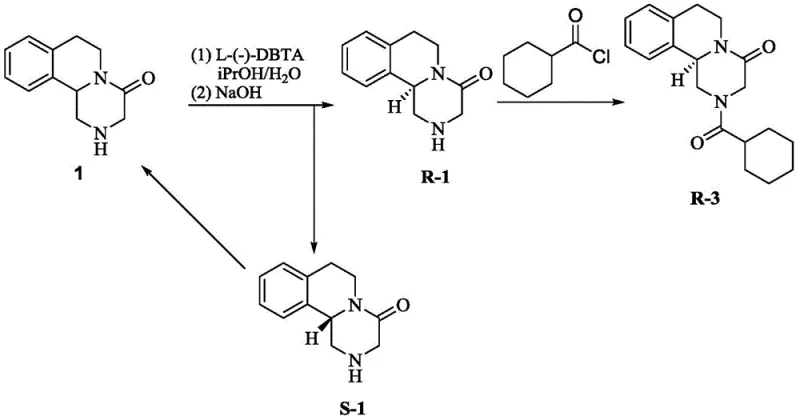 Once planar, the stereochemical information at the chiral center is lost. Subsequent re-addition of hydrogen from the catalyst surface occurs non-stereoselectively, resulting in a statistical 1:1 mixture of R and S configurations. This mechanism is critically dependent on the protection of the secondary amine by the acyl group; as noted in the patent background, unprotected amines might undergo irreversible oxidation or aromatization. The acyl protection group effectively blocks the formation of stable aromatic isoquinoline byproducts, channeling the reaction exclusively towards the desired racemization pathway. This precise control over the reaction trajectory ensures that the recovered racemic intermediate retains the necessary structural integrity for downstream processing, avoiding the complex impurity profiles seen in oxidative racemization attempts.
Once planar, the stereochemical information at the chiral center is lost. Subsequent re-addition of hydrogen from the catalyst surface occurs non-stereoselectively, resulting in a statistical 1:1 mixture of R and S configurations. This mechanism is critically dependent on the protection of the secondary amine by the acyl group; as noted in the patent background, unprotected amines might undergo irreversible oxidation or aromatization. The acyl protection group effectively blocks the formation of stable aromatic isoquinoline byproducts, channeling the reaction exclusively towards the desired racemization pathway. This precise control over the reaction trajectory ensures that the recovered racemic intermediate retains the necessary structural integrity for downstream processing, avoiding the complex impurity profiles seen in oxidative racemization attempts.
Furthermore, the choice of solvent and catalyst plays a pivotal role in suppressing side reactions and maintaining high purity. The patent data highlights that solvents such as toluene, xylene, and alcohols like isopropanol provide an optimal medium for this transformation, balancing solubility with thermal stability. The use of Pd/C is particularly advantageous due to its high activity for hydrogen activation at moderate pressures (10-25 atm). Comparative experiments detailed in the patent demonstrate that attempting racemization with strong acids (HCl, H2SO4) or bases (KOH, NaOCH3) results in either no reaction or significant degradation of the starting material. This confirms that the racemization is strictly catalytic and surface-mediated rather than solution-phase ionic. The ability to achieve 99% yield with 95% HPLC purity in optimized examples indicates that the catalyst does not promote hydrolysis of the amide bond or hydrogenolysis of the benzyl-like positions under these specific conditions. This mechanistic selectivity is crucial for a reliable pharmaceutical intermediate supplier, as it minimizes the need for extensive downstream purification, thereby reducing solvent consumption and waste generation.
How to Synthesize Racemic Praziquantel Intermediate Efficiently
The implementation of this recovery process requires careful attention to reaction parameters to ensure maximum conversion and safety. The general procedure involves dissolving the waste S-praziquantel intermediate in a selected solvent, adding the heterogeneous catalyst, and pressurizing the reactor with hydrogen gas. The reaction is then heated to the specified range to initiate the racemization cycle. Detailed standardized synthetic steps for this process are provided in the guide below.
- Dissolve the waste S-praziquantel intermediate (S-1) in a suitable solvent such as toluene or xylene.
- Add a metal catalyst, preferably Pd/C, Raney-Ni, or Pt/C, to the reaction mixture under an inert atmosphere.
- Heat the mixture to 130-150°C under hydrogen pressure of 10-25 atm for approximately 48 hours to achieve racemization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this recovery technology translates directly into enhanced operational resilience and significant cost optimization. By integrating this recycling loop, manufacturers can effectively double the utility of their initial raw material inputs, as the 'waste' from the resolution step is no longer a disposal cost but a valuable asset. This fundamentally alters the cost structure of L-Praziquantel production, mitigating the impact of fluctuating raw material prices. Moreover, the reliance on commodity catalysts like Pd/C and common solvents eliminates the supply chain risks associated with sourcing specialized, expensive chiral ligands or auxiliaries. The process simplifies the manufacturing workflow by removing the need for complex multi-step oxidative racemization sequences, which often require stringent safety controls and specialized waste treatment for oxidants. Consequently, this leads to a drastic simplification of the production schedule and a reduction in the overall environmental footprint, aligning with modern green chemistry mandates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the elimination of material waste. In traditional resolution processes, nearly half of the synthesized intermediate is discarded, representing a sunk cost in reagents, energy, and labor. By recovering this S-isomer with high efficiency, the effective yield of the entire synthetic sequence is dramatically improved without increasing the input of starting materials. Additionally, the avoidance of expensive chiral catalysts for the racemization step and the use of robust, reusable heterogeneous catalysts further lower the variable costs per kilogram. The high purity of the recovered product also reduces the burden on quality control and purification departments, minimizing solvent usage for recrystallization or chromatography. These factors combine to create a substantially leaner cost profile for the final API, offering a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic chiral reagents can introduce significant vulnerability into the supply chain. This method utilizes widely available industrial chemicals and standard catalysts, diversifying the supply base and reducing the risk of production stoppages due to material shortages. The robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, ensuring consistent output. Furthermore, by internalizing the recovery process, manufacturers gain greater control over their inventory levels of key intermediates. The ability to recycle off-spec or excess S-isomer inventory back into the production line provides a buffer against demand fluctuations, ensuring a steady supply of the racemic precursor needed for resolution. This stability is critical for maintaining long-term contracts with global pharmaceutical partners who require guaranteed continuity of supply.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is highly favorable as it relies on unit operations that are standard in the fine chemical industry, such as high-pressure hydrogenation and filtration. There is no need for specialized equipment for handling hazardous oxidants or cryogenic conditions, which simplifies facility requirements and safety protocols. The use of heterogeneous catalysts allows for easy separation via filtration, facilitating catalyst recovery and reuse, which further enhances sustainability. Environmentally, the process significantly reduces the volume of chemical waste generated, as the S-isomer is converted rather than disposed of. This reduction in waste load lowers the costs associated with hazardous waste treatment and disposal. The alignment with green chemistry principles—specifically atom economy and waste prevention—makes this technology attractive for companies aiming to improve their environmental, social, and governance (ESG) ratings while maintaining high production efficiency.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this recovery technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on the feasibility and benefits of the process for potential adopters.
Q: Why is recovering the S-isomer of praziquantel intermediate critical for cost reduction?
A: Traditional chiral resolution methods discard approximately 50% of the material as the unwanted S-isomer. Recovering this waste stream effectively doubles the yield of usable starting material for subsequent resolution cycles, drastically lowering raw material costs.
Q: What are the advantages of catalytic hydrogenation over acid/base racemization?
A: Conventional acid or base-mediated racemization often leads to decomposition and impurity formation due to harsh conditions. The patented catalytic hydrogenation method operates under controlled thermal and pressure conditions, preserving the structural integrity of the tetrahydroisoquinoline ring while achieving high optical purity conversion.
Q: Is this recovery process scalable for industrial production?
A: Yes, the process utilizes standard heterogeneous catalysts like Pd/C and common solvents like toluene. The reaction conditions (130-150°C, 10-25 atm H2) are compatible with standard high-pressure hydrogenation reactors used in fine chemical manufacturing, ensuring easy scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Praziquantel Intermediate Supplier
The technological advancements detailed in CN107814797B underscore the importance of innovative process chemistry in driving down the cost of essential medicines. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in catalytic processes to deliver superior value to our global partners. As a dedicated CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into industrial reality. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards. We understand that in the competitive landscape of pharmaceutical intermediates, consistency and reliability are paramount, and our infrastructure is designed to deliver exactly that.
We invite you to explore how our advanced manufacturing capabilities can optimize your supply chain for Praziquantel and related antiparasitic agents. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective but also resilient and environmentally responsible, positioning your organization for long-term success in the global market.
