Advanced Catalytic Hydrogenation for High-Purity Aminobenzenesulfonic Acid Manufacturing
Advanced Catalytic Hydrogenation for High-Purity Aminobenzenesulfonic Acid Manufacturing
The global demand for high-purity aromatic amines, particularly aminobenzenesulfonic acids, has necessitated a paradigm shift away from archaic, pollution-heavy synthesis methods toward cleaner, catalytic technologies. Patent CN101362710A introduces a robust catalytic hydrogenation methodology that fundamentally transforms the production landscape for these critical dye and pharmaceutical intermediates. By leveraging hydrogen gas as a clean reducing agent in conjunction with heterogeneous catalysts such as Raney Nickel or active Pd/C, this technology achieves conversion efficiencies that were previously unattainable with traditional stoichiometric reductants. The process operates effectively within a temperature range of 20°C to 180°C and pressures up to 3 MPa, ensuring versatility across various substituted nitrobenzenesulfonic acid substrates. For R&D directors and procurement specialists, this patent represents a viable pathway to secure a reliable aminobenzenesulfonic acid supplier capable of delivering materials with superior impurity profiles.
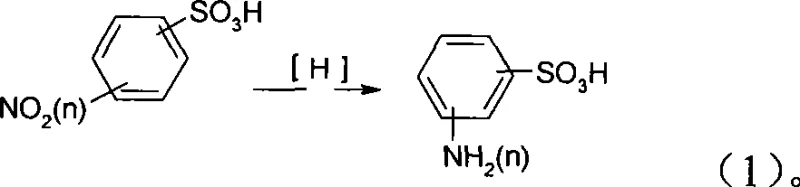
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of aminobenzenesulfonic acids, including meta-sulfanilic acid and ortho-amino phenol sulphonic, has relied heavily on the iron powder reduction method. This legacy technology is fraught with significant operational and environmental deficiencies that render it increasingly obsolete in modern regulatory environments. The core issue lies in the stoichiometric consumption of iron powder, which generates vast quantities of iron mud sludge as a byproduct. This iron mud is not only difficult to filter and separate from the product liquor but also constitutes a severe hazardous waste disposal challenge, driving up operational expenditures related to environmental compliance. Furthermore, the presence of residual iron ions often complicates downstream purification, leading to products with inconsistent color and purity levels that are unacceptable for high-end dye and electronic chemical applications. The batch nature of the iron reduction process, requiring prolonged boiling and multiple filtration steps, inherently limits throughput and scalability.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a catalytic hydrogenation strategy that circumvents the generation of solid inorganic waste entirely. By employing molecular hydrogen over a reusable metal catalyst, the reaction proceeds with atom economy that approaches theoretical limits, converting the nitro group directly to an amino group without introducing extraneous metallic contaminants. This method supports a wide substrate scope, accommodating various isomers such as those depicted in the structural diagrams below, ensuring flexibility for manufacturers producing diverse dye intermediate portfolios. The ability to recover and recycle the catalyst further enhances the economic viability of the process, transforming what was once a linear, waste-generating operation into a circular, sustainable manufacturing loop. This technological leap positions producers to meet the rigorous quality standards required for cost reduction in dye intermediate manufacturing while adhering to strict environmental mandates.
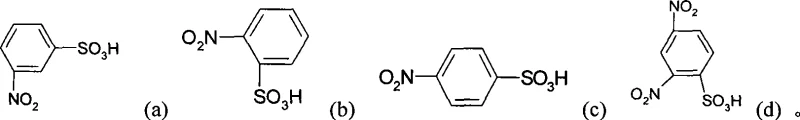
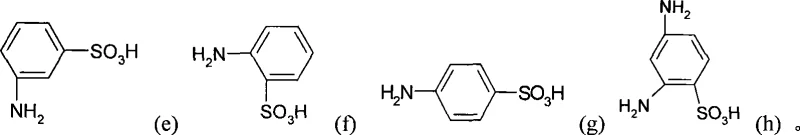
Mechanistic Insights into Catalytic Hydrogenation of Nitro Groups
The core of this technological advancement lies in the heterogeneous catalysis mechanism occurring on the surface of the Raney Nickel or Pd/C particles. During the reaction, hydrogen molecules adsorb onto the active metal sites and dissociate into atomic hydrogen, which is highly reactive. Simultaneously, the nitrobenzenesulfonic acid substrate adsorbs onto the catalyst surface, orienting the nitro group for sequential reduction. The transformation proceeds through several intermediate stages, typically involving nitroso and hydroxylamine species, before finally yielding the stable amine functionality. The choice of solvent, such as methanol or ethanol as exemplified in the patent data, plays a critical role in solubilizing the sulfonic acid substrate and facilitating mass transfer to the catalyst surface. Understanding this surface phenomenon is crucial for R&D teams aiming to optimize reaction kinetics and minimize the formation of azo or hydrazo coupling byproducts, which can occur if the reaction conditions are not tightly controlled.
Impurity control in this system is inherently superior to iron reduction due to the absence of competing redox reactions with metal powders. In the traditional iron method, over-reduction or side reactions with iron salts can lead to complex impurity profiles that are difficult to remove. In the catalytic hydrogenation regime, selectivity is governed primarily by the catalyst type and reaction parameters like temperature and pressure. For instance, maintaining the pressure between 0.2 MPa and 3 MPa ensures sufficient hydrogen availability to drive the reaction to completion without promoting excessive hydrogenolysis of other sensitive functional groups that might be present on the aromatic ring. The result is a product stream with significantly higher chromatographic purity, often exceeding 95% content as demonstrated in the patent examples, thereby reducing the burden on downstream crystallization and purification units.
How to Synthesize Aminobenzenesulfonic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from laboratory scale to commercial production. The process begins with the charging of the nitrobenzenesulfonic acid raw material and an appropriate organic solvent into a high-pressure hydrogenation reactor. Following the addition of the catalyst, typically at a loading of 0.5% to 15% relative to the substrate, the system undergoes a rigorous purging sequence to ensure safety and reaction efficiency. Detailed standard operating procedures regarding temperature ramping, pressure maintenance, and endpoint determination are critical for reproducibility. For a comprehensive breakdown of the specific operational parameters and safety protocols required for implementation, please refer to the standardized synthesis guide below.
- Load nitrobenzenesulfonic acid and organic solvent (methanol/ethanol) into a high-pressure reactor with Raney Nickel or Pd/C catalyst.
- Replace air with nitrogen, then exchange with hydrogen three times, and pressurize to 0.2-3 MPa at 20-180°C.
- React for 2-10 hours until hydrogen absorption ceases, then filter catalyst and isolate the product via acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic hydrogenation technology offers profound strategic advantages beyond mere technical superiority. The elimination of iron mud waste translates directly into substantial cost savings by removing the need for expensive sludge treatment and disposal services, which have become increasingly costly under tightening environmental regulations. Furthermore, the simplified workflow reduces the number of unit operations required, shortening the overall production cycle time and enhancing facility throughput. This efficiency gain allows suppliers to respond more agilely to market fluctuations, ensuring a more reliable supply of high-purity aminobenzenesulfonic acid for downstream customers. The ability to recycle the catalyst also mitigates exposure to volatile raw material costs associated with stoichiometric reductants, stabilizing the long-term cost structure of the manufacturing process.
- Cost Reduction in Manufacturing: The shift from stoichiometric iron powder to catalytic hydrogenation fundamentally alters the cost basis of production by eliminating the purchase of tons of iron powder and the associated disposal fees for iron mud. Since the catalyst can be recovered and reused multiple times without significant loss of activity, the variable cost per kilogram of product is drastically reduced. Additionally, the higher yield and purity reduce the loss of valuable raw materials during purification, maximizing the return on investment for every batch produced. These factors combine to create a leaner, more cost-effective manufacturing model that protects margins even in competitive markets.
- Enhanced Supply Chain Reliability: Traditional iron powder methods are often bottlenecked by waste disposal capacity and filtration limitations, leading to unpredictable production delays. The catalytic process, being cleaner and faster, decouples production rates from waste handling constraints, allowing for more consistent and predictable output schedules. This reliability is critical for customers who depend on just-in-time delivery of dye intermediates to maintain their own production lines. By adopting this technology, suppliers can offer more robust service level agreements and reduce the risk of supply disruptions caused by environmental shutdowns or equipment fouling.
- Scalability and Environmental Compliance: Scaling up the catalytic hydrogenation process is straightforward, as it relies on standard high-pressure reactor technology widely available in the fine chemical industry. Unlike the iron powder method, which faces severe scaling challenges due to heat management and sludge handling, the hydrogenation route scales linearly with reactor volume. Moreover, the process generates no hazardous solid waste, aligning perfectly with green chemistry principles and ensuring long-term compliance with global environmental standards. This future-proofs the supply chain against regulatory shocks and enhances the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this catalytic hydrogenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing supply chains or R&D pipelines. The answers reflect the specific advantages of using Raney Nickel or Pd/C catalysts under the defined pressure and temperature conditions.
Q: What are the primary advantages of catalytic hydrogenation over iron powder reduction for sulfanilic acid?
A: The catalytic hydrogenation method described in CN101362710A eliminates the generation of massive amounts of iron mud waste, significantly reducing environmental treatment costs and improving product purity compared to traditional iron powder reduction.
Q: Which catalysts are suitable for this hydrogenation process?
A: The patent specifies the use of Raney Nickel (Ni-Al or Ni-Zn types) or active Palladium on Carbon (Pd/C) as effective catalysts, with loadings ranging from 0.5% to 15% of the raw material weight.
Q: What represent the typical reaction conditions for this synthesis?
A: Optimal reaction conditions involve temperatures between 20°C and 180°C and hydrogen pressures ranging from 0.2 to 3 MPa, typically completing within 2 to 10 hours depending on the specific substrate and catalyst activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminobenzenesulfonic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced catalytic processes is key to securing a competitive edge in the global fine chemicals market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN101362710A are fully realized in practical, large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aminobenzenesulfonic acid meets the exacting requirements of the dye and pharmaceutical industries. Our commitment to technological excellence ensures that our clients receive materials that facilitate their own downstream processing efficiency and product quality.
We invite you to engage with our technical procurement team to discuss how this innovative hydrogenation route can be tailored to your specific volume and quality needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our catalytically produced intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize your supply chain performance and reduce overall manufacturing costs.
