Advanced Catalytic Hydrogenation Route for High-Purity Para Aminoacet Anilide Production
Advanced Catalytic Hydrogenation Route for High-Purity Para Aminoacet Anilide Production
The chemical industry is constantly seeking more sustainable and efficient pathways for producing critical intermediates, and the technology disclosed in patent CN103787908A represents a significant leap forward in the synthesis of para aminoacet anilide. This compound serves as a vital building block in the manufacture of dispersed dyes and other specialized colorants, where purity and consistency are paramount for downstream performance. Traditional manufacturing routes have long been plagued by severe environmental liabilities and complex waste management issues, but this novel approach utilizes a streamlined two-step process involving direct acylation followed by catalytic hydrogenation. By shifting away from archaic reduction methods, this technology not only enhances the ecological footprint of production but also delivers superior yields and product quality that meet the rigorous standards of modern pharmaceutical and agrochemical supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of para aminoacet anilide has relied heavily on the nitration of acetanilide followed by reduction using iron powder in the presence of strong acids. This legacy process is fraught with significant operational and environmental drawbacks that modern manufacturers can no longer ignore. The use of iron powder generates vast quantities of solid iron mud sludge, which is difficult to dispose of and poses a serious contamination risk to soil and groundwater if not managed perfectly. Furthermore, the reaction conditions require large volumes of acidic media, resulting in highly corrosive wastewater that demands expensive neutralization and treatment facilities before discharge. These factors collectively drive up the operational expenditure and create substantial regulatory hurdles for facilities attempting to maintain compliance with increasingly stringent global environmental protection laws.
The Novel Approach
In stark contrast, the methodology outlined in the referenced patent introduces a cleaner and more atom-economical route that begins with p-nitroaniline as the starting material. Instead of generating hazardous sludge, the process employs glacial acetic acid for the initial acylation step, creating a reaction system where the byproduct is primarily water, which is easily removed via distillation to drive the equilibrium forward. The subsequent reduction step utilizes catalytic hydrogenation rather than stoichiometric metal reductants, effectively eliminating the generation of heavy metal waste streams entirely. This transition to a catalytic regime not only simplifies the post-reaction workup but also allows for the recovery and reuse of the acetic acid filtrate, thereby closing the loop on material usage and significantly reducing the overall consumption of raw materials per kilogram of finished product.
Mechanistic Insights into Acylation and Catalytic Hydrogenation
The first stage of this synthesis involves the nucleophilic acyl substitution where the amino group of p-nitroaniline attacks the carbonyl carbon of glacial acetic acid. This reaction is conducted under reflux conditions at temperatures ranging from 120°C to 130°C, which provides sufficient thermal energy to overcome the activation barrier while maintaining the reaction mixture in a liquid state. A critical aspect of this mechanism is the continuous removal of water formed during the condensation; by distilling off the water azeotrope, the reaction equilibrium is shifted decisively towards the formation of paranitroacetanilide, ensuring high conversion rates without the need for excessive excesses of reagents. The resulting intermediate precipitates upon cooling, allowing for simple filtration and washing, which inherently purifies the substance before it even enters the reduction phase.
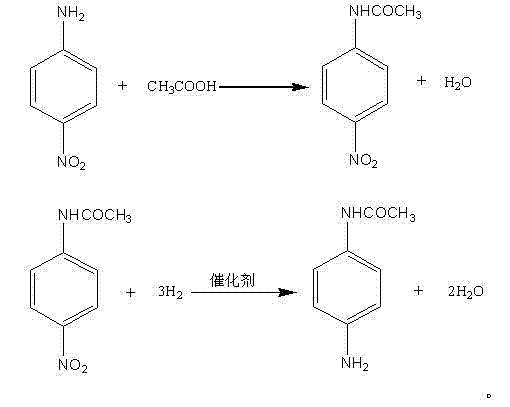
The second stage leverages the power of heterogeneous catalysis to reduce the nitro group to an amino group with exceptional selectivity. Using catalysts such as palladium on carbon or Raney nickel, hydrogen gas is activated on the metal surface and transferred to the nitro functionality of the paranitroacetanilide substrate. This process occurs under moderate pressures of 0.5 to 1.5 MPa and temperatures between 40°C and 100°C, conditions that are mild enough to prevent the hydrolysis of the acetamide protecting group, which is a common side reaction in harsher acidic or basic reduction environments. The preservation of the acetamide group throughout the reduction is crucial, as it avoids the need for a re-protection step, thereby shortening the synthetic sequence and minimizing the formation of deacetylated impurities that would otherwise complicate the final purification.
How to Synthesize Para Aminoacet Anilide Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and safety, particularly during the high-pressure hydrogenation step. The process begins with the careful mixing of p-nitroaniline and glacial acetic acid in a molar ratio optimized between 1:4 and 1:8 to ensure complete dissolution and reaction kinetics. Following the acylation and isolation of the intermediate, the material is charged into an autoclave with the chosen catalyst and a solvent such as methanol or isopropanol. It is imperative to purge the system thoroughly with nitrogen and hydrogen to remove oxygen before pressurizing, ensuring a safe environment for the exothermic reduction reaction to proceed. For a detailed breakdown of the specific operational parameters and safety protocols required for this transformation, please refer to the standardized synthesis guide below.
- Acylation: React p-nitroaniline with glacial acetic acid under reflux at 120-130°C to form paranitroacetanilide, removing water via distillation.
- Hydrogenation: Reduce paranitroacetanilide using hydrogen gas (0.5-1.5 MPa) and a palladium carbon or Raney nickel catalyst in an organic solvent.
- Purification: Filter the reaction mixture, recover the solvent, and recrystallize the crude product to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic hydrogenation technology translates directly into tangible improvements in cost structure and supply reliability. By eliminating the need for iron powder and the associated waste disposal fees, manufacturers can achieve a significant reduction in the variable costs associated with production. The ability to recycle the acetic acid filtrate further compounds these savings, reducing the dependency on fluctuating raw material markets and insulating the supply chain from price volatility. Moreover, the simplified workflow reduces the total processing time and labor hours required per batch, enhancing the overall throughput capacity of existing manufacturing assets without the need for major capital expansion.
- Cost Reduction in Manufacturing: The elimination of stoichiometric metal reductants removes a major cost center associated with both the purchase of iron powder and the disposal of hazardous iron sludge. Additionally, the recycling of the acetic acid mother liquor drastically cuts down on solvent consumption, leading to substantial operational savings that improve the gross margin profile of the final product. This efficiency gain allows suppliers to offer more competitive pricing structures while maintaining healthy profitability margins in a tight market.
- Enhanced Supply Chain Reliability: Because the process relies on widely available commodity chemicals like p-nitroaniline and glacial acetic acid, the risk of supply disruption due to specialty reagent shortages is minimized. The robustness of the catalytic hydrogenation step ensures consistent batch-to-batch quality, reducing the likelihood of production delays caused by out-of-specification material that requires reprocessing or scrapping. This stability is critical for downstream customers who rely on just-in-time delivery models for their own dye manufacturing operations.
- Scalability and Environmental Compliance: The technology is inherently scalable, utilizing standard high-pressure reactors that are common in fine chemical plants, facilitating a smooth transition from pilot scale to multi-ton commercial production. Furthermore, the drastic reduction in wastewater toxicity and solid waste generation simplifies the permitting process and ensures long-term compliance with evolving environmental regulations, securing the social license to operate for manufacturing facilities in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of para aminoacet anilide using this advanced catalytic method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this intermediate into their broader supply networks and product formulations.
Q: What are the environmental advantages of this synthesis method compared to traditional iron powder reduction?
A: Unlike traditional methods that generate massive amounts of acidic wastewater and solid iron mud sludge, this catalytic hydrogenation route produces minimal waste. The acetic acid filtrate can be recycled, and the process eliminates the need for hazardous iron powder handling and disposal.
Q: What catalysts are suitable for the reduction step in this patent?
A: The patent specifies the use of heterogeneous catalysts such as palladium carbon (Pd/C) or Raney nickel. These catalysts facilitate the efficient reduction of the nitro group to an amino group under moderate hydrogen pressure (0.5-1.5 MPa) and temperatures between 40-100°C.
Q: How is high purity (99.8%) achieved in the final product?
A: High purity is achieved through a combination of selective catalytic hydrogenation which minimizes side reactions, followed by a rigorous purification step. The crude product is dissolved in a solvent like methanol or ethanol, heated, cooled to crystallize, and then vacuum dried to remove residual impurities and solvents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Para Aminoacet Anilide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of our clients' final products, which is why we have invested heavily in mastering advanced synthetic routes like the one described in patent CN103787908A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every shipment of para aminoacet anilide meets the exacting standards required for high-performance dye applications, giving you confidence in your supply chain.
We invite you to contact our technical procurement team to discuss how we can tailor our production capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized processes can lower your total landed costs. We are ready to provide specific COA data and route feasibility assessments to demonstrate our commitment to being your trusted partner in the global fine chemicals market.
